WO2010022897A2 - Methoden zur verbesserung des pflanzenwachstums - Google Patents
Methoden zur verbesserung des pflanzenwachstums Download PDFInfo
- Publication number
- WO2010022897A2 WO2010022897A2 PCT/EP2009/006064 EP2009006064W WO2010022897A2 WO 2010022897 A2 WO2010022897 A2 WO 2010022897A2 EP 2009006064 W EP2009006064 W EP 2009006064W WO 2010022897 A2 WO2010022897 A2 WO 2010022897A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- methyl
- plants
- cyanamide
- oxido
- sulfanylidene
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 *C(CCC1)S1(=N*)=O Chemical compound *C(CCC1)S1(=N*)=O 0.000 description 1
- DHLJVFUXQAPJBB-UHFFFAOYSA-N CC(c1ccc(C(C(F)(F)F)(F)F)nc1)S(C)(=NC#N)=O Chemical compound CC(c1ccc(C(C(F)(F)F)(F)F)nc1)S(C)(=NC#N)=O DHLJVFUXQAPJBB-UHFFFAOYSA-N 0.000 description 1
- OJTNEKSDKJYDMH-UHFFFAOYSA-N CC(c1ccc(C(Cl)(Cl)Cl)nc1)S(C)(=NC#N)=O Chemical compound CC(c1ccc(C(Cl)(Cl)Cl)nc1)S(C)(=NC#N)=O OJTNEKSDKJYDMH-UHFFFAOYSA-N 0.000 description 1
- MIEDBILRKQWLQR-UHFFFAOYSA-N N#CN=S1(C(Cc2cnc(C(F)(F)F)[s]2)CCC1)=O Chemical compound N#CN=S1(C(Cc2cnc(C(F)(F)F)[s]2)CCC1)=O MIEDBILRKQWLQR-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/40—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having a double or triple bond to nitrogen, e.g. cyanates, cyanamides
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/34—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom
- A01N43/40—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom six-membered rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/74—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms five-membered rings with one nitrogen atom and either one oxygen atom or one sulfur atom in positions 1,3
- A01N43/78—1,3-Thiazoles; Hydrogenated 1,3-thiazoles
Definitions
- the invention relates to processes using sulfoximines to increase plant's own defenses and / or to improve plant growth and / or to increase the resistance of plants to plant diseases caused by fungi, bacteria, viruses, Mycoplasma-like organisms and or RLO (Rickettsia-like organisms) are suitable.
- plants react to natural stress conditions, such as, for example, cold, heat, dryness, wounding, pathogen infestation (viruses, bacteria, fungi), insects, etc., but also to herbicides with specific or nonspecific defense mechanisms (plant biochemistry, p. 393 -462, Spektrum Akademischer Verlag, Heidelberg, Berlin, Oxford, Hans W. Heldt, 1996. Biochemistry and Molecular Biology of Plants, pp. 1102-1203, American Society of Plant Physiologists, Rockville, Maryland, eds. Buchanan, Gruissem, Jones, 2000). It serve z. B.
- enzymatic proteins such as chitinases, glucanases, or (d) specific inhibitors of essential proteins, such as protease inhibitors, xylanase inhibitors, which act as "proteinaceous proteins" (PR proteins) attack the pathogen directly or impede its proliferation (Dangl and Jones, Nature 411, 826-833, 2001; Kessler and Baldwin, Annual Review of Plant Biology, 53, 299-328, 2003).
- HR hypersensitive reaction
- a known synthetic product which assumes one of the salicylic acid similar function and can provide a protective effect against phytopathogenic fungi, bacteria and viruses, is benzothiadiazole (CGA 245704; Common name: acibenzolar-S-methyl; trade name: Bion ®) (Achuo et al. , Plant Pathology 53 (1), 65-72, 2004, Tamblyn et al., Pesticide Science 55 (6), 676-677, 1999, EP-OS 0 313 512).
- pathogenesis-related proteins which primarily support plants in the defense against biotic stressors, such as phytopathogens Fungi, bacteria and viruses (DE 10 2005 045 174 A; DE 10 2005 022 994 A and WO 2006/122662 A; Thielert Maischutz- sympatheticen Bayer, 59 (1), 73-86, 2006; Francis et al., European Journal of Plant Pathology, publ. online 23.1.2009).
- PR proteins pathogenesis-related proteins
- plants have a plurality of endogenous reaction mechanisms which can effect effective defense against various harmful organisms (biotic stress) and / or abiotic stress.
- the seed is sown in special containers, eg Styrofoam perforated trays, in special potting soil based on peat culture substrate and then cultivated in containers with suitable nutrient solution until the desired transplant size is reached (Figure 1). It is allowed to drive the containers on the nutrient solution, from which derives the name of the culture method (Leal, 2001, supra).
- insecticides from the class of neonicotinoids chlornicotinyls
- the plants are usually sprayed with neonicotinoid (chloronicotinyl) insecticides in the float process shortly before transplantation or are applied to the field with neonicotinoid (chloronicotinyl) insecticides immediately before or during transplantation.
- neonicotinoid chloronicotinyl
- corona neonicotinoid
- Fungicides and insecticides are used to protect the crops of seed or plant material from fungal pathogens and pests until they are transplanted.
- the choice of pesticides, the place and time of application and the application rate of the funds depend here mainly on the type of fungal diseases and pests occurring, the specific mode of action and duration of action of the agents and their plant compatibility, and thus directly to the specific requirements of different Adapted to crops and regions.
- Sulfoximines have become known, for example, as agents for combating animal pests, in particular insects (for example, US Patent Application 2005/228027 A1, WO 2006/060029 A2, WO 2007/095229 A2, WO 2007/149134 A1, WO 2008 / 027539 Al, WO 2008/027073 Al and WO 2008/097235 Al). Furthermore, the insecticidal increase in activity for a subgroup of sulfoximines is described by the addition of suitable salts and optionally additives (WO 2007/068355).
- the sulfoximines lead to a good protection of the plant from damage by fungal, bacterial or viral pathogens. Without wishing to be bound by theory, it is currently believed that defense of the pathogens by induction of PR proteins occurs as a result of treatment with at least one sulfoximine.
- the use according to the invention in seed treatment, in soil treatment, in special cultivation and cultivation processes eg floating box, Rockwool, Hydroponic
- stem and leaf treatment has the advantages described.
- Combinations of sulfoximines with, inter alia, insecticides, fungicides and bactericides show a synergistic effect in the control of plant diseases.
- the combined use of sulfoximines with genetically modified varieties for increased abiotic stress tolerance results in a synergistic improvement in growth.
- sulfoxic acids not only enhance the pathogen defense in plants, but also improve plant growth and / or increase the resistance of plants to plant diseases caused by fungi, bacteria, viruses, Mycoplasma-like organisms. and / or RLO (rickettsia-like organisms), in particular to soil-borne fungal diseases, and / or to increase the resistance of plants to abiotic stresses.
- Abiotic stress conditions may include, for example, drought, cold and heat conditions, osmotic stress, waterlogging, increased soil salinity, increased exposure to minerals, ozone conditions, high light conditions, limited availability of nitrogen nutrients, limited availability of phosphorous nutrients, or avoidance of shade.
- the present invention therefore relates firstly to the use of at least one compound selected from the class of sulfoximines for increasing plant-specific defense forces and / or for improving plant growth and / or for increasing the resistance of plants to plant diseases caused by fungi, Bacteria, viruses, MLO (Mycoplasma -like organisms) and / or RLO (Rickettsia-like organisms) are caused, especially against soil-borne fungal diseases, and / or to increase the resistance of plants to abiotic stress factors.
- MLO Mycoplasma -like organisms
- RLO Rosia-like organisms
- X is NO 2 , CN or COOR 4 ,
- R 1 is C 1 -C 4 -alkyl
- R 1 , sulfur and L together represent a 4-, 5- or 6-membered ring
- R 2 and R 3 independently represent hydrogen, methyl, ethyl, fluorine, chlorine or bromine stand, or
- R 2 and R 3 together represent - (CH 2 ⁇ -, - (CH 2 ) 3 -, - (CH 2 ) 4 - or - (CH 2 ) S - and with the carbon atom to which they are attached form a Form 4-, 5- or 6-ring,
- n 0, 1, 2 or 3
- Z represents halogen, Ci-C4-alkyl, Ci-C4-haloalkyl, Ci-C 4 -alkoxy or C r C 4 haloalkoxy, and
- R 4 is d-Cj-alkyl.
- the compounds of the formula (I) can also be present in different compositions as optical isomers or mixtures of isomers, which can optionally be separated in a customary manner. Both the pure isomers and the isomer mixtures, their use and compositions containing them are the subject of the present invention. In the following, however, for the sake of simplicity, reference is always made to compounds of the formula (I), although both the pure compounds and, if appropriate, mixtures with different proportions of isomeric compounds are meant.
- R 1 , sulfur, and L together form a 5-membered ring
- X is NO 2 or CN
- Y is 6-halo-pyrid-3-yl or 6- (C 1 -C 4 -haloalkyl) -pyrid-3-yl, particularly preferred for 6-chloropyrid-3-yl or 6-trifluoromethyl-pyrid-3-yl
- n is preferably O:
- R 1 , sulfur, and L together form a 5-membered ring
- X is NO 2 or CN
- Y is 6-halo-pyrid-3-yl or 6- (C 1 -C 4 -haloalkyl) -pyrid-3-yl, particularly preferably 6-chloropyrid-3-yl or 6-trifluoromethyl-pyrid-3-yl
- n is preferably 0:
- R 1 is methyl
- X is NO 2 or CN
- L is a single bond and n is preferably 1:
- R 1 is methyl
- R 2 and R 3 independently of one another, are hydrogen or methyl
- X is NO 2 or CN
- n is preferably 1:
- R 1 is methyl
- R 2 and R 3 together are - (CH 2 ) 2 - and form a 3-membered ring with the carbon atom to which they are bonded
- X is NO 2 or CN
- n is preferably 1:
- the compounds of the general formula (I) can, if appropriate, depending on the nature of the substituents, be present as geometrical and / or as optically active isomers or corresponding isomer mixtures in different compositions.
- the invention relates to both the pure isomers and the isomer mixtures.
- sulfoximines When sulfoximines are used in the context of the present invention, these are generally sulfoximines of the general formula (I), where the general formula (I) includes in particular the compounds of the groups (Ia) to (Dc) in particular, the compounds of the general formulas (I-1) to (1-23) can be subsumed.
- the sulfoximines cause an activity according to the invention for plant growth.
- plant growth is understood to mean various advantages for plants which are not directly linked to the known pesticidal activity, preferably the insecticidal activity of the sulfoximines, in particular the sulfoximines of the general formula (I).
- Such advantageous properties are, for example, the improved plant characteristics mentioned below: accelerated germination and emergence of the seed and planting material, improved rooting in terms of surface area and depth, increased tailing or tillering, stronger and more productive shoots and tillers, improvement in shoot growth, increased stability, increased size Shoot base diameter, increased leaf area, greener leaf color, higher yields Nutrients and ingredients such as carbohydrates, fats, oils, proteins, vitamins, minerals, essential oils, dyes, fibers, better fiber quality, earlier flowering, increased number of flowers, reduced content of toxic products such as mycotoxins, reduced levels of residues or unfavorable components of any kind or better digestibility, improved storage stability of the crop, improved tolerance to unfavorable temperatures, improved tolerance to drought and drought, as well as lack of oxygen due to excess water, improved tolerance to increased salt levels in soils and water, increased tolerance to UV radiation, increased tolerance to ozone stress, improved tolerance to herbicides and other plant treatment agents, improved water uptake and photosynthetic performance, beneficial plant properties, such as acceleration of maturation,
- Soche terms are, for example, the following: phytotonic effect, resistance to stress factors, less plant stress, plant health, healthy plants, plant fitness, plant wellness, plant concept, vigor effect, Stress Shield, Protective Shield, Crop Health, Crop Health Properties, Crop Health Products, Crop Health Management, Crop Health Therapy, Plant Health, Plant Health Properties, Plant Health Products "Plant Health Management”, “Plant Health Therapy”, “Greening Effect” or “Greening Effect”, “Freshness” or other terms that are well known to a person skilled in the art.
- At least one leaf area increased by generally 5%, in particular 10%, particularly preferably 15%, especially 20%,
- Fertilizers which can be used according to the invention together with the sulfoximines explained in more detail above are in general organic and inorganic nitrogen-containing compounds such as, for example, ureas, urea-formaldehyde condensation products, amino acids, ammonium salts and nitrates, potassium salts (preferably chlorides, sulfates, nitrates ), Phosphoric acid salts and / or salts of phosphorous acid (preferably potassium salts and ammonium salts).
- NPK fertilizers ie fertilizers containing nitrogen, phosphorus and potassium, calcium ammonium nitrate, ie fertilizers that still contain calcium, ammonium sulfate nitrate (general formula (NKU) 2 SO 4 NH 4 NO 3 ), ammonphos - phat and ammonium sulfate.
- NKU ammonium sulfate nitrate
- the fertilizers may also contain salts of micronutrients (preferably calcium, sulfur, boron, manganese, magnesium, iron, boron, copper, zinc, molybdenum and cobalt) and phytohormones (eg, vitamin B1 and indol-3-ylacetic acid (IAA)). or mixtures thereof.
- Fertilizers used according to the invention may also contain other salts such as monoammonium phosphate (MAP), diamonium phosphate (DAP), potassium sulfate, potassium chloride or magnesium sulfate.
- MAP monoammonium phosphate
- DAP diamonium phosphate
- potassium sulfate potassium chloride or magnesium sulfate.
- Suitable amounts for the secondary nutrients or trace elements are amounts of 0.5 to 5 wt .-%, based on the total fertilizer.
- Other possible ingredients are crop protection products, Insecticides or fungicides, growth regulators or mixtures thereof. Further details follow below.
- the fertilizers can be used, for example, in the form of powders, granules, prills or compactates. However, the fertilizers can also be used in liquid form dissolved in an aqueous medium. In this case, dilute aqueous ammonia can be used as nitrogen fertilizer. Further possible ingredients for fertilizers are described, for example, in Ullmann's Encyclopedia of Industrial Chemistry, 5th Edition, 1987, Volume A 10, pages 363 to 401, DE-A 41 28 828, DE-A 19 05 834 and DE-A 196 31 764 ,
- the general composition of the fertilizers which in the context of the present invention may be single nutrient and / or multi-nutrient fertilizers, for example nitrogen, potassium or phosphorus, may vary within a wide range.
- a content of 1 to 30 wt .-% of nitrogen preferably 5 to 20 wt .-%), from 1 to 20 wt .-% potassium (preferably 3 to 15 wt .-%) and a content of 1 to 20% by weight of phosphorus (preferably 3 to 10% by weight) is advantageous.
