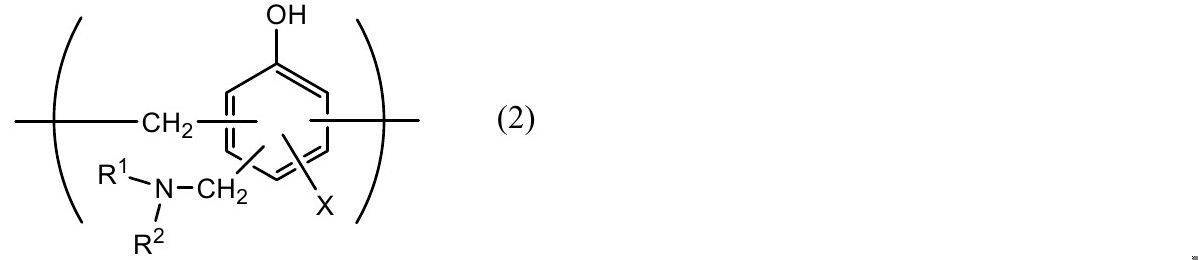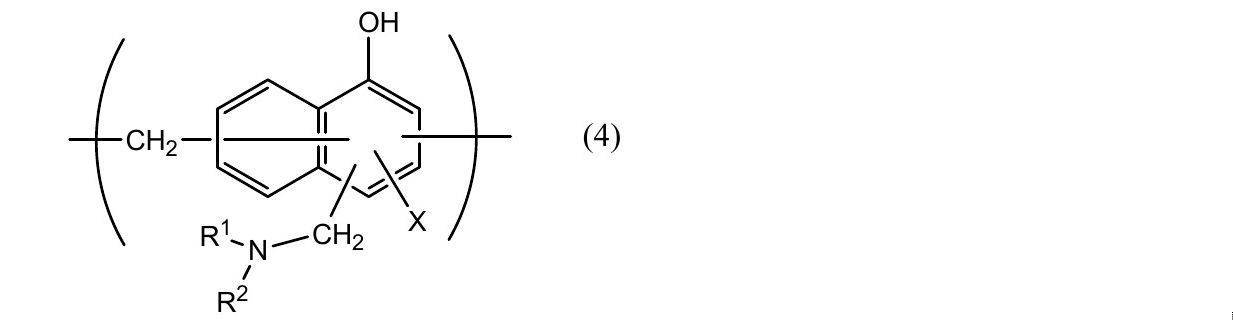WO2014208518A1 - 樹脂組成物 - Google Patents
樹脂組成物 Download PDFInfo
- Publication number
- WO2014208518A1 WO2014208518A1 PCT/JP2014/066616 JP2014066616W WO2014208518A1 WO 2014208518 A1 WO2014208518 A1 WO 2014208518A1 JP 2014066616 W JP2014066616 W JP 2014066616W WO 2014208518 A1 WO2014208518 A1 WO 2014208518A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- meth
- acrylate
- resin composition
- layer
- group
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J175/00—Adhesives based on polyureas or polyurethanes; Adhesives based on derivatives of such polymers
- C09J175/04—Polyurethanes
- C09J175/14—Polyurethanes having carbon-to-carbon unsaturated bonds
- C09J175/16—Polyurethanes having carbon-to-carbon unsaturated bonds having terminal carbon-to-carbon unsaturated bonds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/67—Unsaturated compounds having active hydrogen
- C08G18/671—Unsaturated compounds having only one group containing active hydrogen
- C08G18/672—Esters of acrylic or alkyl acrylic acid having only one group containing active hydrogen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/70—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the isocyanates or isothiocyanates used
- C08G18/81—Unsaturated isocyanates or isothiocyanates
- C08G18/8108—Unsaturated isocyanates or isothiocyanates having only one isocyanate or isothiocyanate group
- C08G18/8116—Unsaturated isocyanates or isothiocyanates having only one isocyanate or isothiocyanate group esters of acrylic or alkylacrylic acid having only one isocyanate or isothiocyanate group
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L63/00—Compositions of epoxy resins; Compositions of derivatives of epoxy resins
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/10—Primary casings; Jackets or wrappings
- H01M50/116—Primary casings; Jackets or wrappings characterised by the material
- H01M50/124—Primary casings; Jackets or wrappings characterised by the material having a layered structure
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F222/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a carboxyl radical and containing at least one other carboxyl radical in the molecule; Salts, anhydrides, esters, amides, imides, or nitriles thereof
- C08F222/10—Esters
- C08F222/1006—Esters of polyhydric alcohols or polyhydric phenols
- C08F222/106—Esters of polycondensation macromers
- C08F222/1065—Esters of polycondensation macromers of alcohol terminated (poly)urethanes, e.g. urethane(meth)acrylates
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
Definitions
- the present invention relates to a resin composition capable of firmly bonding a plurality of members in a short time. Furthermore, this invention relates to the packaging material for batteries which used the hardened
- adhesives for bonding between the same or different members such as resin and resin, resin and metal are known.