- the content of microelements is usually in the ppm range, preferably in the range of 1 to 1000 ppm.
- the application is carried out in a functional context, in particular within a period of generally 24 hours, preferably 18 hours, more preferably 12 hours, especially 6 hours, more particularly 4 hours, even more special within 2 hours.
- the active compounds of the general formula (I) and of the fertilizer according to the invention are used in a time frame of less than 1 hour, preferably less than 30 minutes, more preferably less than 15 minutes.
- dimensionally stable mixtures for example in the form of rods, granules, tablets, etc., starting from at least one active ingredient to be used according to the invention and at least one fertilizer.
- the corresponding constituents can be mixed with one another and optionally extruded or the at least one active compound of the general formula (I) to be used according to the invention can be applied to the fertilizer.
- formulation auxiliaries in the dimensionally stable mixtures such as Extender or pressure-sensitive adhesive, used to achieve dimensional stability of the resulting mixture.
- corresponding mixtures are suitable, in particular for use in the "Home &Garden” sector, ie for a private user or home gardener, who can use the dimensionally stable mixture or the constituents contained therein with a predetermined, clearly defined amount and without special aids ,
- the mixtures of at least one of the active ingredients to be used according to the invention and the at least one fertilizer may also be liquid, so that-for example, in the case of a professional user in the field of agriculture-the resulting mixture can be applied as a so-called tank solution.
- the active compounds to be used according to the invention can, if appropriate in combination with fertilizers, preferably be applied to the following plants, the following listing not being restrictive.
- the forest stock includes trees for the production of wood, pulp, paper and products made from parts of the trees.
- crops refers to crops used as plants for the production of food, feed, fuel or for technical purposes.
- count z Among the crops that can be improved with the method according to the invention, count z.
- the following plant species turf, vines, cereals, for example wheat, barley, rye, oats, rice, maize and millet; Beets, for example sugar beets and fodder beets; Fruits, such as pome fruit, stone fruit and soft fruit, such as apples, pears, plums, peaches, almonds, cherries and berries, eg.
- Strawberries, raspberries, blackberries Legumes, such as beans, lentils, peas and soybeans; Oil crops such as rapeseed, mustard, poppy, olive, sunflower, coconut, castor oil, cocoa beans and peanuts; Cucurbits, for example squash, cucumbers and melons; Fiber plants, for example cotton, flax, hemp and jute; Citrus fruits, such as oranges, lemons, grapefruit and mandarins.
- Vegetables such as spinach, (head) salad, asparagus, cabbages, carrots, onions, tomatoes, potatoes and peppers
- Vegetables such as spinach, (head) salad, asparagus, cabbages, carrots, onions, tomatoes, potatoes and peppers
- Laurel family for example avocado, cinnamomum, camphor or also plants such as tobacco, nuts, coffee, eggplant, sugar cane, tea, pepper, vines, hops, bananas, natural rubber plants and ornamentals, for example flowers, shrubs, deciduous trees and conifers. This list is not a limitation.
- Particularly suitable target crops for the application of the method according to the invention are the following plants: bamboo wool, eggplant, turf, pome fruit, stone fruit, berry fruit, corn, wheat, barley, cucumber, tobacco, vines, rice, cereals, pear, beans, soybeans, rapeseed , Tomato, paprika, melons, cabbage, potato and apple.
- Examples of trees that can be improved according to the method of the invention are: Abies sp., Eucalyptus sp., Picea sp., Pinus sp., Aesculus sp., Platanus sp., Tilia sp., Acer sp., Tsuga sp , Fraxinus sp., Sorbus sp., Betula sp., Crataegus sp., Ulmus sp., Quercus sp., Fagus sp., Salix sp., Populus sp.
- Preferred trees which can be improved according to the method according to the invention may be mentioned: From the tree species Aesculus: A. hippocastanum, A. pariflora, A. carnea; from the tree species Platanus: P. aceriflora, P. occidentalis, P. racemosa; from the tree species Picea: P. abies; from the species Pinus: P. radiate, P. ponderosa, P. contorta, P. sylvestre, P. elliottii, P. montecola, P. albicaulis, P. resinosa, P. palustris, P. taeda, P. flexilis, P jeffregi, P. baksiana, P. strobes; from the tree species Eucalyptus: E. grandis, E. globulus, E. camadentis, E. nitens, E. obliqua, E. regnans, E. pilularus.
- trees which can be improved according to the method of the invention, may be mentioned: From the species Pinus: P. radiate, P. ponderosa, P. contorta, P. sylvestre, P. strobes; from the tree species Eucalyptus: E. grandis, E. globulus, E. camadentis.
- the present invention may also be practiced on any turfgrasses, including "cool season turfgrasses” and “warm season turfgrasses.”
- grass species for the cold season are blue grasses (Poa spp.). such as “Kentucky bluegrass” (Poa pratensis L.), “rough bluegrass” (Poa trivialis L.), “Canada bluegrass” (Poa compressa L.), “annual bluegrass” (Poa annua L.), “upland bluegrass” (Poa glaucantha Gaudin), "wood bluegrass “(Poa nemoralis L.) and” bulbous bluegrass “(Poa bulbosa L.); Straussgräser f “Bentgrass”, Agrostis spp.), Such as “creeping bentgrass” (Agrostis palustris Huds.), “Colonial bentgrass” (Agrostis tenuis Sibth.), “Velvet bentgrass” (Agro
- Fescue Fescues, Festucu spp.
- red fescue Festuca rubra L. spp. Rubra
- creeping fescue Festuca rubra L.
- chewings fescue Festuca rubra commutata Gaud.
- sheep fescue (Festuca ovina L.)
- hard fescue (Festuca longifolia Thuill.)
- hair fescue (Festucu capillata Lam.)
- tall fescue (Festuca arundinacea Schreb.) and” meadow fescue "(Festuca elanor L.);
- Lolium ryegrasses, Lolium spp.
- wheat grasses such as "wheatgrasses", Agropyron spp.), such as “fairway wheatgrass” (Agropyron cristatum (L.) Gaertn.), “crested wheatgrass” (Agropyron desertorum (fish.) Schult.) and “western wheatgrass”. (Agropyron smithii Rydb.).
- Examples of other "cool season turfgrasses” are “beachgrass” (Ammophila breviligulata Fern.), “Smooth bromegrass” (Bromus inermis leyss.), Reeds ("cattails") such as “Timothy” (Phleum pratensis L.), “ sand cattail “(Phleum subulatum L.),” orchardgrass “(Dactylis glomerata L.),” weeping alkaligrass “(Puccinellia distans (L.) Pari.) and” crested dog's-tail “(Cynosurus cristatus L.).
- Examples of “warm season turfgrasses” are “3ermudagrass” (Cynodon spp., LC Rieh), “zoysia-grass” (Zoysia spp., Willd.), “St. Augustine grass” (Stenotaphrum lakeundatum Walt Kuntze), "tippedegrass”.
- sulfoximines of the general formula (I) lead to an increased expression of genes from the series of "pathogenesis-related proteins" (PR proteins) .
- PRO proteins primarily support the plants in the defense against biotic stressors, such as phyto- pathogenic fungi, bacteria and viruses, which means that plants are better protected after the use of sulfoximines, in particular sulfoximines of the general formula (I) against infections of phytopathogenic fungi, bacteria and viruses.
- PR proteins pathogenesis-related proteins
- the active compounds can be converted into the customary formulations, such as solutions, emulsions, wettable powders, water- and oil-based suspensions, powders, dusts, pastes, soluble powders, soluble granules, scattering granules, suspension-emulsion concentrates, active substance-impregnated natural products, active ingredient impregnated synthetic materials, fertilizers and micro-encapsulants in polymeric materials.
- customary formulations such as solutions, emulsions, wettable powders, water- and oil-based suspensions, powders, dusts, pastes, soluble powders, soluble granules, scattering granules, suspension-emulsion concentrates, active substance-impregnated natural products, active ingredient impregnated synthetic materials, fertilizers and micro-encapsulants in polymeric materials.
- formulations are prepared in a known manner, e.g. by mixing the active compounds with extenders, ie liquid solvents and / or solid carriers, if appropriate using surface-active agents, ie emulsifiers and / or dispersants and / or foam-forming agents.
- extenders ie liquid solvents and / or solid carriers
- surface-active agents ie emulsifiers and / or dispersants and / or foam-forming agents.
- Excipients which can be used are those which are suitable for imparting special properties to the composition itself and / or preparations derived therefrom (for example spray liquor, seed dressing), such as certain technical properties and / or also particular biological properties.
- Typical auxiliaries are: extenders, solvents and carriers.
- polar and non-polar organic chemical liquids e.g. from the classes of aromatic and non-aromatic hydrocarbons (such as paraffins, alkylbenzenes, alkylnaphthalenes, chlorobenzenes), alcohols and polyols (which may also be substituted, etherified and / or esterified), ketones (such as acetone, cyclohexanone), Esters (including fats and oils) and (poly) ethers, simple and substituted amines, amides, lactams (such as N-alkylpyrrolidones) and lactones, sulfones and sulfoxides (such as dimethyl sulfoxide).
- aromatic and non-aromatic hydrocarbons such as paraffins, alkylbenzenes, alkylnaphthalenes, chlorobenzenes
- alcohols and polyols which may also be substituted, etherified and / or esterified
- ketones such
- organic solvents can also be used as auxiliary solvents.
- Suitable liquid solvents are essentially: aromatics, such as xylene, toluene, or alkylnaphthalenes, chlorinated aromatics and chlorinated aliphatic hydrocarbons, such as chlorobenzenes, chloroethylenes or methylene chloride, aliphatic hydrocarbons, such as cyclohexane or paraffins, for example petroleum fractions, mineral and vegetable oils , Alcohols, such as butanol or glycol, and their ethers and esters, ketones, such as acetone, clay, methyl ethyl ketone, methyl isobutyl ketone or cyclohexanone, strongly polar solvents, such as dimethyl sulfoxide, and water.
- aromatics such as xylene, toluene, or alkylnaphthalenes
- chlorinated aromatics and chlorinated aliphatic hydrocarbons such as chlorobenz
- Suitable carriers are:
- Ammonium salts and ground natural minerals such as kaolins, clays, talc, chalk, quartz, attapulgite, montmorillonite or diatomaceous earth, and ground synthetic minerals, such as fumed silica, alumina and silicates, as solid carriers for granules are suitable: e.g. crushed and fractionated natural rocks such as calcite, marble, pumice, sepiolite, dolomite and synthetic granules of inorganic and organic flours and granules of organic material such as paper, sawdust, coconut shells, corn cobs and tobacco stalks; suitable emulsifiers and / or foam formers are: e.g.
- nonionic and anionic emulsifiers such as polyoxyethylene fatty acid esters, polyoxyethylene fatty alcohol ethers, e.g. Alkylaryl polyglycol ethers, alkylsulfonates, alkyl sulfates, arylsulfonates and protein hydrolysates;
- suitable dispersants are non-ionic and / or ionic substances, e.g.
- Adhesives such as carboxymethylcellulose, natural and synthetic powdery, granular or latex-form polymers such as gum arabic, polyvinyl alcohol, polyvinyl acetate, and natural phospholipids such as cephalins and lecithins and synthetic phospholipids may be used in the formulations.
- Dyes such as inorganic pigments, e.g. Iron oxide, titanium oxide, ferrocyan blue and organic dyes such as alizarin, azo and metal phthalocyanine dyes and trace nutrients such as salts of iron, manganese, boron, copper, cobalt, molybdenum and zinc.
- inorganic pigments e.g. Iron oxide, titanium oxide, ferrocyan blue and organic dyes such as alizarin, azo and metal phthalocyanine dyes and trace nutrients such as salts of iron, manganese, boron, copper, cobalt, molybdenum and zinc.
- additives may be fragrances, mineral or vegetable optionally modified oils, waxes and nutrients (also micronutrients), such as salts of iron, manganese, boron, copper, cobalt, molybdenum and zinc.
- Stabilizers such as cold stabilizers, preservatives, antioxidants, light stabilizers or other chemical and / or physical stability-improving agents may also be present.
- the formulations generally contain between 0.01 and 98% by weight of active compound, preferably between 0.5 and 90%.
- the active compound according to the invention can be present in its commercially available formulations as well as in the formulations prepared from these formulations in admixture with other active ingredients such as insecticides, attractants, sterilants, bactericides, acaricides, nematicides, fungicides, growth-regulating substances, herbicides, safeners, fertilizers or semiochemicals.
- active ingredients such as insecticides, attractants, sterilants, bactericides, acaricides, nematicides, fungicides, growth-regulating substances, herbicides, safeners, fertilizers or semiochemicals.
- the invention further relates to the use of sulfoximines, in particular of sulfoximines of the general formula (I), to plants against plant diseases caused by fungi, bacteria, viruses, MLO (Mycoplasma -like organisms) and / or RLO (Rickettsia-like organisms) be protected.
- sulfoximines regardless of insect control, provide good protection of the plant from damage by fungal, bacterial or viral pathogens.
- the plants may be treated with single agents or with combinations of sulfoximines of the general formula (I).
- this protection is provided by the induction of PR proteins as a result of treatment with sulfoximines of general formula (I).
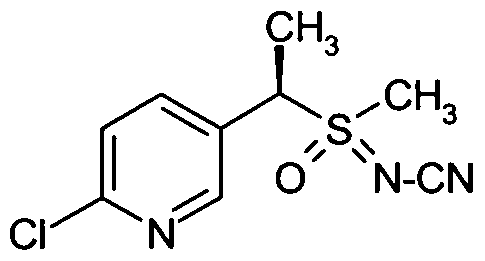
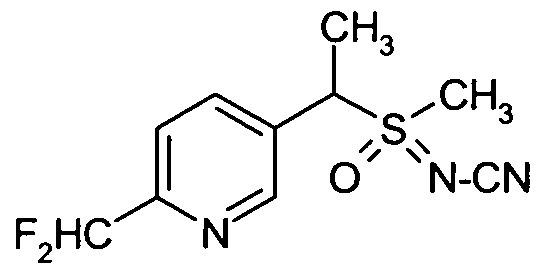
- Preferred sulfoximines of the general formula (I) are:
- Plant varieties are understood as meaning plants new traits that have been bred either by conventional breeding, by mutagenesis or by recombinant DNA techniques. Crop plants can accordingly be plants which can be obtained by conventional breeding and optimization methods or by biotechnological and genetic engineering methods or combinations of these methods, including the transgenic plants and including the plant varieties which can or can not be protected by variety protection rights.
- the treatment method according to the invention can thus also for the treatment of genetically modified organisms (GMOs), z.
- GMOs genetically modified organisms
- Genetically modified plants are plants in which a heterologous gene has been stably integrated into the genome.
- the term "heterologous gene” essentially refers to a gene that is provided or assembled outside the plant and that when introduced into the nuclear genome, chloroplast genome or mitochondrial genome imparts new or improved agronomic or other properties to the transformed plant Expressing protein or polypeptide, or that it downregulates or shuts down another gene present in the plant or other genes present in the plant (for example by means of antisense technology, cosuppression technology or RNAi technology [RNA Interference]).
- a heterologous gene present in the genome is also referred to as a transgene.
- a transgene defined by its specific presence in the plant genome is referred to as a transformation or transgenic event.
- Plants and plant varieties which are preferably treated according to the invention include all plants which have genetic material conferring on these plants particularly advantageous, useful features (whether obtained by breeding and / or biotechnology).
- Plants and plant varieties which are also preferably treated according to the invention are resistant to one or more biotic stressors, i. H. These plants have an improved defense against animal and microbial pests such as nematodes, insects, mites, phytopathogenic fungi, bacteria, viruses and / or viroids.
- Plants and plant varieties which can also be treated according to the invention are those plants which are resistant to one or more abiotic stress factors.
- Abiotic stress conditions may include, for example, drought, cold and heat conditions, osmotic stress, waterlogging, increased soil salinity, increased exposure to minerals, ozone conditions, high light conditions, limited availability of nitrogen nutrients, limited availability of phosphorous nutrients, or avoidance of shade.
- Plants and plant varieties which can also be treated according to the invention are those plants which are characterized by increased yield properties. An increased yield can in these plants z.
- the yield may be further influenced by improved plant architecture (under stress and non-stress conditions), including early flowering, control of flowering for hybrid seed production, seedling vigor, plant size, internode count and spacing, root growth, seed size, Fruit size, pod size, pod or ear number, number of seeds per pod or ear, seed mass, increased seed filling, reduced seed drop, reduced pod popping and stability.
- improved plant architecture under stress and non-stress conditions
- Other yield-related traits include seed composition such as carbohydrate content, protein content, oil content and composition, nutritional value, reduction in nontoxic compounds, improved processability, and improved shelf life.
- Plants which can be treated according to the invention are hybrid plants which already express the properties of the heterosis or of the hybrid effect, which generally leads to higher yields, higher vigor, better health and better resistance to biotic and abiotic stress factors.
- Such plants are typically produced by crossing an inbred male sterile parental line (the female crossover partner) with another inbred male fertile parent line (the male crossbred partner).
- the hybrid seed is typically harvested from the male sterile plants and sold to propagators.
- Pollen sterile plants can sometimes be produced (eg in maize) by delaving (ie mechanical removal of the male reproductive organs or the male flowers); however, it is more common for male sterility to be due to genetic determinants in the plant genome.
- pollen sterile plants can also be obtained using plant biotechnology methods such as genetic engineering.
- a particularly convenient means of producing male-sterile plants is described in WO 89/10396, wherein, for example, a ribonuclease such as a barnase is selectively expressed in the tapetum cells in the stamens. The fertility can then be restorated by expression of a ribonuclease inhibitor such as barstar in the tapetum cells (eg WO 1991/002069).
- Plants or plant varieties obtained by methods of plant biotechnology, such as genetic engineering which can be treated according to the invention are herbicide-tolerant plants, i. H. Plants tolerant to one or more given herbicides. Such plants can be obtained either by genetic transformation or by selection of plants containing a mutation conferring such herbicide tolerance.
- Herbicide-tolerant plants are, for example, glyphosate-tolerant plants, i. H. Plants tolerant to the herbicide glyphosate or its salts.
- glyphosate-tolerant plants can be obtained by transforming the plant with a gene encoding the enzyme 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS).
- EPSPS 5-enolpyruvylshikimate-3-phosphate synthase
- EPSPS 5-enolpyruvylshikimate-3-phosphate synthase
- AroA gene mutant CT7 of the bacterium Salmonella typhimurium (Comai et al., Science (1983), 221, 370-371)
- the CP4 gene of the bacterium Agrobacterium sp. Barry et al., Curr Topics Plant Physiol.
- Glyphosate-tolerant plants can also be obtained by expressing a gene coding for a glyphosate oxidoreductase enzyme as described in US 5,776,760 and US 5,463,175. Glyphosate-tolerant plants can also be obtained by expressing a gene encoding a glyphosate acetyltransferase enzyme as described in e.g. As WO 2002/036782, WO 2003/092360, WO 2005/012515 and WO 2007/024782 is encoded. Glyphosate-tolerant plants can also be obtained by selecting plants containing naturally occurring mutations of the abovementioned genes, as described, for example, in WO 2001/024615 or WO 2003/013226.
- herbicide-resistant plants are, for example, plants tolerant to herbicides which inhibit the enzyme glutamine synthase, such as bialaphos, phosphinotricin or glufosinate have been made. Such plants can be obtained by expressing an enzyme which detoxifies the herbicide or a mutant of the enzyme glutamine synthase, which is resistant to inhibition.
- an effective detoxifying enzyme is, for example, an enzyme encoding a phosphinotricin acetyltransferase (such as the bar or pat protein of Streptomyces species).
- Plants expressing an exogenous phosphinotricin acetyltransferase are described, for example, in US 5,561,236; US 5,648,477; US 5,646,024; US 5,273,894; US 5,637,489; US 5,276,268; US 5,739,082; US 5,908,810 and US 7,112,665.
- hydroxyphenylpyruvate dioxygenase HPPD
- HPPD hydroxyphenylpyruvate dioxygenases
- HPPD inhibitors may be treated with a gene encoding a naturally occurring resistant HPPD enzyme or a gene encoding an imitated HPPD enzyme according to WO 1996/038567, WO 1999/024585 and WO 1999 / 024586, are transformed.
- Tolerance to HPPD inhibitors can also be achieved by transforming plants with genes encoding certain enzymes that allow the formation of homogentisate despite inhibition of the native HPPD enzyme by the HPPD inhibitor. Such plants and genes are described in WO 1999/034008 and WO 2002/36787.
- the tolerance of plants to HPPD inhibitors can also be improved by transforming plants in addition to a gene coding for an HPPD-tolerant enzyme with a gene coding for a prephenate dehydrogenase enzyme, as described in WO 2004 / 024928 is described.
- ALS inhibitors include sulfonylurea, imidazolinone, triazolopyrimidines, pyrimidinyloxy (thio) benzoates and / or sulfonylaminocarbonyltriazolinone herbicides.
- ALS also known as acetohydroxy acid synthase, AHAS
- AHAS acetohydroxy acid synthase
- plants which are tolerant to imidazolinone and / or sulfonylurea can be obtained by induced mutagenesis, selection in cell cultures in the presence of the herbicide or by mutation breeding, as for example for the soybean in US 5,084,082, for rice in WO 1997/41218, for the sugar beet in US 5,773,702 and WO 1999/057965, for salad in US 5,198,599 or for the sunflower in WO 2001/065922.
- Plants or plant varieties obtained by plant biotechnology methods such as genetic engineering which can also be treated according to the invention are insect-resistant transgenic plants, i. Plants that have been made resistant to attack by certain target insects. Such plants can be obtained by genetic transformation or by selection of plants containing a mutation conferring such insect resistance.
- insect-resistant transgenic plant includes any plant containing at least one transgene comprising a coding sequence encoding:
- an insecticidal crystal protein from Bacillus thuringiensis or an insecticidal portion thereof such as the insecticidal crystal proteins described by Crickmore et al., Microbiology and Molecular Biology Reviews (1998), 62, 807-813, by Crickmore et al. (2005) in the Bacillus thuringia 5w toxin nomenclature, online at: http://www.lifesci.sussex.ac.uk/Home/Neil_Crickmore/Bt/), or insecticidal portions thereof, e.g. Proteins of the cry protein classes CrylAb, CrylAc, CrylF, Cry2Ab, Cry3Ae or Cry3Bb or insecticidal parts thereof; or
- Bacillus thuringiensis crystal protein or a part thereof which is insecticidal in the presence of a second, other crystal protein than Bacillus thuringiensis or a part thereof, such as the binary toxin consisting of the crystal proteins Cy34 and Cy35 (Moellenbeck et al. Nat. Biotechnol. (2001), 19, 668-72; Schnepf et al., Applied Environment Microb. (2006), 71, 1765-1774); or
- an insecticidal hybrid protein comprising parts of two different insecticidal crystal proteins from Bacillus thuringiensis, such as a hybrid of the proteins of
- VIPs vegetative insecticidal proteins
- a secreted protein from Bacillus thuringiensis or Bacillus cereus which is insecticidal in the presence of a second secreted protein from Bacillus thuringiensis or B. cereus, such as the binary toxin consisting of the proteins VIPlA and VIP2A (WO 1994/21795); or
- a hybrid insecticidal protein comprising parts of various secreted proteins of Bacillus thuringiensis or Bacillus cereus, such as a hybrid of the proteins of 1) or a hybrid of the proteins of 2) above; or
- 8) a protein according to any one of items 1) to 3) above, in which some, in particular 1 to 10, amino acids have been replaced by another amino acid in order to achieve a higher insecticidal activity against a target insect species and / or the spectrum of the corresponding target insect species and / or due to changes induced in the coding DNA during cloning or transformation (preserving the coding for an insecticidal protein), such as the protein VIP3Aa in the cotton event
- insect-resistant transgenic plants in the present context also include any plant comprising a combination of genes encoding the proteins of any of the above classes 1 to 8.
- an insect resistant plant contains more than one transgene encoding a protein of any one of the above 1 to 8 in order to extend the spectrum of the corresponding target insect species or to delay the development of resistance of the insects to the plants by various Use proteins that are insecticidal for the same target insect species, but have a different mode of action, such as binding to different receptor binding sites in the insect.
- Plants or plant varieties obtained by plant biotechnology methods such as genetic engineering), which can also be treated according to the invention, are tolerant to abiotic stressors. Such plants can be obtained by genetic transformation or by selection of plants containing a mutation conferring such stress resistance. Particularly useful plants with stress tolerance include the following:
- Plants which contain a transgene which may reduce the expression and / or activity of the gene for the poly (ADP-ribose) polymerase (PARP) in the plant cells or plants, as described in WO 2000/004173 or EP 04077984.5 or EP 06009836.5 is described.
- Plants which contain a stress tolerance-enhancing transgene encoding a plant-functional enzyme of the nicotinamide adenine dinucleotide salvage biosynthetic pathway including
- Plants or plant varieties obtained by plant biotechnology methods such as genetic engineering which can also be treated according to the invention have a changed amount, quality and / or storability of the harvested product and / or altered characteristics of certain components of the harvested product, such as:
- transgenic plants which synthesize a modified starch are described, for example, in EP 0571427, WO 1995/004826, EP 0719338, WO 1996/15248, WO 1996/19581, WO 1996/27674, WO 1997/11188, WO 1997/26362, WHERE 1997/32985, WO 1997/42328, WO 1997/44472, WO 1997/45545, WO 1998/27212, WO 1998/40503, WO 99/58688, WO 1999/58690, WO 1999/58654, WO 2000/008184, WO 2000/008185, WO 2000/28052, WO 2000/77229, WO 2001/12782, WO 2001/12826, WO 2002/101059, WO 2003/071860, WO 2004/056999, WO 2005/030942, WO 2005/030941, WO 2005/095632, WO 2005/095617, WO 2005/095619, WO 2005/095618, WO
- Transgenic plants that synthesize non-starch carbohydrate polymers or non-starch carbohydrate polymers whose properties are altered compared to wild-type plants without genetic modification.
- plants which produce polyfructose, in particular of the inulin and levan type, as described in EP 0663956, WO 1996/001904, WO 1996/021023, WO 1998/039460 and WO 1999/024593, plants which are alpha-1 , 4-glucans, as described in WO 1995/031553, US 2002/031826, US 6,284,479, US 5,712,107, WO 1997/047806, WO 1997/047807, WO 1997/047808 and WO 2000/14249, plants which alpha-1, 6-branched alpha-1,4-glucans, as described in WO 2000/73422, and plants producing alternan, as described in WO 2000/047727, EP 06077301.7, US 5,908,975 and EP 0728213 is.
- Plants or plant varieties obtained by plant biotechnology methods such as genetic engineering, which can also be treated according to the invention, are plants such as cotton plants with altered fiber properties. Such plants can be obtained by genetic transformation or by selection of plants containing a mutation conferring such altered fiber properties; these include:
- plants such as cotton plants, which contain an altered form of cellulose synthase genes, as described in WO 1998/000549
- plants such as cotton plants, which contain an altered form of rsw2 or rsw3 homologous nucleic acids, as described in WO 2004/053219;
- plants such as cotton plants having an increased expression of sucrose phosphate synthase, as described in WO 2001/017333;
- plants such as cotton plants with an increased expression of sucrose synthase, as described in WO 02/45485;
- plants such as cotton plants with modified reactivity fibers, e.g. By expression of the N-acetylglucosamine transferase gene, including nodC, and of chitin synthase genes, as described in WO 2006/136351.
- Plants or plant varieties obtained by plant biotechnology methods such as genetic engineering, which can also be treated according to the invention, are plants such as oilseed rape or related Brassica plants with altered properties of the oil composition. Such plants can be obtained by genetic transformation or by selection of plants containing a mutation conferring such altered oil properties; these include:
- plants such as rape plants producing oil of high oleic acid content, as described, for example, in US 5,969,169, US 5,840,946 or US 6,323,392 or US 6,063,947;
- plants such as oilseed rape plants which produce low linolenic acid oil, as described in US 6,270,828, US 6,169,190 or US 5,965,755.
- plants such as oilseed rape plants, which produce oil with a low saturated fatty acid content, as described, for. As described in US 5,434,283.
- transgenic plants which can be treated according to the invention are plants with one or more genes coding for one or more toxins, the transgenic plants offered under the following commercial names: YIELD GARD® (for example maize, cotton, Soybeans), KnockOut® (for example corn), Bite-Gard® (for example maize), BT-Xtra® (for example corn), StarLink® (for example maize), Bollgard® (cotton), Nucotn® (cotton), Nucotn 33B® (cotton), NatureGard® (for example corn), Protecta® and NewLeaf® (potato).
- YIELD GARD® for example maize, cotton, Soybeans
- KnockOut® for example corn
- Bite-Gard® for example maize
- BT-Xtra® for example corn
- StarLink® for example maize
- Bollgard® cotton
- Nucotn® cotton
- Nucotn 33B® cotton
- NatureGard® for example corn
- Herbicide-tolerant crops to be mentioned are, for example, corn, cotton and soybean varieties sold under the following tradenames: Roundup Ready® (glyphosate tolerance, for example corn, cotton, soybean), Liberty Link® (phosphinotricin tolerance, for example rapeseed) , MI® (imidazolinone tolerance) and SCS® (sylphonylurea tolerance), for example corn.