- a resin composition is generally used.
- an adhesive made of a resin composition is used in order to bond a base material layer formed of a resin and a metal layer.
- polyurethane adhesives are often used as adhesives for bonding between a base material layer and a metal layer.
- an aging process for heating for a long time is provided in order to increase the adhesive force. ing.
- Patent Document 1 discloses an adhesive composition for laminating, which has an ionizing radiation reactive group and a curing reactive group or a crosslinking reactive group due to heat and / or humidity. Things are disclosed.
- Patent Document 2 discloses a method for improving the adhesion of each layer in a laminated film by using an ionizing radiation curable adhesive composition containing dimer acid or a dimer acid derivative.
- the present inventor bonded the base material layer and the metal layer via the adhesive composition as disclosed in Patent Documents 1 and 2 to produce a battery packaging material. Then, in order to confirm the moisture and heat resistance of these battery packaging materials, when the battery packaging material is placed in a humid heat environment, the base material layer and the metal layer may be peeled off from the adhesive layer. It became clear that the strength could not be maintained.
- the present invention has been made in view of these problems.
- the main object of the present invention is to provide a resin composition capable of firmly bonding a plurality of members in a short time without providing a long aging step.
- another object of the present invention is to provide a battery packaging material in which a base material layer and a metal layer are firmly bonded using the resin composition and are excellent in moisture and heat resistance.
- the present inventor has intensively studied to solve the above problems. As a result, selected from the group consisting of (1) urethane (meth) acrylate, (2) (meth) acrylate having a phosphate group, (meth) acrylic silane coupling agent, and (meth) acrylate having an isocyanate group It is clear that by using a resin composition containing at least one kind and (3) (meth) acrylate having an epoxy group, a plurality of members can be firmly bonded without providing a long-time aging process. It became. Furthermore, it is also clear that by bonding the base material layer and the metal layer using the resin composition, the base material layer and the metal layer are firmly bonded, and a battery packaging material having excellent moisture and heat resistance can be obtained. became. The present invention has been completed by further studies based on these findings.
- this invention provides the resin composition of the aspect hung up below, the battery packaging material which adhere
- Item 1 (1) urethane (meth) acrylate, (2) at least one selected from the group consisting of a (meth) acrylate having a phosphate group, a (meth) acrylic silane coupling agent, and a (meth) acrylate having an isocyanate group; (3) (meth) acrylate having an epoxy group;
- a resin composition comprising: Item 2.
- the (1) urethane (meth) acrylate, the (2) (meth) acrylate having a phosphate group, a (meth) acrylic silane coupling agent, and an (meth) isocyanate group is 99.8: 0.
- the mass ratio ((1) :( 2) + (3)) of at least one selected from the group consisting of acrylates and (3) the total of (meth) acrylates having an epoxy group is 99.8: 0.
- the resin composition according to Item 1 which is in the range of 2 to 80:20.
- the resin composition according to Item 1 or 2 which is cured by heating or irradiation with ionizing radiation.
- Item 5 At least a base material layer, an adhesive layer, a metal layer, and a sealant layer are laminated in this order, Item 4.
- At least a method for producing a battery packaging material comprising a step of laminating a base material layer, an adhesive layer, a metal layer, and a sealant layer in this order to obtain a laminate, In the step, from the group consisting of (1) urethane (meth) acrylate, (2) (meth) acrylate having a phosphate group, (meth) acrylic silane coupling agent, and (meth) acrylate having an isocyanate group.
- the manufacturing method of the packaging material for batteries which forms the said contact bonding layer by hardening the resin composition containing at least 1 sort (s) selected and (3) (meth) acrylate which has an epoxy group.
- Item 4. A cured product of the resin composition according to any one of Items 1 to 3, comprising a laminate in which at least a base material layer, an adhesive layer, a metal layer, and a sealant layer are laminated in this order.