- Herbicide-resistant plants (plants traditionally grown for herbicide tolerance) to be mentioned include the varieties sold under the name Clearfield® (for example corn).
- transgenic plants that can be treated according to the invention are plants that contain transformation events, or a combination of transformation events, and that are listed, for example, in the files of various national or regional authorities (see, for example, http: // /gmoinfo.jrc.it/gmp_browse.aspx and http://www.agbios.com/dbase.php).
- Preferred sulfoximines of general formula (I) for this application to transgenic plants and transgenic seeds are:
- the defense against the following pathogens is preferably enhanced: Botrytis cinerea, Phytophthora nicotianae, Peronospora tabacinae, Phytophthora infestans, Sphaerotheca fuliginea, Phakopsora pachyrhizi, Ramularia gossypii, Rhizoctonia solani, Curvularia spec, Pyrenophora spec, Sclerotinia homoeoca ⁇ a, Erysiphe graminis, Colletotrichum graminicola, Pythium ultimum, Pythium aphanidermatum.
- Blumeria species such as Blumeria graminis
- Podosphaera species such as Podosphaera leucotricha
- Sphaerotheca species such as Sphaerotheca fuliginea
- Uncinula species such as Uncinula necator
- Diseases caused by pathogens of rust diseases such as Gymnosporangium species such as, for example, Gymnosporangium sabinae Hemileia species such as Hemileia vastatrix;
- Phakopsora species such as Phakopsora pachyrhizi and Phakopsora meibomiae
- Puccinia species such as Puccinia recondita
- Uromyces species such as Uromyces appendiculatus
- Bremia species such as Bremia lactucae
- Peronospora species such as Peronospora pisi or P. brassicae
- Phytophthora species such as Phytophthora infestans
- Plasmopara species such as Plasmopara viticola
- Pseudoperonospora species such as Pseudoperonospora humuli or
- Pythium species such as Pythium ultimum; • leaf spot diseases and leaf wilt caused by e.g.
- Alternaria species such as Alternaria solani;
- Cercospora species such as Cercospora beticola
- Cladosporium species such as Cladosporium cucumerinum
- Cochliobolus species such as Cochliobolus sativus: (conidia form: Drechslera, Syn: Helminthosporium);
- Colletotrichum species such as Colletotrichum lindemuthanium
- Cycloconium species such as cycloconium oleaginum; Diaporthe species, such as Diaporthe citri;
- Elsinoe species such as Elsinoe fawcettii
- Gloeosporium species such as, for example, Gloeosporium laeticolor
- Glomerella species such as Glomerella cingulata
- Guignardia species such as Guignardia bidwelli;
- Leptosphaeria species such as Leptosphaeria maculans
- Magnaporthe species such as Magnaporthe grisea
- Mycosphaerella species such as Mycosphaerella graminicola and Mycosphaerella fijiensis;
- Phaeosphaeria species such as Phaeosphaeria nodorum
- Pyrenophora species such as, for example, Pyrenophora teres
- Ramularia species such as Ramularia collo-cygni
- Rhynchosporium species such as Rhynchosporium secalis
- Septoria species such as Septoria apii
- Typhula species such as Typhula incarnata
- Venturia species such as Venturia inaequalis
- Corticium species such as Corticium graminearum
- Fusarium species such as Fusarium oxysporum
- Gaeumannomyces species such as Gaeumannomyces graminis
- Rhizoctonia species such as Rhizoctonia solani
- Tapesia species such as Tapesia acuformis
- Thielaviopsis species such as Thielaviopsis basicola
- Ear and panicle diseases including corncob, caused by eg
- Alternaria species such as Alternaria spp .
- Aspergillus species such as Aspergillus flavus
- Cladosporium species such as Cladosporium cladosporioides
- Claviceps species such as Claviceps purpurea
- Fusarium species such as Fusarium culmorum
- Gibberella species such as Gibberella zeae
- Monographella species such as Monographella nivalis; • Diseases caused by fire fungi, such as
- Sphacelotheca species such as Sphacelotheca reiliana
- Tilletia species such as Tilletia caries
- Urocystis species such as Urocystis occulta
- Ustilago species such as Ustilago nuda
- Aspergillus species such as Aspergillus flavus
- Botrytis species such as Botrytis cinerea
- Penicillium species such as Penicillium expansum and Penicillium purpurogenum
- Sclerotinia species such as Sclerotinia sclerotiorum
- Verticilium species such as Verticilium alboatrum
- Seed and soil-borne rots and wilting, and seedling diseases caused by eg
- Alternaria species such as Alternaria brassicicola
- Aphanomyces species such as Aphanomyces euteiches; Ascochyta species, such as Ascochyta lentis;
- Aspergillus species such as Aspergillus flavus
- Cladosporium species such as Cladosporium herbarum
- Cochliobolus species such as Cochliobolus sativus
- Fusarium species such as Fusarium culmorum
- Gibberella species such as Gibberella zeae
- Macrophomina species such as Macrophomina phaseolina
- Monographella species such as Monographella nivalis
- Penicillium species such as Penicillium expansum
- Phoma species such as Phoma Hungary
- Phomopsis species such as Phomopsis sojae
- Phytophthora species such as Phytophthora cactorum
- Pyrenophora species such as Pyrenophora graminea
- Pyricularia species such as Pyricularia oryzae
- Pythium species such as Pythium ultimum
- Rhizoctonia species such as Rhizoctonia solani
- Rhizopus species such as Rhizopus oryzae
- Sclerotium species such as Sclerotium rolfsii
- Septoria species such as Septoria nodorum
- Typhula species such as Typhula incarnata
- Verticillium species such as Verticillium dahliae
- Nectria species such as Nectria galligena
- Monilinia species such as Monilinia laxa
- Taphrina species such as Taphrina deformans
- Esca species such as Phaeomoniella chlamydospora and Phaeoacremonium aleophilum and Fomitiporia mediterranea;
- Botrytis species such as Botrytis cinerea
- Rhizoctonia species such as Rhizoctonia solani
- Hehninthosporium species such as Helminthosporium solani
- Xanthomonas species such as Xanthomonas campestris pv. Oryzae;
- Pseudomonas species such as Pseudomonas syringae pv. Lachrymans; Erwinia species, such as Erwinia amylovora;
- the following diseases of soybean beans can be controlled:
- Alternaria leaf spot (Alternaria spec. Atrans tenuissima), Anthracnose (Colletotrichum gloeospoides dematium var. Truncatum), Brown spot (Septoria glycines), Cercospora leaf spot and blight (Cercospora kikuchii), Choanephora leaf blight (Choanephora infundibulifera trispora (Syn.
- Dactuliophora leaf spot (Dactuliophora glycines), Downy Mildew (Peronospora manshurica), Drechslera blight (Drechslera glycini), Frogeye leaf spot (Cercospora sojina), Leptosphaerulina leaf spot (Leptosphaerulina trifolii), Phyllostica leaf spot (Phyllosticta sojaecola), Pod and Star Blight (Phomopsis sojae), Powdery Mildew (Microsphaera diffusa), Pyrenochaeta Leaf Spot (Pyrenochaeta glycines), Rhizoctonia Aerial, Foliage, and Web Blight (Rhizoctonia solani), Rust (Phakopsora pachyrhizi), Scab (Sphaceloma glycines), Stemphylium Leaf Blight (Stemphylium botryosum), Target Spot
- Black Root Red (Calonectria crotalariae), Charcoal Red (Macrophomina phaseolina), Fusarium Blight or Wiit, Root Red, and Pod and Collar Red (Fusarium oxysporum, Fusarium orthoceras, Fusarium semitectum, Fusarium equiseti), Mycoleptodiscus Root Red (Mycoleptodiscus terrestris), Neocosmospora (Neocosmopspora vasinfecta), Pod and Star Blight (Diaporthe phaseolorum), Stem Canker (Diaporthe phaseolorum var.
- Phytophthora red (Phytophthora megasperma), Brown Stem Red (Phialophora gregata), Pythium Red (Pythium aphanidermatum, Pythium irregular , Pythium debaryanum, Pythium myriotylum, Pythium ultimum), Rhizoctonia Root Red, Stem Decay, and Damping Off (Rhizoctonia solani), Sclerotinia Stem Decay (Sclerotinia sclerotium), Sclerotinia Southern Blight (Sclerotinia rolfsii), Thielaviopsis Root Red (Thielaviopsis basicola).
- Preferred times for the application of sulfoximines for pathogen defense are seed, soil, Nähratess-, stem and / or leaf treatments with the approved application rates.
- the amounts of a sulfoximine to achieve the properties of the invention can be varied within a substantial range.
- concentrations of from 0.00001% to 0.05% are used, more preferably from 0.000025% to 0.025%, and most preferably from 0.000025% to 0.005%.
- concentration of active compound combinations is preferably between 0.000025% and 0.005%, more preferably between 0.00005% and 0.001%.
- the active compounds according to the invention can also be used in the treatment of seed. Preference is given to mention the active ingredients mentioned above as being particular, preferred, particularly preferred and very particularly preferred.
- the compounds of the general formulas (Ia) to (Dc) and the specific compounds of the formers (I-1) to (1-23) should be mentioned in particular.
- the present invention therefore more particularly relates to a method for protecting seed and germinating plants for improving plant growth and / or enhancing the resistance of plants to plant diseases caused by fungi, bacteria, viruses, MLO and / or RLO, the seed and / or the seed mende plant is treated with an active ingredient according to the invention.
- the invention also relates to the corresponding use of the active ingredients according to the invention for the treatment of seed.
- the invention relates to seed which has been treated with an active ingredient according to the invention.
- the present invention also relates to appropriate nutrient solutions, in particular for the cultivation of plants and / or germinating plants, containing one for increasing the plant's own defenses and / or for improving plant growth and / or for increasing the resistance of plants to plant diseases caused by fungi, bacteria, viruses, MLO (Mycoplasma -like organisms) and / or RLO (Rickett's -like organisms), effective amount of at least one sulfoximine, in particular a sulfoximine of the general formula (I).
- the nutrient solutions preferably have a content of at least one sulfoximine of from 0.0005 to 0.025% by weight, based on the total weight of the nutrient solution.
- the at least one sulfoximine is present in the form of an NMP-free formulation containing 10 to 50% by weight of propylene carbonate.
- the method of protecting seed and germinating plants to improve plant growth and increase the resistance of plants to plant diseases caused by fungi, bacteria, viruses, MLO and / or RLO, by using a seed of the invention Active substance in the so-called “float process” or “floating process” (Leal, RS, The use of Confidor S in the float, a new tobacco seedling production system in the South of Brazil.) Crop Protection News Bayer (German Edition) (2001), 54 (3), pages 337 to 352; Rudolph, RD; Rogers, WD Crop Protection News Bayer (German Edition) (2001), The efficacy of imidacloprid treatment for reduction in the severity of insect vectored virus diseases ), 54 (3), pages 311 to 336).
- the seed is kept in special containers, e.g. Styrofoam perforated trays, seeded in special potting soil based on peat culture substrate and then cultivated in containers with suitable nutrient solution until reaching the desired transplantation size (see Figure 1).
- the containers are allowed to run on the nutrient solution, from which the name of the growing method is derived (Leal, 2001, supra).
- insecticides from the class of neonicotinoids chlornicotinyls
- the active compounds according to the invention can be used in particular also in transgenic seed.
- the active compounds to be used according to the invention are suitable for protecting and supporting seed of any plant variety as mentioned above, which is used in agriculture, in the greenhouse, in forests or in horticulture.
- these are corn, peanut, canola, rapeseed, poppy, soybean, cotton, turnip (eg sugarbeet and fodder beet), rice, millet, wheat, barley, oats, rye, sunflower, tobacco, potatoes or vegetables ( eg tomatoes, cabbage).
- the active compounds to be used according to the invention are likewise suitable for the treatment of the seed of fruit plants and vegetables as already mentioned above. Of particular importance is the treatment of the seeds of maize, soya, cotton, wheat and canola or rapeseed.
- the active compounds according to the invention are applied to the seed alone or in a suitable formulation.
- the seed is treated in a state where it is so stable that no damage occurs during the treatment.
- the treatment of the seed can be done at any time between harvesting and sowing.
- seed is used which has been separated from the plant and freed from flasks, shells, stems, hull, wool or pulp.
- the amount of the active compounds according to the invention and / or further additives applied to the seed is chosen such that the germination of the seed is not impaired or the resulting plant is not damaged. This is especially important for active ingredients, which can show phytotoxic effects in certain application rates.
- the agents according to the invention can be applied directly, ie without containing further components and without being diluted. In general, it is preferable to apply the agents to the seed in the form of a suitable formulation.
- Suitable formulations and methods for seed treatment are known to those skilled in the art and are described, for example, in the following documents: US 4,272,417 A, US 4,245,432 A, US 4,808,430 A, US 5,876,739 A, US 2003/0176428 A1, WO 2002/080675 A1, WO 2002 / 028186 A2.
- the active compounds which can be used according to the invention can be converted into the customary seed dressing formulations, such as solutions, emulsions, suspensions, powders, foams, slurries or other seed coating compositions, as well as ULV formulations.
- formulations are prepared in a known manner by mixing the active compounds of the invention with conventional additives, such as conventional extenders and solvents or diluents, dyes, wetting agents, dispersants, emulsifiers, defoamers, preservatives, secondary thickeners, adhesives, gibberellins and water ,
- conventional additives such as conventional extenders and solvents or diluents, dyes, wetting agents, dispersants, emulsifiers, defoamers, preservatives, secondary thickeners, adhesives, gibberellins and water ,
- Dyes which may be present in the seed dressing formulations which can be used according to the invention are all dyes customary for such purposes. Both water-insoluble pigments and water-soluble dyes are useful in this case. Examples which may be mentioned under the names rhodamine B, CI. Pigment Red 112 and CI. Solvent Red 1 known dyes.
- Suitable wetting agents which may be present in the seed dressing formulations which can be used according to the invention are all wetting-promoting substances customary for the formulation of agrochemical active compounds.
- Preferably usable are alkylnaphthalene sulfonates such as diisopropyl or diisobutylnaphthalene sulfonates.
- Suitable dispersants and / or emulsifiers which may be present in the seed dressing formulations which can be used according to the invention are all nonionic, anionic and cationic dispersants customary for the formulation of agrochemical active compounds.
- Preferably usable are nonionic or anionic dispersants or mixtures of nonionic or anionic dispersants.
- Particularly suitable nonionic dispersants are, in particular, ethylene oxide-propylene oxide, block polymers, alkylphenol polyglycol ethers and tristryrylphenol polyglycol ethers and their phosphated or sulfated derivatives.
- Suitable anionic dispersants are in particular lignosulfonates, polyacrylic acid salts and arylsulfonate-formaldehyde condensates.
- defoamers all foam-inhibiting substances customary for the formulation of agrochemical active substances can be present in the seed dressing formulations which can be used according to the invention.
- foam-inhibiting substances customary for the formulation of agrochemical active substances can be present in the seed dressing formulations which can be used according to the invention.
- Preferably usable are silicone defoamers and magnesium stearate.
- all substances which can be used for such purposes in agrochemical compositions can be present in the seed dressing formulations which can be used according to the invention.
- examples include dichlorophen and Benzylalkoholhemiformal.
- Suitable secondary thickeners which may be present in the seed dressing formulations which can be used according to the invention are all substances which can be used for such purposes in agrochemical compositions. Preference is given to cellulose derivatives, acrylic acid derivatives, xanthan, modified clays and highly dispersed silicic acid.
- Suitable adhesives which may be present in the seed dressing formulations which can be used according to the invention are all customary binders which can be used in pickling agents.
- Preferably mentioned are polyvinylpyrrolidone, polyvinyl acetate, polyvinyl alcohol and Tylose.
- the gibberellins are known (see R. Wegler "Chemie der convinced- und Swdlingsbekungsstoff", Vol. 2, Springer Verlag, 1970, pp. 401-412).
- the seed dressing formulations which can be used according to the invention can be used either directly or after prior dilution with water for the treatment of seed of various kinds, including seed of transgenic plants.
- the seed dressing formulations which can be used according to the invention or the preparations prepared therefrom by the addition of water
- all mixing devices customarily usable for the dressing can be considered.
- the seed is placed in a mixer which adds either desired amount of seed dressing formulations either as such or after prior dilution with water and mixes until evenly distributed the formulation on the seed.
- a drying process follows.
- the active compounds according to the invention can generally be used in their commercial formulations and in the forms of application prepared from these formulations Mixtures with other active ingredients, such as insecticides, attractants, sterilants, acaricides, nematicides, fungicides, growth regulators or herbicides.
- active ingredients such as insecticides, attractants, sterilants, acaricides, nematicides, fungicides, growth regulators or herbicides.
- inhibitors of nucleic acid synthesis such as benalaxyl, benalaxyl-M, bupirimate, clozylacone, dimethirimol, ethirimol, furalaxyl, hymexazole, metalaxyl, metalaxyl-M, ofurace,
- Mitosis and cell division inhibitors such as benomyl, carbendazim, chlorfenazole, diethofencarb, ethaboxam, fuberidazole, pencycuron, thiabendazole, thiophanate, thiophanate-methyl and zoxamide.
- inhibitors of respiration such as diflumetorim as an inhibitor at complex I of the respiratory chain; Bixafen, boscalid, carboxin, fenfuram, flutolanil, fluopyram, furametpyr, furmecyclox, isopyrazam (mixture of the syn-epimeric racemate 1RS, 4SR, 9RS and the anti-epimeric racemate 1RS, 4SR, 9SR), isopyrazam (syn epimeric racemate 1RS, 4SR, 9RS), isopyrazam (syn-epimeric enantiomer 1R, 4S, 9R), isopyrazam (syn-epimeric enantiomer 1S, 4R, 9S), isopyrazam (anti-epimeric racemate 1RS, 4SR, 9SR), isopyrazam (anti-epimeric racemate 1RS, 4SR, 9SR), isopyrazam (anti-epimeric racemate 1RS, 4SR,
- decouplers such as binapacryl, dinocap, fluazinam and meptyldinocap.
- inhibitors of ATP production such as, for example, fentin acetate, fentin chloride, fentin hydroxide and silthiofam.
- inhibitors of amino acid and protein biosynthesis such as andoprim, blasticidin-S, cyprodinil, kasugamycin, kasugamycin hydrochloride hydrate, mepanipyrim and pyrimethanil.
- signal transduction inhibitors such as fenpiclonil, fludioxonil and quinoxyfen.
- Inhibitors of lipid and membrane synthesis such as biphenyl, chlozolinate, edifenphos, etridiazole, iodocarb, Iprobenfos, iprodione, isoprothiolane, procymidone, propamocarb, propamocarb hydrochloride, pyrazophos, tolclofos-methyl and vinclozolin.
- inhibitors of ergosterol biosynthesis such as aldimorph, azaconazole, bitertanol, bromuconazole, cyproconazole, diclobutrazole, difenoconazole, diniconazole, diniconazole-M,
- inhibitors of cell wall synthesis such as Benthiavalicarb, Dimethomo ⁇ h, Flumo ⁇ h, Iprovalicarb, Mandipropamid, Polyoxins, Polyoxorim, Prothiocarb, Validamycin A and valefenalate.
- inhibitors of melanin biosynthesis such as, for example, caryropamide, diclocymet, fenoxanil, fthalide, pyroquilone and tricyclazole.
- resistance inducers such as acibenzolar-S-methyl, probenazole and tiadinil.
- Acetylcholinesterase (AChE) inhibitors such as carbamates, e.g. Alanycarb, aldicarb, bendiocarb, benfuracarb, butocarboxime, butoxycarboxime, carbaryl, carbofuran, carbosulfane, ethiofencarb, fenobucarb, formetanate, furathiocarb, isoprocarb, methiocarb, methomyl, metolcarb, oxamyl, pirimicarb, propoxur, thiodicarb, thiofanox, triazamates, Trimethacarb, XMC and xylylcarb; or
- AChE Acetylcholinesterase
- Organophosphates eg acephates, azamethiphos, azinphos (-methyl, -ethyl), cadusafos, chiorethoxyfos, chlorfenvinphos, chlormephos, chlorpyrifos (-methyl), coumaphos, cyanophos, demeton-s-methyl, diazinon, dichlorvos / DDVP, Dicrotophos, Dimethoate, Dimethylvinphos, Disulphon, EPN, Ethion, Ethoprophos, Famphur, Fenamiphos, Fenitrothion, Fenthion, Fosthiazate, Heptenophos, Isofenphos, Isopropyl O- (methoxyaminothio-phosphoryl) salicylate, isoxathione, Malathion, mecarbam, methamidophos, methidathion, mevinphos, monocrotophos, naled, o
- GABA-controlled chloride channel antagonists such as organochlorines, e.g. Chlordane and endosulfan (alpha); or fiproles (phenylpyrazoles), e.g. Ethiprole, fipronil, pyrroliprole and pyriprole.
- sodium channel modulators / voltage dependent sodium channel blockers such as pyrethroids, e.g. Acrinathrin, allethrin (d-cis-trans, d-trans), bifenthrin, bioallethrin, bioalmosthrin S-cyclopentenyl, bioresmethrin, cycloprothrin, cyfluthrin (beta), cyhalothrin (gamma, lambda), cypermethrin (alpha) , beta-, theta-, ceta-), cyphenothrin [(IR) -trans-lsome ⁇ e], deltamethrin, dimefluthrin, empenthrin [(£ Z) - (1R) -isomers], esfenvalerate, etofenprox, fenpropathrine, Fenvalerates, flucythrinates, flumethrin, fluvalinates (
- nicotinergic acetylcholine receptor agonists such as neonicotinoids, e.g. Acetamido, clothianidin, dinotefuran, imidacloprid, nitenpyram, thiacloprid, thiamethoxam; or nicotine.
- Allosteric acetylcholine receptor modulators such as spinosyns, e.g. Spinetoram and spinosad.
- chloride channel activators such as avermectins / milbemycins, e.g. Abamectin, Emamectin benzoate, Lepimectin and Milbemectin.
- Juvenile hormone analogs e.g. Hydroprene, kinoprene, methoprene; or fenoxycarb; Pyritexyfen.
- agents with unknown or non-specific mechanisms of action such as
- Fumigant e.g. Methyl bromide and other alkyl halides; or chloropicrin; Sulfuryl fluoride; Borax; Tartar emetic.
- selective feed inhibitors eg, pymetrozines; or flonicamide.
- mite growth inhibitors eg clofentezine, diflovidazine, hexythiazox, etoxazole.
- Microbial disruptors of insect intestinal membrane such as Bacillus thuringiensis subsp. israelensis, Bacillus sphaericus, Bacillus thuringiensis subsp. aizawai, Bacillus thuringiensis subsp. kurstaki, Bacillus thuringiensis subsp. tenebrionis, and BT plant proteins, e.g. Cryl Ab, Cryl Ac, CrylFa, Cry2Ab, mCry3A, Cry3Ab, Cry3Bb, Cry34 / 35Abl.
- insect intestinal membrane such as Bacillus thuringiensis subsp. israelensis, Bacillus sphaericus, Bacillus thuringiensis subsp. aizawai, Bacillus thuringiensis subsp. kurstaki, Bacillus thuringiensis subsp. tenebrionis, and BT plant proteins, e.g. Cryl Ab
- inhibitors of oxidative phosphorylation, ATP disruptors such as diafenthiourone; or organotin compounds, e.g. Azocyclotine, cyhexatin, fenbutatin oxide; or propriety; Tetradifon.
- Nicotinergic acetylcholine receptor antagonists such as Bensultap, Cartap (hydrochloride), thiocylam, and thiosultap (sodium).
- inhibitors of chitin biosynthesis type 0, such as benzoylureas, e.g. Bistrifluron, chlorofluorazuron, diflubenzuron, flucycloxuron, flufenoxuron, hexaflumuron, vufenon, novaluron, noviflumuron, teflubenzuron and triflumuron.
- benzoylureas e.g. Bistrifluron, chlorofluorazuron, diflubenzuron, flucycloxuron, flufenoxuron, hexaflumuron, vufenon, novaluron, noviflumuron, teflubenzuron and triflumuron.
- inhibitors of chitin biosynthesis type 1, such as buprofezin.
- Moulting agents such as Cyromazine.
- Diacylhydrazines e.g. Chromafenozide, Halofenozide, Methoxyfenozide and Tebufenozide.
- Octopaminergic agonists such as amitraz.
- side-I electron transport inhibitors / side II electron transport inhibitors such as, for example, hydramethylnone; acequinocyl; fluacrypyrim; or Cyflumetofen and Cyenopyrafen.
- Electron transport inhibitors such as side-I electron transport inhibitors, from the group of METI acaricides, e.g. Fenazaquin, Fenpyroximate, Pyrimidifen, Pyridaben, Tebfenpyrad, Tolfenpyrad; or Rotenone (Derris).
- METI acaricides e.g. Fenazaquin, Fenpyroximate, Pyrimidifen, Pyridaben, Tebfenpyrad, Tolfenpyrad; or Rotenone (Derris).
- side-IV electron transport inhibitors such as phosphines, e.g. Aluminum phosphide, calcium phosphide, phosphine, zinc phosphide; or cyanide.
- ryanodine receptor effectors such as diamides, e.g. Chlorantraniliprole (Rynaxypyr), Cyantraniliprole (Cyazypyr) and Flubendiamide.
- agents with unknown mechanism of action such as azadirachtin, amidoflumet, benzoximate, bifenazates, quinomethionate, cryolites, dicofol, flufenerim, pyridalyl and pyrifluquinazone; or the following known effective compounds
- Figure 1 Floating box filled with nutrient solution.
- Figure 2 Floating box with floating styrofoam shells filled with potting soil and tobacco seeds.
- Figure 3 Styrofoam shells with tobacco plants after cultivation in a floating box.
- Tomato seeds (Solarium lycopersicum, Rentita ') are grown in rock wool. After germination, the rockwool blocks are transferred to a Floating Box provided with the preparation and further cultivated under suitable climatic conditions.
- the nutrient solution of the floating box was not ventilated (oxygen deficiency stress).