- the present invention it is possible to provide a resin composition capable of firmly bonding a plurality of members in a short time. Moreover, according to this invention, the cured resin composition formed by hardening
- the resin composition of the present invention comprises (1) urethane (meth) acrylate, (2) (meth) acrylate having a phosphate group, (meth) acrylic silane coupling agent, and (meth) acrylate having an isocyanate group. And at least one selected from the group consisting of (3) and (meth) acrylate having an epoxy group.
- the resin composition of the present invention, the battery packaging material in which the base material layer and the metal layer are bonded using the resin composition, and the production method thereof will be described in detail.
- the resin composition of the present invention has (1) urethane (meth) acrylate, (2) (meth) acrylate having a phosphate group, (meth) acrylic silane coupling agent, and an isocyanate group ( At least one selected from the group consisting of (meth) acrylates and (3) (meth) acrylates having an epoxy group.
- urethane (meth) acrylate is a general term for urethane acrylate and urethane methacrylate.
- a compound containing (meth) such as (meth) acrylate is a general term for a compound having “meta” in the name and a compound not having “meta”.
- the urethane (meth) acrylate is not particularly limited as long as it is a compound having one or more (meth) acryloyl groups and one or more urethane bonds in the molecule.
- the urethane (meth) acrylate of the present invention is a compound obtained by esterifying a polyurethane oligomer obtained by reaction of a polyisocyanate with a polyhydric alcohol such as polyether polyol or polyester polyol with (meth) acrylic acid. .
- the number of (meth) acryloyl groups in the molecule of urethane (meth) acrylate is not particularly limited, but from the viewpoint of increasing the curing rate of the resin composition and further strengthening the adhesion between members, for example, 2
- the number is preferably at least 2, more preferably 2 to 6, and even more preferably 2 to 4.
- the number of (meth) acryloyl groups is one, the curing rate of the resin composition is slow, so the practicality may be lowered.
- the number of (meth) acryloyl groups exceeds 6, the number of crosslinks increases, and the cured product (cured resin composition) of the resin composition becomes too hard, and the adhesive strength may be weakened.
- the weight average molecular weight of the urethane (meth) acrylate is not particularly limited, but from the viewpoint of further improving the moldability of the bonded member while further increasing the curing rate of the resin composition and further strengthening the adhesion between the members.
- it is preferably in the range of about 6,000 to 15,000, and more preferably in the range of about 8,000 to 13,000.
- the weight average molecular weight of the urethane (meth) acrylate is less than 6,000, the cured product of the resin composition may become too hard and the adhesive strength may be weakened.
- the weight average molecular weight of urethane (meth) acrylate is a value measured by gel permeation chromatography (GPC) using polystyrene as a standard sample.
- the apparent molecular weight between the crosslinking points of the urethane (meth) acrylate is not particularly limited, but it is, for example, in the range of about 2,000 to 15,000. Is more preferable, and it is more preferably in the range of about 3,000 to 15,000.
- the curing rate of the resin composition is further increased, and the adhesion between members is further strengthened.
- the moldability of the bonded member can be further improved.
- the apparent molecular weight (Y) between cross-linking points is a value calculated according to the following formula.
- Y (M ⁇ X) ⁇ 2
- M number average molecular weight of urethane (meth) acrylate
- X number of (meth) acryloyl groups
- the apparent crosslinking point molecular weight in the present invention means the molecular weight between (meth) acryloyl groups in the molecule of urethane (meth) acrylate.
- the number average molecular weight of urethane (meth) acrylate is a value measured by gel permeation chromatography (GPC) using polystyrene as a standard sample.
- the apparent molecular weight between cross-linking points of (meth) acrylate can be appropriately selected in consideration of the number average molecular weight of urethane (meth) acrylate and the number of (meth) acryloyl groups.
- Urethane (meth) acrylate can be produced by a known method, and a commercially available product can also be used. Only one type of urethane (meth) acrylate may be used, or two or more types may be used.
- the (meth) acrylate having a phosphate group is not particularly limited as long as it is a compound having a phosphate group and a (meth) acryloyl group in the molecule. Since the (meth) acrylate having a phosphoric acid group has a (meth) acryloyl group in the molecule, it functions as a curing agent that reacts with the (meth) acryloyl group of the urethane (meth) acrylate to cure the resin composition. .
- Examples of the (meth) acrylate having a phosphoric acid group include phosphate esters of (meth) acrylate such as 2-acryloyloxyethyl acid phosphate and 2-acryloyloxybutyl acid phosphate.
- phosphate esters of (meth) acrylate such as 2-acryloyloxyethyl acid phosphate and 2-acryloyloxybutyl acid phosphate.
- As the (meth) acrylate having a phosphoric acid group one produced by a known method can be used, and a commercially available product can also be used. Only one type of (meth) acrylate having a phosphoric acid group may be used, or two or more types may be used.
- the (meth) acrylic silane coupling agent is not particularly limited as long as it is a silane coupling agent having a (meth) acryloyl group in the molecule. Since the (meth) acrylic silane coupling agent has a (meth) acryloyl group in the molecule, it functions as a curing agent that reacts with the (meth) acryloyl group of urethane (meth) acrylate to cure the resin composition. .
- Examples of (meth) acrylic silane coupling agents include 3-acryloxypropyltrimethoxysilane, 3-acryloxypropyltriethoxysilane, 3-methacryloxypropyltrimethoxysilane, 3-methacryloxypropylmethyldimethoxysilane, A silane coupling agent having an alkoxysilyl group and a (meth) acryloyl group in the molecule, such as 3-methacryloxypropyltriethoxysilane and 3-methacryloxypropylethyldiethoxysilane, can be used.
- the (meth) acrylic silane coupling agent those produced by known methods can be used, and commercially available products can also be used. Only one type of (meth) acrylic silane coupling agent may be used, or two or more types may be used.
- the (meth) acrylate having an isocyanate group is not particularly limited as long as it is a compound having an isocyanate group and a (meth) acryloyl group in the molecule. Since the (meth) acrylate having an isocyanate group has a (meth) acryloyl group in the molecule, it reacts with the (meth) acryloyl group of the urethane (meth) acrylate and functions as a curing agent that cures the resin composition.
- the (meth) acrylate having an isocyanate group for example, (meth) acrylic acid such as 2-isocyanatoethyl acrylate and 2- (2-methacryloyloxyethyloxy) ethyl isocyanate and a hydroxyl group-containing isocyanate compound
- an esterified product can be used.
- the (meth) acrylate having an isocyanate group one produced by a known method can be used, and a commercially available product can also be used. Only one type of (meth) acrylate having an isocyanate group may be used, or two or more types may be used.
- the (meth) acrylate having an epoxy group is not particularly limited as long as it is a compound having an epoxy group and a (meth) acryloyl group in the molecule.
- the (meth) acryloyl group which has an epoxy group reacts with the (meth) acryloyl group of urethane (meth) acrylate, and functions as a hardening
- the (meth) acrylate having an epoxy group for example, a compound having an epoxy group and a (meth) acryloyl group such as bisphenol A type epoxy acrylate and 4-hydroxybutyl acrylate glycidyl ether can be used.
- (1) mass of urethane (meth) acrylate, (2) (meth) acrylate having a phosphate group, (meth) acrylic silane coupling agent, and (meth) acrylate having an isocyanate group is not particularly limited. It is preferably in the range of 99.8: 0.2 to 80:20, and more preferably in the range of 98: 2 to 90:10. When the mass ratio is in such a range, a plurality of members can be firmly bonded in a short time.
- the mass ratio of urethane (meth) acrylate to (meth) acrylate having a phosphate group is preferably in the range of 99.8: 0.2 to 80:20, and 99: 1 to More preferably, it is in the range of 90:10.
- the mass ratio of urethane (meth) acrylate and (meth) acrylic silane coupling agent is preferably in the range of 99.9: 0.1 to 95: 5, and 99.8: 0.2. More preferably, it is in the range of ⁇ 98: 2.
- the mass ratio of urethane (meth) acrylate and (meth) acrylate having an isocyanate group is preferably in the range of 99.8: 0.2 to 80:20, and is preferably 99: 1 to 90:10. More preferably, it is in the range.
- the weight ratio of urethane (meth) acrylate to (meth) acrylate having an epoxy group is preferably in the range of 99.8: 0.2 to 80:20, and is preferably 99: 1 to 90:10. More preferably, it is in the range.
- the resin composition of the present invention may further contain other resins for the purpose of increasing the curing rate or enhancing the adhesiveness.
- resin a well-known thing can be used and a commercial item can also be used.