- the maximum length of the root per tomato plant is measured and the average root length per floating box and treatment is calculated.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Agronomy & Crop Science (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Dentistry (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Fertilizers (AREA)
Abstract
Description
Claims
Priority Applications (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| MX2011001934A MX2011001934A (es) | 2008-08-29 | 2009-08-17 | Procedimientos para la mejora del crecimiento de plantas. |
| KR1020117007081A KR101649556B1 (ko) | 2008-08-29 | 2009-08-17 | 식물 성장 촉진방법 |
| EP09778023.3A EP2323488B1 (de) | 2008-08-29 | 2009-08-17 | Methode zur verbesserung des pflanzenwachstums |
| CN200980142927.6A CN102196727B (zh) | 2008-08-29 | 2009-08-17 | 改良植物生长的方法 |
| BRPI0917104A BRPI0917104B1 (pt) | 2008-08-29 | 2009-08-17 | uso de sulfoximinas para intensificar a resistência de plantas a fatores abióticos de estresse de deficência de oxigênio, solução nutriente, e seu uso |
| ES09778023.3T ES2677600T3 (es) | 2008-08-29 | 2009-08-17 | Procedimientos para la mejora del crecimiento de las plantas |
| US13/061,360 US8796175B2 (en) | 2008-08-29 | 2009-08-17 | Method for enhancing plant intrinsic defense |
| JP2011524237A JP5583669B2 (ja) | 2008-08-29 | 2009-08-17 | 植物成長を向上させるための方法 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE102008041695A DE102008041695A1 (de) | 2008-08-29 | 2008-08-29 | Methoden zur Verbesserung des Pflanzenwachstums |
| DE102008041695.9 | 2008-08-29 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2010022897A2 true WO2010022897A2 (de) | 2010-03-04 |
| WO2010022897A3 WO2010022897A3 (de) | 2011-01-06 |
Family
ID=41605883
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2009/006064 Ceased WO2010022897A2 (de) | 2008-08-29 | 2009-08-17 | Methoden zur verbesserung des pflanzenwachstums |
Country Status (11)
| Country | Link |
|---|---|
| US (1) | US8796175B2 (de) |
| EP (1) | EP2323488B1 (de) |
| JP (1) | JP5583669B2 (de) |
| KR (1) | KR101649556B1 (de) |
| CN (1) | CN102196727B (de) |
| BR (1) | BRPI0917104B1 (de) |
| DE (1) | DE102008041695A1 (de) |
| ES (1) | ES2677600T3 (de) |
| MX (1) | MX2011001934A (de) |
| TR (1) | TR201807878T4 (de) |
| WO (1) | WO2010022897A2 (de) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2012000946A2 (de) | 2010-06-30 | 2012-01-05 | Bayer Cropscience Ag | Wirkstoffkombinationen |
| JP2012250978A (ja) * | 2011-05-10 | 2012-12-20 | Sumitomo Chemical Co Ltd | 植物の生長を促進する方法 |
| JP2012250979A (ja) * | 2011-05-10 | 2012-12-20 | Sumitomo Chemical Co Ltd | 植物の生長を促進する方法 |
| US20230189805A1 (en) * | 2020-05-15 | 2023-06-22 | Clarke Mosquito Control Products, Inc. | Multi-solvent insecticidal compositions including sulfoximine |
Families Citing this family (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102333445B (zh) * | 2008-12-29 | 2014-09-03 | 拜尔农作物科学股份公司 | 改善利用转基因植物生产潜力的方法 |
| IN2014DN09428A (de) * | 2012-04-18 | 2015-07-17 | Dow Agrosciences Llc | |
| KR101437570B1 (ko) * | 2013-07-23 | 2014-09-04 | 주식회사 이티엔에스 | 식물 보습제 제조방법 |
| DE102013012500A1 (de) | 2013-07-26 | 2015-01-29 | Skw Stickstoffwerke Piesteritz Gmbh | Verwendung von Phosphorsäureamiden als Mittel zur Verbesserung der Trockenstresstoleranz |
| DE102013012498A1 (de) | 2013-07-26 | 2015-01-29 | Skw Stickstoffwerke Piesteritz Gmbh | Verwendung von einfachen 1,2,4-Triazol-Derivaten als Mittel zur Verbesserung der Trockenstresstoleranz |
| DE102013021933A1 (de) | 2013-12-20 | 2015-06-25 | Skw Stickstoffwerke Piesteritz Gmbh | Verwendung von Pyrazol-Derivaten als Mittel zur Verbesserung der Trockenstresstoleranz |
| DE102016107338A1 (de) | 2016-04-20 | 2017-10-26 | Skw Stickstoffwerke Piesteritz Gmbh | Verwendung von Imidamid-Derivaten als Mittel zur Verbesserung der Trockenstresstoleranz |
| CN106332755A (zh) * | 2016-08-25 | 2017-01-18 | 马家庆 | 一种牛油果栽培营养基料以及制备方法 |
| RU2654589C1 (ru) * | 2017-04-12 | 2018-05-21 | Федеральное государственное бюджетное научное учреждение "Всероссийский научно-исследовательский институт биологической защиты растений" | Регулятор роста озимой пшеницы и кукурузы |
| CN106977237A (zh) * | 2017-04-25 | 2017-07-25 | 龙胜裕丰农业发展有限公司 | 西红柿种植喷施营养液 |
| CN110810438A (zh) * | 2019-09-29 | 2020-02-21 | 西南大学 | 烟草疫霉菌纳米氧化铜抗菌剂及其制备方法 |
| CN117965606A (zh) * | 2024-01-25 | 2024-05-03 | 中国热带农业科学院三亚研究院 | 同时突变橡胶树HbMLO14和HbMLO23基因的编辑靶点、基因编辑载体和应用 |
Family Cites Families (198)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE1905834C3 (de) | 1969-02-06 | 1972-11-09 | Basf Ag | Verfahren zur Vermeidung des Staubens und Zusammenbackens von Salzen bzw.Duengemitteln |
| US4272417A (en) * | 1979-05-22 | 1981-06-09 | Cargill, Incorporated | Stable protective seed coating |
| US4245432A (en) * | 1979-07-25 | 1981-01-20 | Eastman Kodak Company | Seed coatings |
| US4761373A (en) | 1984-03-06 | 1988-08-02 | Molecular Genetics, Inc. | Herbicide resistance in plants |
| US5331107A (en) | 1984-03-06 | 1994-07-19 | Mgi Pharma, Inc. | Herbicide resistance in plants |
| US5304732A (en) | 1984-03-06 | 1994-04-19 | Mgi Pharma, Inc. | Herbicide resistance in plants |
| ATE57390T1 (de) | 1986-03-11 | 1990-10-15 | Plant Genetic Systems Nv | Durch gentechnologie erhaltene und gegen glutaminsynthetase-inhibitoren resistente pflanzenzellen. |
| US5637489A (en) | 1986-08-23 | 1997-06-10 | Hoechst Aktiengesellschaft | Phosphinothricin-resistance gene, and its use |
| US5273894A (en) | 1986-08-23 | 1993-12-28 | Hoechst Aktiengesellschaft | Phosphinothricin-resistance gene, and its use |
| US5276268A (en) | 1986-08-23 | 1994-01-04 | Hoechst Aktiengesellschaft | Phosphinothricin-resistance gene, and its use |
| US5013659A (en) | 1987-07-27 | 1991-05-07 | E. I. Du Pont De Nemours And Company | Nucleic acid fragment encoding herbicide resistant plant acetolactate synthase |
| US5605011A (en) | 1986-08-26 | 1997-02-25 | E. I. Du Pont De Nemours And Company | Nucleic acid fragment encoding herbicide resistant plant acetolactate synthase |
| US5378824A (en) | 1986-08-26 | 1995-01-03 | E. I. Du Pont De Nemours And Company | Nucleic acid fragment encoding herbicide resistant plant acetolactate synthase |
| US4808430A (en) * | 1987-02-27 | 1989-02-28 | Yazaki Corporation | Method of applying gel coating to plant seeds |
| ATE82668T1 (de) | 1987-08-21 | 1992-12-15 | Ciba Geigy Ag | Benzothiadiazole und ihre verwendung in verfahren und mitteln gegen pflanzenkrankheiten. |
| US5638637A (en) * | 1987-12-31 | 1997-06-17 | Pioneer Hi-Bred International, Inc. | Production of improved rapeseed exhibiting an enhanced oleic acid content |
| GB8810120D0 (en) | 1988-04-28 | 1988-06-02 | Plant Genetic Systems Nv | Transgenic nuclear male sterile plants |
| US5084082A (en) | 1988-09-22 | 1992-01-28 | E. I. Du Pont De Nemours And Company | Soybean plants with dominant selectable trait for herbicide resistance |
| US6013861A (en) * | 1989-05-26 | 2000-01-11 | Zeneca Limited | Plants and processes for obtaining them |
| ATE203276T1 (de) | 1989-08-10 | 2001-08-15 | Aventis Cropscience Nv | Pflanzen mit modifizierten blüten |
| US5739082A (en) | 1990-02-02 | 1998-04-14 | Hoechst Schering Agrevo Gmbh | Method of improving the yield of herbicide-resistant crop plants |
| US5908810A (en) | 1990-02-02 | 1999-06-01 | Hoechst Schering Agrevo Gmbh | Method of improving the growth of crop plants which are resistant to glutamine synthetase inhibitors |
| AU639319B2 (en) * | 1990-04-04 | 1993-07-22 | Pioneer Hi-Bred International, Inc. | Production of improved rapeseed exhibiting a reduced saturated fatty acid content |
| US5198599A (en) | 1990-06-05 | 1993-03-30 | Idaho Resarch Foundation, Inc. | Sulfonylurea herbicide resistance in plants |
| JP3173784B2 (ja) | 1990-06-25 | 2001-06-04 | モンサント カンパニー | グリホセート耐性植物 |
| EP0539588A1 (de) | 1990-07-05 | 1993-05-05 | Nippon Soda Co., Ltd. | Aminderivat |
| FR2667078B1 (fr) * | 1990-09-21 | 1994-09-16 | Agronomique Inst Nat Rech | Sequence d'adn conferant une sterilite male cytoplasmique, genome mitochondrial, mitochondrie et plante contenant cette sequence, et procede de preparation d'hybrides. |
| DE4104782B4 (de) | 1991-02-13 | 2006-05-11 | Bayer Cropscience Gmbh | Neue Plasmide, enthaltend DNA-Sequenzen, die Veränderungen der Karbohydratkonzentration und Karbohydratzusammensetzung in Pflanzen hervorrufen, sowie Pflanzen und Pflanzenzellen enthaltend dieses Plasmide |
| US5731180A (en) | 1991-07-31 | 1998-03-24 | American Cyanamid Company | Imidazolinone resistant AHAS mutants |
| DE4128828A1 (de) | 1991-08-30 | 1993-03-04 | Basf Ag | Ammonium- oder harnstoffhaltige duengemittel und verfahren zu ihrer herstellung |
| US6270828B1 (en) * | 1993-11-12 | 2001-08-07 | Cargrill Incorporated | Canola variety producing a seed with reduced glucosinolates and linolenic acid yielding an oil with low sulfur, improved sensory characteristics and increased oxidative stability |
| DE4227061A1 (de) | 1992-08-12 | 1994-02-17 | Inst Genbiologische Forschung | DNA-Sequenzen, die in der Pflanze die Bildung von Polyfructanen (Lävanen) hervorrufen, Plasmide enthaltend diese Sequenzen sowie Verfahren zur Herstellung transgener Pflanzen |
| GB9218185D0 (en) | 1992-08-26 | 1992-10-14 | Ici Plc | Novel plants and processes for obtaining them |
| DK0664835T3 (da) | 1992-10-14 | 2004-09-27 | Syngenta Ltd | Nye planter og fremgangsmåde til opnåelse af dem |
| GB9223454D0 (en) | 1992-11-09 | 1992-12-23 | Ici Plc | Novel plants and processes for obtaining them |
| HU220714B1 (hu) | 1993-03-25 | 2002-04-29 | Novartis Ag. | Új peszticid proteinek és törzsek |
| AU695940B2 (en) * | 1993-04-27 | 1998-08-27 | Cargill Incorporated | Non-hydrogenated canola oil for food applications |
| WO1995004826A1 (en) | 1993-08-09 | 1995-02-16 | Institut Für Genbiologische Forschung Berlin Gmbh | Debranching enzymes and dna sequences coding them, suitable for changing the degree of branching of amylopectin starch in plants |
| DE4330960C2 (de) | 1993-09-09 | 2002-06-20 | Aventis Cropscience Gmbh | Kombination von DNA-Sequenzen, die in Pflanzenzellen und Pflanzen die Bildung hochgradig amylosehaltiger Stärke ermöglichen, Verfahren zur Herstellung dieser Pflanzen und die daraus erhaltbare modifizierte Stärke |
| GB9318985D0 (en) | 1993-09-14 | 1993-10-27 | Xaar Ltd | Passivation of ceramic piezoelectric ink jet print heads |
| CA2150667C (en) | 1993-10-01 | 2007-01-09 | Mari Iwabuchi | A gene which determines cytoplasmic sterility and a method of producing hybrid plants using said gene |
| AU692791B2 (en) * | 1993-10-12 | 1998-06-18 | Agrigenetics, Inc. | Brassica napus variety AG019 |
| PL180543B1 (pl) * | 1993-11-09 | 2001-02-28 | Du Pont | Zrekombinowany konstrukt DNA, sposób wytwarzania fruktanui sposób zwiekszania poziomu fruktanu w roslinach PL PL |
| EP0754235A1 (de) | 1994-03-25 | 1997-01-22 | National Starch and Chemical Investment Holding Corporation | Verfahren zur herstellung veränderten stärke aus kartoffelpflanzen |
| PT759993E (pt) | 1994-05-18 | 2007-08-10 | Bayer Bioscience Gmbh | ''sequências de dna que codificam para enzimas capazes de facilitar a síntese de alfa-1,4-glucanos lineares em plantas, fungos e microrganismos'' |
| US5824790A (en) * | 1994-06-21 | 1998-10-20 | Zeneca Limited | Modification of starch synthesis in plants |
| JPH10507622A (ja) | 1994-06-21 | 1998-07-28 | ゼネカ・リミテッド | 新規植物およびその入手法 |
| NL1000064C1 (nl) | 1994-07-08 | 1996-01-08 | Stichting Scheikundig Onderzoe | Produktie van oligosacchariden in transgene planten. |
| DE4441408A1 (de) | 1994-11-10 | 1996-05-15 | Inst Genbiologische Forschung | DNA-Sequenzen aus Solanum tuberosum kodierend Enzyme, die an der Stärkesynthese beteiligt sind, Plasmide, Bakterien, Pflanzenzellen und transgene Pflanzen enhaltend diese Sequenzen |
| DE4447387A1 (de) | 1994-12-22 | 1996-06-27 | Inst Genbiologische Forschung | Debranching-Enzyme aus Pflanzen und DNA-Sequenzen kodierend diese Enzyme |
| ATE373094T1 (de) | 1995-01-06 | 2007-09-15 | Plant Res Int Bv | Für kohlenhydratpolymere-bildende enzyme- kodierende dna-sequenzen und verfahren zur herstellung transgener pflanzen |
| DE19509695A1 (de) | 1995-03-08 | 1996-09-12 | Inst Genbiologische Forschung | Verfahren zur Herstellung einer modifizieren Stärke in Pflanzen, sowie die aus den Pflanzen isolierbare modifizierte Stärke |
| CA2218526C (en) | 1995-04-20 | 2012-06-12 | American Cyanamid Company | Structure-based designed herbicide resistant products |
| US5853973A (en) | 1995-04-20 | 1998-12-29 | American Cyanamid Company | Structure based designed herbicide resistant products |
| US6825342B1 (en) | 1995-05-05 | 2004-11-30 | National Starch And Chemical Investment Holding Corporation | Plant starch composition |
| FR2734842B1 (fr) | 1995-06-02 | 1998-02-27 | Rhone Poulenc Agrochimie | Sequence adn d'un gene de l'hydroxy-phenyl pyruvate dioxygenase et obtention de plantes contenant un gene de l'hydroxy-phenyl pyruvate dioxygenase, tolerantes a certains herbicides |
| US6284479B1 (en) * | 1995-06-07 | 2001-09-04 | Pioneer Hi-Bred International, Inc. | Substitutes for modified starch and latexes in paper manufacture |
| US5712107A (en) * | 1995-06-07 | 1998-01-27 | Pioneer Hi-Bred International, Inc. | Substitutes for modified starch and latexes in paper manufacture |
| GB9513881D0 (en) | 1995-07-07 | 1995-09-06 | Zeneca Ltd | Improved plants |
| FR2736926B1 (fr) * | 1995-07-19 | 1997-08-22 | Rhone Poulenc Agrochimie | 5-enol pyruvylshikimate-3-phosphate synthase mutee, gene codant pour cette proteine et plantes transformees contenant ce gene |
| ES2335494T3 (es) * | 1995-09-19 | 2010-03-29 | Bayer Bioscience Gmbh | Procedimiento para la produccion de un almidon modificado. |
| GB9524938D0 (en) | 1995-12-06 | 1996-02-07 | Zeneca Ltd | Modification of starch synthesis in plants |
| DE19601365A1 (de) | 1996-01-16 | 1997-07-17 | Planttec Biotechnologie Gmbh | Nucleinsäuremoleküle aus Pflanzen codierend Enzyme, die an der Stärkesynthese beteiligt sind |
| DE19608918A1 (de) * | 1996-03-07 | 1997-09-11 | Planttec Biotechnologie Gmbh | Nucleinsäuremoleküle, die neue Debranching-Enzyme aus Mais codieren |
| US5773704A (en) | 1996-04-29 | 1998-06-30 | Board Of Supervisors Of Louisiana State University And Agricultural And Mechanical College | Herbicide resistant rice |
| DE19618125A1 (de) * | 1996-05-06 | 1997-11-13 | Planttec Biotechnologie Gmbh | Nucleinsäuremoleküle, die neue Debranching-Enzyme aus Kartoffel codieren |
| DE19619918A1 (de) | 1996-05-17 | 1997-11-20 | Planttec Biotechnologie Gmbh | Nucleinsäuremoleküle codierend lösliche Stärkesynthasen aus Mais |
| BR9709487A (pt) | 1996-05-29 | 1999-08-10 | Hoechst Schering Agrevo Gmbh | Moléculas de ácido nucléico que codificam enzimas de trigo que est o envolvidas nas síntese de amigo |
| CA2257621C (en) | 1996-06-12 | 2003-02-04 | Pioneer Hi-Bred International, Inc. | Substitutes for modified starch in paper manufacture |
| EP0904453B1 (de) | 1996-06-12 | 2001-12-19 | Pioneer Hi-Bred International, Inc. | Ersatzmaterial für modifizierte stärke in der papierherstellung |
| WO1997047807A1 (en) | 1996-06-12 | 1997-12-18 | Pioneer Hi-Bred International, Inc. | Substitutes for modified starch in paper manufacture |
| US5876739A (en) * | 1996-06-13 | 1999-03-02 | Novartis Ag | Insecticidal seed coating |
| AUPO069996A0 (en) | 1996-06-27 | 1996-07-18 | Australian National University, The | Manipulation of plant cellulose |
| US5850026A (en) * | 1996-07-03 | 1998-12-15 | Cargill, Incorporated | Canola oil having increased oleic acid and decreased linolenic acid content |
| US5773702A (en) | 1996-07-17 | 1998-06-30 | Board Of Trustees Operating Michigan State University | Imidazolinone herbicide resistant sugar beet plants |
| DE19631764A1 (de) * | 1996-08-06 | 1998-02-12 | Basf Ag | Neue Nitrifikationsinhibitoren sowie die Verwendung von Polysäuren zur Behandlung von Mineraldüngemitteln die einen Nitrifikationsinhibitor enthalten |
| GB9623095D0 (en) | 1996-11-05 | 1997-01-08 | Nat Starch Chem Invest | Improvements in or relating to starch content of plants |
| US6232529B1 (en) | 1996-11-20 | 2001-05-15 | Pioneer Hi-Bred International, Inc. | Methods of producing high-oil seed by modification of starch levels |
| DE19653176A1 (de) | 1996-12-19 | 1998-06-25 | Planttec Biotechnologie Gmbh | Neue Nucleinsäuremoleküle aus Mais und ihre Verwendung zur Herstellung einer modifizierten Stärke |
| CA2193938A1 (en) | 1996-12-24 | 1998-06-24 | David G. Charne | Oilseed brassica containing an improved fertility restorer gene for ogura cytoplasmic male sterility |
| US5981840A (en) | 1997-01-24 | 1999-11-09 | Pioneer Hi-Bred International, Inc. | Methods for agrobacterium-mediated transformation |
| DE19708774A1 (de) * | 1997-03-04 | 1998-09-17 | Max Planck Gesellschaft | Nucleinsäuremoleküle codierend Enzyme die Fructosylpolymeraseaktivität besitzen |
| DE19709775A1 (de) | 1997-03-10 | 1998-09-17 | Planttec Biotechnologie Gmbh | Nucleinsäuremoleküle codierend Stärkephosphorylase aus Mais |
| GB9718863D0 (en) | 1997-09-06 | 1997-11-12 | Nat Starch Chem Invest | Improvements in or relating to stability of plant starches |
| DE19749122A1 (de) | 1997-11-06 | 1999-06-10 | Max Planck Gesellschaft | Nucleinsäuremoleküle codierend Enzyme, die Fructosyltransferaseaktivität besitzen |
| US6245968B1 (en) * | 1997-11-07 | 2001-06-12 | Aventis Cropscience S.A. | Mutated hydroxyphenylpyruvate dioxygenase, DNA sequence and isolation of plants which contain such a gene and which are tolerant to herbicides |
| FR2770854B1 (fr) | 1997-11-07 | 2001-11-30 | Rhone Poulenc Agrochimie | Sequence adn d'un gene de l'hydroxy-phenyl pyruvate dioxygenase et obtention de plantes contenant un tel gene, tolerantes aux herbicides |
| FR2772789B1 (fr) | 1997-12-24 | 2000-11-24 | Rhone Poulenc Agrochimie | Procede de preparation enzymatique d'homogentisate |
| WO1999053072A1 (en) | 1998-04-09 | 1999-10-21 | E.I. Du Pont De Nemours And Company | Starch r1 phosphorylation protein homologs |
| AU3536699A (en) * | 1998-04-27 | 1999-11-16 | Ihara Chemical Industry Co. Ltd. | 3-arylphenyl sulfide derivatives and insecticides and miticides |
| DE19820608A1 (de) * | 1998-05-08 | 1999-11-11 | Hoechst Schering Agrevo Gmbh | Nucleinsäuremoleküle codierend Enzyme aus Weizen, die an der Stärkesynthese beteiligt sind |
| DE19820607A1 (de) * | 1998-05-08 | 1999-11-11 | Hoechst Schering Agrevo Gmbh | Nucleinsäuremoleküle codierend Enzyme aus Weizen, die an der Stärkesynthese beteiligt sind |
| JP4494634B2 (ja) * | 1998-05-13 | 2010-06-30 | バイエル バイオサイエンス ゲーエムベーハー | プラスチドadp/atp転移因子の改変された活性を有するトランスジェニック植物 |
| DE19821614A1 (de) | 1998-05-14 | 1999-11-18 | Hoechst Schering Agrevo Gmbh | Sulfonylharnstoff-tolerante Zuckerrübenmutanten |
| JP2002518015A (ja) | 1998-06-15 | 2002-06-25 | ナショナル スターチ アンド ケミカル インベストメント ホールディング コーポレイション | 植物および植物製品の改良またはそれらに関する改良 |
| US6693185B2 (en) | 1998-07-17 | 2004-02-17 | Bayer Bioscience N.V. | Methods and means to modulate programmed cell death in eukaryotic cells |
| DE19836098A1 (de) * | 1998-07-31 | 2000-02-03 | Hoechst Schering Agrevo Gmbh | Pflanzen, die eine modifizierte Stärke synthetisieren, Verfahren zur Herstellung der Pflanzen, ihre Verwendung sowie die modifizierte Stärke |
| DE19836099A1 (de) * | 1998-07-31 | 2000-02-03 | Hoechst Schering Agrevo Gmbh | Nukleinsäuremoleküle kodierend für eine ß-Amylase, Pflanzen, die eine modifizierte Stärke synthetisieren, Verfahren zur Herstellung der Pflanzen, ihre Verwendung sowie die modifizierte Stärke |
| AU6018399A (en) | 1998-08-25 | 2000-03-14 | Pioneer Hi-Bred International, Inc. | Plant glutamine: fructose-6-phosphate amidotransferase nucleic acids |
| WO2000014249A1 (en) | 1998-09-02 | 2000-03-16 | Planttec Biotechnologie Gmbh | Nucleic acid molecules encoding an amylosucrase |
| DK1117802T3 (da) * | 1998-10-09 | 2010-05-25 | Bayer Bioscience Gmbh | Nukleinsyremolekyler, der koder for et forgreningsenzym fra bakterier af slægten Neisseria samt fremgangsmåde til fremstilling af alfa-1,6-forgrenede alfa-1,4-glucaner |
| DE19924342A1 (de) | 1999-05-27 | 2000-11-30 | Planttec Biotechnologie Gmbh | Genetisch modifizierte Pflanzenzellen und Pflanzen mit erhöhter Aktivität eines Amylosucraseproteins und eines Verzweigungsenzyms |
| JP2002529094A (ja) | 1998-11-09 | 2002-09-10 | プランテック バイオテクノロジー ゲーエムベーハー | コメ由来核酸分子および改変デンプンの生産のためのその使用 |
| US6503904B2 (en) * | 1998-11-16 | 2003-01-07 | Syngenta Crop Protection, Inc. | Pesticidal composition for seed treatment |
| US6531648B1 (en) | 1998-12-17 | 2003-03-11 | Syngenta Participations Ag | Grain processing method and transgenic plants useful therein |
| DE19905069A1 (de) * | 1999-02-08 | 2000-08-10 | Planttec Biotechnologie Gmbh | Nucleinsäuremoleküle codierend Alternansucrase |
| US6323392B1 (en) * | 1999-03-01 | 2001-11-27 | Pioneer Hi-Bred International, Inc. | Formation of brassica napus F1 hybrid seeds which exhibit a highly elevated oleic acid content and a reduced linolenic acid content in the endogenously formed oil of the seeds |
| HUP0201018A2 (en) | 1999-04-29 | 2002-07-29 | Syngenta Ltd | Herbicide resistant plants |
| PL354925A1 (en) | 1999-04-29 | 2004-03-22 | Syngenta Ltd | Herbicide resistant plants |
| DE19926771A1 (de) | 1999-06-11 | 2000-12-14 | Aventis Cropscience Gmbh | Nukleinsäuremoleküle aus Weizen, transgene Pflanzenzellen und Pflanzen und deren Verwendung für die Herstellung modifizierter Stärke |
| DE19937348A1 (de) * | 1999-08-11 | 2001-02-22 | Aventis Cropscience Gmbh | Nukleinsäuremoleküle aus Pflanzen codierend Enzyme, die an der Stärkesynthese beteiligt sind |
| DE19937643A1 (de) * | 1999-08-12 | 2001-02-22 | Aventis Cropscience Gmbh | Transgene Zellen und Pflanzen mit veränderter Aktivität des GBSSI- und des BE-Proteins |
| AU7647000A (en) | 1999-08-20 | 2001-03-19 | Basf Plant Science Gmbh | Increasing the polysaccharide content in plants |
| US6423886B1 (en) * | 1999-09-02 | 2002-07-23 | Pioneer Hi-Bred International, Inc. | Starch synthase polynucleotides and their use in the production of new starches |
| US6472588B1 (en) | 1999-09-10 | 2002-10-29 | Texas Tech University | Transgenic cotton plants with altered fiber characteristics transformed with a sucrose phosphate synthase nucleic acid |
| GB9921830D0 (en) | 1999-09-15 | 1999-11-17 | Nat Starch Chem Invest | Plants having reduced activity in two or more starch-modifying enzymes |
| AR025996A1 (es) | 1999-10-07 | 2002-12-26 | Valigen Us Inc | Plantas no transgenicas resistentes a los herbicidas. |
| EP1261252B1 (de) | 2000-03-09 | 2013-04-24 | E.I. Du Pont De Nemours And Company | Sulfonylurea-tolerante sonnenblumenpflanzen |
| DE60111613T2 (de) | 2000-03-09 | 2006-05-18 | Monsanto Technology Llc. | Verfahren zum herstellen von glyphosat-toleranten pflanzen |
| EP1325136A1 (de) | 2000-09-29 | 2003-07-09 | Syngenta Limited | Herbizid-resistente pflanzen |
| US6660690B2 (en) | 2000-10-06 | 2003-12-09 | Monsanto Technology, L.L.C. | Seed treatment with combinations of insecticides |
| US6734340B2 (en) | 2000-10-23 | 2004-05-11 | Bayer Cropscience Gmbh | Monocotyledon plant cells and plants which synthesise modified starch |
| WO2002036782A2 (en) | 2000-10-30 | 2002-05-10 | Maxygen, Inc. | Novel glyphosate n-acetyltransferase (gat) genes |
| FR2815969B1 (fr) | 2000-10-30 | 2004-12-10 | Aventis Cropscience Sa | Plantes tolerantes aux herbicides par contournement de voie metabolique |
| CN1326996C (zh) | 2000-12-08 | 2007-07-18 | 联邦科学及工业研究组织 | 蔗糖合酶的基因表达在植物组织中的修饰及其用途 |
| US20020134012A1 (en) | 2001-03-21 | 2002-09-26 | Monsanto Technology, L.L.C. | Method of controlling the release of agricultural active ingredients from treated plant seeds |
| US20040107461A1 (en) | 2001-03-30 | 2004-06-03 | Padma Commuri | Glucan chain length domains |
| JP4460282B2 (ja) | 2001-06-12 | 2010-05-12 | バイエル・クロップサイエンス・アーゲー | 高アミロースデンプンを合成するトランスジェニック植物 |
| US20030084473A1 (en) | 2001-08-09 | 2003-05-01 | Valigen | Non-transgenic herbicide resistant plants |
| BRPI0213423A2 (pt) | 2001-10-17 | 2017-05-02 | Basf Plant Science Gmbh | amido e método para produção do mesmo |
| WO2003092360A2 (en) | 2002-04-30 | 2003-11-13 | Verdia, Inc. | Novel glyphosate-n-acetyltransferase (gat) genes |
| FR2844142B1 (fr) * | 2002-09-11 | 2007-08-17 | Bayer Cropscience Sa | Plantes transformees a biosynthese de prenylquinones amelioree |
| AU2003275859A1 (en) | 2002-10-29 | 2004-05-25 | Basf Plant Science Gmbh | Compositions and methods for identifying plants having increased tolerance to imidazolinone herbicides |
| US20040110443A1 (en) | 2002-12-05 | 2004-06-10 | Pelham Matthew C. | Abrasive webs and methods of making the same |
| DK1578973T3 (da) | 2002-12-19 | 2008-11-24 | Bayer Cropscience Ag | Planteceller og planter, der syntetiserer en stivelse med foröget slutviskositet |