- an acrylic resin, an epoxy resin, etc. are mentioned.
- acrylic resin for example, an acrylic resin acrylate can be used.
- epoxy resin for example, a glycidyl ether type epoxy resin can be used.
- the mass ratio of the urethane (meth) acrylate and the other resin is not particularly limited, but is preferably in the range of 99: 1 to 70:30, and 95: 5 More preferably, it is in the range of 80:20.
- the mass ratio is in such a range, the curing rate of the resin composition can be further increased, and adhesion between members can be further strengthened.
- the method for curing the resin composition of the present invention is not particularly limited.
- heating or ionizing radiation irradiation is preferable, and ionizing radiation irradiation is more preferable.
- ionizing radiation means an electromagnetic wave or charged particle beam having an energy quantum capable of polymerizing or cross-linking molecules, and usually ultraviolet (UV) or electron beam (EB) is used.
- electromagnetic waves such as rays and ⁇ rays, and charged particle rays such as ⁇ rays and ion rays.
- the curing conditions for the resin composition can be appropriately set according to urethane (meth) acrylate, the type of member to be bonded, and the like.
- the heating temperature when the resin composition is cured by heating is not particularly limited, but is usually about 30 ° C. to 70 ° C., preferably about 35 ° C. to 45 ° C.
- the heating time is not particularly limited, but can be usually about 24 to 120 hours, preferably about 48 to 72 hours.
- the ionizing radiation is not particularly limited, but for example, ultraviolet rays and electron beams can be used, and electron beams are preferable.
- the irradiation condition of ionizing radiation is usually about 25 kGy to 200 kGy, preferably about 30 kGy to 100 kGy.
- the resin composition of the present invention can be used as an adhesive for bonding two or more members.
- a cured resin composition is obtained, and a plurality of members can be firmly bonded in a short time.
- As a kind of member to adhere resin, a metal, etc. are mentioned, for example.
- the shape of the member to be bonded include a plate shape and a film shape.
- the thickness of the resin composition of the present invention can also be appropriately set depending on the type and shape of the member to be bonded, but is usually about 2 ⁇ m to 15 ⁇ m, preferably about 3 ⁇ m to 8 ⁇ m.
- the resin composition of the present invention can be suitably used particularly as an adhesive for bonding between films of laminated films.
- the above resin composition is applied to at least one film, and the other film is laminated using a dry laminating method, etc.
- a laminated film can be produced by curing the resin composition by irradiating the composition with ionizing radiation.
- the resin composition of the present invention can be firmly bonded not only between the same kind of materials such as resin and resin but also between different kinds of materials such as resin and metal in a short time. Can be suitably used.
- Specific examples of the laminated film in which the resin composition of the present invention can be suitably used as an adhesive include battery packaging materials.
- the structure of the packaging material for batteries manufactured using the resin composition of this invention is explained in full detail.
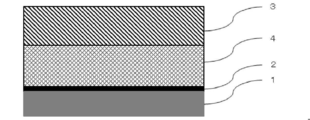
- the battery packaging material of the present invention includes at least a base material layer 1, an adhesive layer 2 formed of a cured product of the above resin composition, a metal layer 3, and a sealant.
- the layer 4 is composed of a laminated body laminated in this order.
- the base material layer 1 is the outermost layer
- the sealant layer 4 is the innermost layer. That is, when the battery is assembled, the battery element is wrapped with the battery packaging material so that the sealant layer 4 of the battery packaging material is on the inside of the battery, and the sealant layers 4 positioned at the periphery of the battery element are heat-welded. The battery element is sealed by sealing the battery element.
- the battery packaging material of the present invention only needs to include at least the base material layer 1, the adhesive layer 2, the metal layer 3, and the sealant layer 4, and may further include other layers.
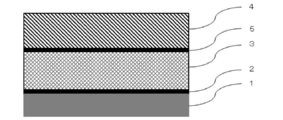
- an adhesive layer 5 may be provided between the metal layer 2 and the sealant layer 4 as necessary for the purpose of further improving the adhesion. Good.
- other layers may be further formed on the outside of the base material layer 1 for the purpose of, for example, imparting design properties.
- the base material layer 1 is a layer constituting the outermost layer when the battery is assembled.
- the raw material which forms the base material layer 1 will not be restrict
- the material for forming the base material layer 1 include resin films such as polyester resin, polyamide resin, epoxy resin, acrylic resin, fluorine resin, polyurethane resin, silicon resin, phenol resin, and mixtures and copolymers thereof. Can be mentioned.
- polyester resin examples include polyethylene terephthalate, polybutylene terephthalate, polyethylene naphthalate, polybutylene naphthalate, copolymerized polyester, and polycarbonate.
- polyamide resin examples include nylon 6, nylon 6,6, a copolymer of nylon 6 and nylon 6,6, nylon 6,10, polymetaxylylene adipamide (MXD6), and the like. It is done.
- the material forming the base layer 1 is preferably nylon or polyester, more preferably biaxially stretched nylon, biaxially stretched polyester, and particularly preferably biaxially stretched nylon.
- the base material layer 1 may be formed from one resin film or may be formed from two or more multilayer resin films.
- the pinhole resistance and insulation of the battery packaging material of the present invention can be improved.
- two or more resin films can be laminated
- the adhesive used at this time include the resin composition of the present invention and an adhesive for forming the adhesive layer 5 described later.
- the thickness of the base material layer 1 is not particularly limited, but can be, for example, about 10 ⁇ m to 50 ⁇ m, preferably about 15 ⁇ m to 30 ⁇ m.
- the metal layer 3 is a layer that functions as a barrier layer for preventing the penetration of water vapor, oxygen, light, etc. into the battery, in addition to improving the strength of the battery packaging material.
- a metal which comprises the metal layer 3 For example, aluminum, stainless steel, titanium etc. are mentioned, Preferably aluminum is mentioned.
- the metal layer 3 can be formed by metal foil, metal vapor deposition, or the like, preferably by metal foil, and more preferably by aluminum foil.
- the thickness of the metal layer 3 is not particularly limited, but can be, for example, about 10 ⁇ m to 200 ⁇ m, preferably about 20 to 100 ⁇ m.
- the metal layer 3 is preferably subjected to chemical conversion treatment on at least one side, preferably both sides, in order to stabilize adhesion, prevent dissolution and corrosion, and the like.
- the chemical conversion treatment refers to a treatment for forming an acid-resistant film on the surface of the metal layer.
- chromic acid compounds such as chromium nitrate, chromium fluoride, chromium sulfate, chromium acetate, chromium oxalate, chromium biphosphate, chromic acetyl acetate, chromium chloride, potassium sulfate chromium, etc.
- Phosphoric acid chromate treatment using a phosphoric acid compound such as sodium phosphate, potassium phosphate, ammonium phosphate, polyphosphoric acid; aminated phenol having a repeating unit represented by the following general formulas (1) to (4) Examples include chromate treatment using a polymer.
- X represents a hydrogen atom, a hydroxyl group, an alkyl group, a hydroxyalkyl group, an allyl group or a benzyl group.
- R 1 and R 2 are the same or different and each represents a hydroxyl group, an alkyl group, or a hydroxyalkyl group.
- examples of the alkyl group represented by X, R 1 and R 2 include a methyl group, an ethyl group, an n-propyl group, an isopropyl group, an n-butyl group, an isobutyl group, Examples thereof include a linear or branched alkyl group having 1 to 4 carbon atoms such as a tert-butyl group.
- Examples of the hydroxyalkyl group represented by X, R 1 and R 2 include a hydroxymethyl group, a 1-hydroxyethyl group, a 2-hydroxyethyl group, a 1-hydroxypropyl group, a 2-hydroxypropyl group, 3- C1-C4 straight or branched chain in which one hydroxy group such as hydroxypropyl group, 1-hydroxybutyl group, 2-hydroxybutyl group, 3-hydroxybutyl group, 4-hydroxybutyl group is substituted
- An alkyl group is mentioned.
- the alkyl group and hydroxyalkyl group represented by X, R 1 and R 2 may be the same or different.
- X is preferably a hydrogen atom, a hydroxyl group or a hydroxyalkyl group.
- the number average molecular weight of the aminated phenol polymer having a repeating unit represented by the general formulas (1) to (4) is preferably, for example, 500 to 1,000,000, more preferably about 1,000 to 20,000. preferable.
- a metal oxide such as aluminum oxide, titanium oxide, cerium oxide, tin oxide, or barium sulfate fine particles dispersed in phosphoric acid is coated.
- a method of forming a corrosion-resistant treatment layer on the surface of the metal layer 3 by performing a baking treatment at 150 ° C. or higher can be mentioned.