| CN1777677A (zh) | 2003-03-07 | 2006-05-24 | 巴斯福植物科学有限公司 | 增强植物中直链淀粉产量 |
| AU2004227111B2 (en) | 2003-04-09 | 2009-05-28 | Bayer Cropscience Nv. | Methods and means for increasing the tolerance of plants to stress conditions |
| CN1863914B (zh) | 2003-04-29 | 2011-03-09 | 先锋高级育种国际公司 | 新的草甘膦-n-乙酰转移酶(gat)基因 |
| CA2526480A1 (en) | 2003-05-22 | 2005-01-13 | Syngenta Participations Ag | Modified starch, uses, methods for production thereof |
| US9382526B2 (en) | 2003-05-28 | 2016-07-05 | Basf Aktiengesellschaft | Wheat plants having increased tolerance to imidazolinone herbicides |
| EP1493328A1 (de) | 2003-07-04 | 2005-01-05 | Institut National De La Recherche Agronomique | Verfahren zur Herstellung von doppel null fertilität-restaurations Linien von B. napus mit guter agromomischer Qualität |
| US7547819B2 (en) * | 2003-07-31 | 2009-06-16 | Toyo Boseki Kabushiki Kaisha | Plant producing hyaluronic acid |
| EP1692285B1 (de) | 2003-08-15 | 2012-04-11 | Commonwealth Scientific and Industrial Research Organisation | Verfahren und mittel zur veränderung der fasereigenschaften in faserproduzierenden pflanzen |
| BRPI0413917B1 (pt) | 2003-08-29 | 2018-09-25 | Instituto Nacional De Tech Agropecuaria | ácido nucleico ahas de arroz mutagenizado não transgênico, polipeptídeo ahas isolado, e, método de controle de ervas daninhas dentro da vizinhança de uma planta de arroz |
| EP1705987A2 (de) * | 2003-09-24 | 2006-10-04 | Avestha Gengraine Technologies Pvt. Ltd | Proteine mit möglichkeiten als nutrazeutika und biotherapeutika |
| WO2005030941A1 (en) | 2003-09-30 | 2005-04-07 | Bayer Cropscience Gmbh | Plants with increased activity of a class 3 branching enzyme |
| DE602004030613D1 (de) | 2003-09-30 | 2011-01-27 | Bayer Cropscience Ag | Pflanzen mit reduzierter aktivität eines klasse-3-verzweigungsenzyms |
| EP1725666B1 (de) | 2004-03-05 | 2012-01-11 | Bayer CropScience AG | Pflanzen mit reduzierter aktivität des stärkephosphorylierenden enzyms phosphoglucan-wasser-dikinase |
| AR048025A1 (es) | 2004-03-05 | 2006-03-22 | Bayer Cropscience Gmbh | Plantas con actividad aumentada de una enzima fosforilante del almidon |
| AR048026A1 (es) | 2004-03-05 | 2006-03-22 | Bayer Cropscience Gmbh | Procedimientos para la identificacion de proteinas con actividad enzimatica fosforiladora de almidon |
| AR048024A1 (es) | 2004-03-05 | 2006-03-22 | Bayer Cropscience Gmbh | Plantas con actividad aumentada de distintas enzimas fosforilantes del almidon |
| US7432082B2 (en) | 2004-03-22 | 2008-10-07 | Basf Ag | Methods and compositions for analyzing AHASL genes |
| KR101151932B1 (ko) * | 2004-04-08 | 2012-06-01 | 다우 아그로사이언시즈 엘엘씨 | 살충성 n-치환된 설폭시민 |
| CA2570298A1 (en) | 2004-06-16 | 2006-01-19 | Basf Plant Science Gmbh | Polynucleotides encoding mature ahasl proteins for creating imidazolinone-tolerant plants |
| DE102004029763A1 (de) | 2004-06-21 | 2006-01-05 | Bayer Cropscience Gmbh | Pflanzen, die Amylopektin-Stärke mit neuen Eigenschaften herstellen |
| CN101031646B (zh) | 2004-07-30 | 2013-09-25 | 巴斯夫农业化学产品公司 | 抗除草剂的向日葵植物、编码抗除草剂的乙酰羟酸合酶大亚基蛋白的多核苷酸和使用方法 |
| DE102004037506A1 (de) * | 2004-08-03 | 2006-02-23 | Bayer Cropscience Ag | Methode zur Verbesserung der Pflanzenverträglichkeit gegenüber Glyphosate |
| EP1776462A4 (de) | 2004-08-04 | 2010-03-10 | Basf Plant Science Gmbh | Monokotyledon-ahass-sequenzen und verwendungsverfahren |
| ATE459720T1 (de) | 2004-08-18 | 2010-03-15 | Bayer Cropscience Ag | Pflanzen mit erhöhter plastidär aktivität der stärkephosphorylierenden r3-enzyme |
| WO2006021972A1 (en) | 2004-08-26 | 2006-03-02 | Dhara Vegetable Oil And Foods Company Limited | A novel cytoplasmic male sterility system for brassica species and its use for hybrid seed production in indian oilseed mustard brassica juncea |
| TW200626532A (en) * | 2004-09-21 | 2006-08-01 | Syngenta Participations Ag | Novel insecticides |
| WO2006032538A1 (en) | 2004-09-23 | 2006-03-30 | Bayer Cropscience Gmbh | Methods and means for producing hyaluronan |
| PL1794306T3 (pl) | 2004-09-24 | 2010-05-31 | Bayer Cropscience Nv | Rośliny oporne na stres |
| EP1807519B1 (de) | 2004-10-29 | 2012-02-01 | Bayer BioScience N.V. | Stresstolerante baumwollpflanzen |
| AR051690A1 (es) | 2004-12-01 | 2007-01-31 | Basf Agrochemical Products Bv | Mutacion implicada en el aumento de la tolerancia a los herbicidas imidazolinona en las plantas |
| EP1672075A1 (de) | 2004-12-17 | 2006-06-21 | Bayer CropScience GmbH | Transformierte Pflanzen, die Dextransucrase exprimieren und eine veränderte Stärke synthetisieren |
| EP1679374A1 (de) | 2005-01-10 | 2006-07-12 | Bayer CropScience GmbH | Transformierte Pflanzen, die Mutansucrase exprimieren und eine veränderte Stärke synthetisieren |
| JP2006304779A (ja) | 2005-03-30 | 2006-11-09 | Toyobo Co Ltd | ヘキソサミン高生産植物 |
| EP1707632A1 (de) | 2005-04-01 | 2006-10-04 | Bayer CropScience GmbH | Phosphorylierte waxy-Kartoffelstärke |
| EP1710315A1 (de) | 2005-04-08 | 2006-10-11 | Bayer CropScience GmbH | Hoch Phosphat Stärke |
| DE102005022994A1 (de) | 2005-05-19 | 2006-11-30 | Bayer Cropscience Ag | Methode zur Verbesserung des Pflanzenwachstums und der Steigerung der Widerstandsfähigkeit gegen bodenbürtige Schadpilze in Pflanzen |
| EP1731037A1 (de) | 2005-06-04 | 2006-12-13 | Bayer CropScience AG | Erhöhung der Stresstoleranz durch Anwendung von Nikotinoiden auf Planzen, die genmodifiziert worden sind, um stresstolerant zu werden |
| EP1893759B1 (de) | 2005-06-15 | 2009-08-12 | Bayer BioScience N.V. | Verfahren zum erhöhen der widerstandsfähigkeit von pflanzen gegen hypoxische bedingungen |
| KR101468830B1 (ko) | 2005-06-24 | 2014-12-03 | 바이엘 크롭사이언스 엔.브이. | 식물 세포벽의 반응성을 변경하는 방법 |
| AR054174A1 (es) | 2005-07-22 | 2007-06-06 | Bayer Cropscience Gmbh | Sobreexpresion de sintasa de almidon en vegetales |
| MEP3508A (xx) | 2005-08-24 | 2010-02-10 | Pioneer Hi Bred Int | Kompozicije koje obezbjeđuju otpornost na više hibricida i njihov postupak upotrebe |
| MX2008002802A (es) | 2005-08-31 | 2008-04-07 | Monsanto Technology Llc | Secuencias nucleotidicas que codifican proteinas insecticidas. |
| DE102005045174A1 (de) | 2005-09-21 | 2007-03-22 | Bayer Cropscience Ag | Steigerung der Pathogenabwehr in Pflanzen |
| US10428341B2 (en) | 2005-10-05 | 2019-10-01 | Basf Se | Transgenic potato plants with increased hyaluronan production |
| JP2009509557A (ja) | 2005-10-05 | 2009-03-12 | バイエル・クロップサイエンス・アーゲー | 改善されたヒアルロン酸産生方法および手段 |
| BRPI0616844A2 (pt) * | 2005-10-05 | 2011-07-05 | Bayer Cropscience Ag | célula de planta geneticamente modificada, uso da mesma, planta, processo para produção da mesma, material de reprodução de plantas, partes de plantas colhìveis, processo para produção de hialuronano, composição, bem como seu processo de produção |
| DE102005059468A1 (de) | 2005-12-13 | 2007-06-14 | Bayer Cropscience Ag | Insektizide Zusammensetzungen mit verbesserter Wirkung |
| TW201309635A (zh) | 2006-02-10 | 2013-03-01 | Dow Agrosciences Llc | 殺蟲性之n-取代(6-鹵烷基吡啶-3-基)烷基磺醯亞胺(二) |
| US8158856B2 (en) | 2006-03-21 | 2012-04-17 | Bayer Cropscience Nv | Stress resistant plants |
| DE102006015467A1 (de) * | 2006-03-31 | 2007-10-04 | Bayer Cropscience Ag | Substituierte Enaminocarbonylverbindungen |
| DE102006015468A1 (de) * | 2006-03-31 | 2007-10-04 | Bayer Cropscience Ag | Substituierte Enaminocarbonylverbindungen |
| DE102006015470A1 (de) * | 2006-03-31 | 2007-10-04 | Bayer Cropscience Ag | Substituierte Enaminocarbonylverbindungen |
| US8237017B2 (en) | 2006-05-12 | 2012-08-07 | Bayer Cropscience Nv | Stress-related microRNA molecules and uses thereof |
| TWI381811B (zh) | 2006-06-23 | 2013-01-11 | Dow Agrosciences Llc | 用以防治可抵抗一般殺蟲劑之昆蟲的方法 |
| EP1887079A1 (de) * | 2006-08-09 | 2008-02-13 | Bayer CropScience AG | Genetisch modifizierte Pflanzen, die eine Stärke mit erhöhtem Quellvermögen synthetisieren |
| TWI409256B (zh) | 2006-09-01 | 2013-09-21 | Dow Agrosciences Llc | 殺蟲性之n-取代(雜芳基)環烷基磺醯亞胺 |
| MX2009002302A (es) | 2006-09-01 | 2009-03-13 | Dow Agrosciences Llc | Sulfoximinas de (2-sustituido-1,3-tiazol) alquilo n-sustituidas insecticidas. |
| DE102006057036A1 (de) | 2006-12-04 | 2008-06-05 | Bayer Cropscience Ag | Biphenylsubstituierte spirocyclische Ketoenole |
| EP1950303A1 (de) * | 2007-01-26 | 2008-07-30 | Bayer CropScience AG | Genetisch modifizierte Pflanzen, die eine Stärke mit geringem Amylosegehalt und erhöhtem Quellvermögen synthetisieren |
| BRPI0721218A2 (pt) | 2007-02-09 | 2013-01-01 | Dow Agrosciences Llc | processo para a oxidação de determinadas sulfiliminas substituìdas para sulfoximinas inseticidas |
| MX2010000593A (es) * | 2007-07-20 | 2010-08-04 | Dow Agrosciences Llc | Aumentador del vigor de una planta. |
| JP5320460B2 (ja) * | 2008-05-01 | 2013-10-23 | ダウ アグロサイエンシィズ エルエルシー | 相乗的殺虫混合物 |
| CA2732353C (en) * | 2008-08-12 | 2013-09-24 | Jonathan M. Babcock | Synergistic pesticidal compositions comprising an active compound, an ammonium salt, and a nonionic surfactant |
-
2008
- 2008-08-29 DE DE102008041695A patent/DE102008041695A1/de not_active Withdrawn
-
2009
- 2009-08-17 ES ES09778023.3T patent/ES2677600T3/es active Active
- 2009-08-17 US US13/061,360 patent/US8796175B2/en not_active Expired - Fee Related
- 2009-08-17 EP EP09778023.3A patent/EP2323488B1/de not_active Not-in-force
- 2009-08-17 WO PCT/EP2009/006064 patent/WO2010022897A2/de not_active Ceased
- 2009-08-17 CN CN200980142927.6A patent/CN102196727B/zh not_active Expired - Fee Related
- 2009-08-17 MX MX2011001934A patent/MX2011001934A/es active IP Right Grant
- 2009-08-17 JP JP2011524237A patent/JP5583669B2/ja not_active Expired - Fee Related
- 2009-08-17 KR KR1020117007081A patent/KR101649556B1/ko not_active Expired - Fee Related
- 2009-08-17 BR BRPI0917104A patent/BRPI0917104B1/pt not_active IP Right Cessation
- 2009-08-17 TR TR2018/07878T patent/TR201807878T4/tr unknown
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2012000946A2 (de) | 2010-06-30 | 2012-01-05 | Bayer Cropscience Ag | Wirkstoffkombinationen |
| EP3146841A2 (de) | 2010-06-30 | 2017-03-29 | Bayer Intellectual Property GmbH | Wirkstoffkombinationen |
| JP2012250978A (ja) * | 2011-05-10 | 2012-12-20 | Sumitomo Chemical Co Ltd | 植物の生長を促進する方法 |
| JP2012250979A (ja) * | 2011-05-10 | 2012-12-20 | Sumitomo Chemical Co Ltd | 植物の生長を促進する方法 |
| US20230189805A1 (en) * | 2020-05-15 | 2023-06-22 | Clarke Mosquito Control Products, Inc. | Multi-solvent insecticidal compositions including sulfoximine |
Also Published As
| Publication number | Publication date |
|---|---|
| DE102008041695A1 (de) | 2010-03-04 |
| ES2677600T3 (es) | 2018-08-03 |
| JP2012500816A (ja) | 2012-01-12 |
| TR201807878T4 (tr) | 2018-06-21 |
| CN102196727B (zh) | 2015-07-29 |
| MX2011001934A (es) | 2011-03-29 |
| WO2010022897A3 (de) | 2011-01-06 |
| US8796175B2 (en) | 2014-08-05 |
| EP2323488A2 (de) | 2011-05-25 |
| KR20110044925A (ko) | 2011-05-02 |
| CN102196727A (zh) | 2011-09-21 |
| BRPI0917104B1 (pt) | 2017-01-17 |
| KR101649556B1 (ko) | 2016-08-19 |
| EP2323488B1 (de) | 2018-04-18 |
| BRPI0917104A2 (pt) | 2015-07-28 |
| US20110160061A1 (en) | 2011-06-30 |
| JP5583669B2 (ja) | 2014-09-03 |
| BRPI0917104A8 (pt) | 2016-03-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2323488B1 (de) | Methode zur verbesserung des pflanzenwachstums | |
| US11191270B2 (en) | Pesticide composition comprising a tetrazolyloxime derivative and a fungicide or an insecticide active substance | |
| CN101820763B (zh) | 改善植物生长的方法 | |
| CN102448307B (zh) | 琥珀酸脱氢酶抑制剂用于提高植物或植物部位对非生物应激的抗性的用途 | |
| CN102118971B (zh) | 唑类用于提高植物或植物部分的非生物胁迫抗性的用途 | |
| EP2044841A1 (de) | Methode zur Verbesserung des Pflanzenwachstums | |
| WO2016001122A1 (de) | Methoden zur verbesserung des pflanzenwachstums | |
| EP2090168A1 (de) | Methode zur Verbesserung des Pflanzenwachstums |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980142927.6 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09778023 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009778023 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2011/001934 Country of ref document: MX |
|
| ENP | Entry into the national phase |
Ref document number: 2011524237 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13061360 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20117007081 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: PI0917104 Country of ref document: BR Kind code of ref document: A2 Effective date: 20110224 |