- a resin layer obtained by crosslinking a cationic polymer with a crosslinking agent may be further formed on the corrosion-resistant treatment layer.
- examples of the cationic polymer include polyethyleneimine, an ionic polymer complex composed of a polymer having polyethyleneimine and a carboxylic acid, a primary amine graft acrylic resin obtained by graft polymerization of a primary amine on an acrylic main skeleton, and polyallylamine. Or the derivative, aminophenol, etc. are mentioned.
- these cationic polymers only one type may be used, or two or more types may be used in combination.
- examples of the crosslinking agent include a compound having at least one functional group selected from the group consisting of an isocyanate group, a glycidyl group, a carboxyl group, and an oxazoline group, and a silane coupling agent. As these crosslinking agents, only one type may be used, or two or more types may be used in combination.
- chemical conversion treatment only one type of chemical conversion treatment may be performed, or two or more types of chemical conversion processing may be performed in combination. Furthermore, these chemical conversion treatments may be carried out using one kind of compound alone, or may be carried out using a combination of two or more kinds of compounds.
- chemical conversion treatments chromic acid chromate treatment, chromate treatment combining a chromic acid compound, a phosphoric acid compound, and an aminated phenol polymer are preferable.
- the amount of the acid-resistant film formed on the surface of the metal layer 3 in the chemical conversion treatment is not particularly limited.
- a chromic acid compound is present per 1 m 2 of the surface of the metal layer 3.
- a solution containing a compound used for forming an acid-resistant film is applied to the surface of the metal layer by a bar coating method, a roll coating method, a gravure coating method, an immersion method, or the like, and then the temperature of the metal layer is 70. It is carried out by heating so as to reach about 200 ° C.
- the metal layer may be previously subjected to a degreasing treatment by an alkali dipping method, an electrolytic cleaning method, an acid cleaning method, an electrolytic acid cleaning method, or the like. By performing the degreasing treatment in this way, it becomes possible to more efficiently perform the chemical conversion treatment on the surface of the metal layer.
- the adhesive layer 2 is a layer provided between the base material layer 1 and the metal layer 3 in order to adhere the base material layer 1 and the metal layer 3.
- the adhesive layer 2 can be formed of a cured product of the resin composition of the present invention.
- the composition and curing method of the resin composition can be the same as described above.
- the thickness of the adhesive layer 2 is not particularly limited and may be appropriately set depending on the total thickness and strength of the target battery packaging material, but is usually about 2 ⁇ m to 15 ⁇ m, preferably about 3 ⁇ m to 8 ⁇ m. .
- the adhesive layer 2 is formed of a cured product of the specific resin composition, the base material layer 1 and the base material layer 3 are firmly bonded, and is resistant to heat and moisture. Also excellent.
- the sealant layer 4 is a layer constituting the innermost layer of the battery packaging material of the present invention when the battery is assembled. When the battery is assembled, the surfaces of the sealant layer 4 can be brought into contact with each other, and the contacted portion can be heat-welded to seal the battery element.
- the resin component for forming the sealant layer 4 is not particularly limited as long as the sealant layers 4 can be thermally welded to each other.
- examples of such a resin component include polyolefin, cyclic polyolefin, carboxylic acid-modified polyolefin, carboxylic acid-modified cyclic polyolefin, and the like.
- polyolefins include polyethylene such as low density polyethylene, medium density polyethylene, high density polyethylene, and linear low density polyethylene; homopolypropylene, polypropylene block copolymers (for example, block copolymers of propylene and ethylene), polypropylene Crystalline or amorphous polypropylene such as random copolymers (for example, random copolymers of propylene and ethylene); ethylene-butene-propylene terpolymers, and the like.
- polyethylene and polypropylene are preferable.
- Cyclic polyolefin is a copolymer of olefin and cyclic monomer.
- the olefin include ethylene, propylene, 4-methyl-1-pentene, styrene, butadiene, and isoprene.
- the cyclic monomer include cyclic alkenes such as norbornene; cyclic dienes such as cyclopentadiene, dicyclopentadiene, cyclohexadiene, norbornadiene, and the like.
- a cyclic alkene is preferable, and norbornene is more preferable.
- Carboxylic acid-modified polyolefin is a polymer obtained by modifying polyolefin with carboxylic acid.
- carboxylic acid used for modification include maleic acid, acrylic acid, itaconic acid, crotonic acid, maleic anhydride, itaconic anhydride and the like.
- the carboxylic acid-modified cyclic polyolefin is a copolymer obtained by copolymerizing a part of the monomer constituting the cyclic polyolefin in place of the ⁇ , ⁇ -unsaturated carboxylic acid or its anhydride, or ⁇ , ⁇ - It is a polymer obtained by block polymerization or graft polymerization of an unsaturated carboxylic acid or its anhydride.
- the cyclic polyolefin to be modified with carboxylic acid can be the same as the above cyclic polyolefin.
- the carboxylic acid used for modification can be the same as that used for modification of the acid-modified cycloolefin copolymer.
- these resin components preferably crystalline or amorphous polyolefin, cyclic polyolefin, and blended polymers thereof; more preferably polyethylene, polypropylene, copolymers of ethylene and norbornene, and two or more of these The blend polymer of these is mentioned.
- the sealant layer 4 may be formed of only one type of resin component or may be formed of a blend polymer obtained by combining two or more types of resin components. Furthermore, the sealant layer 4 may be formed of only one layer, or may be formed of two or more layers using the same or different resin components.
- the thickness of the sealant layer 4 is not particularly limited, but may be, for example, about 2 ⁇ m to 2000 ⁇ m, preferably about 5 ⁇ m to 1000 ⁇ m, and more preferably 10 ⁇ m to 500 ⁇ m.
- Adhesive layer 5 In the battery packaging material of the present invention, an adhesive layer is provided between the metal layer 3 and the sealant layer 4 for the purpose of firmly bonding the metal layer 3 and the sealant layer 4 as shown in FIG. 5 may be further provided.
- the adhesive layer 5 is formed of an adhesive component capable of bonding the metal layer 3 and the sealant layer 4.
- the adhesive used for forming the adhesive layer 5 is not particularly limited, and may be the resin composition of the present invention or other adhesive, but the metal layer 3 and the sealant layer 4 It is preferable to use the resin composition of the present invention from the viewpoint of firmly adhering to a short time.
- the other adhesive may be a two-component curable adhesive or a one-component curable adhesive.
- adhesive components of other adhesives that can be used for forming the adhesive layer 5 include polyesters such as polyethylene terephthalate, polybutylene terephthalate, polyethylene naphthalate, polybutylene naphthalate, polyethylene isophthalate, polycarbonate, and copolyester.
- Polyether adhesive Polyurethane adhesive; Epoxy resin; Phenol resin resin; Polyamide resin such as nylon 6, nylon 66, nylon 12, copolymer polyamide; polyolefin, carboxylic acid modified polyolefin, metal modified Polyolefin resins such as polyolefin; polyvinyl acetate resins; cellulose adhesives; (meth) acrylic resins; polyimide resins; amino resins such as urea resins and melamine resins; chloroprene rubber; - nitrile rubber, styrene rubber such as butadiene rubber; and silicone resins.
- These adhesive components may be used alone or in combination of two or more.
- the thickness of the adhesive layer 5 is not particularly limited, but is preferably about 1 ⁇ m to 40 ⁇ m, for example, and more preferably about 2 ⁇ m to 30 ⁇ m.
- Production method of battery packaging material is not particularly limited as long as the above-described laminate in which the respective layers having a predetermined composition are laminated is obtained.
- the following method is exemplified. be able to.
- At least the base material layer 1, the adhesive layer 2, the metal layer 3, and the sealant layer 4 are laminated in this order to obtain a laminate.
- a solvent containing the above resin composition is applied to at least one of the base material layer 1 and the metal layer 3.
- the solvent can be applied by an application method such as an extrusion method, a gravure coating method, or a roll coating method.
- the solvent is dried.
- the base material layer 1 and the metal layer 3 are bonded and laminated.
- Lamination of the base material layer 1 and the metal layer 3 through the resin composition can be performed by, for example, a dry lamination method.
- the resin composition located between the base material layer 1 and the metal layer 3 is cured by ionizing radiation irradiation or heating to form the adhesive layer 2.
- the base material layer 1 when the base material layer 1 is transparent, it is preferable to cure the resin composition by irradiating ionizing radiation from the base material layer 1 side.
- the ionizing radiation irradiation can be performed using, for example, an ultraviolet irradiation device or an electron beam irradiation device.
- the irradiation condition of ionizing radiation is usually about 25 kGy to 200 kGy, preferably about 30 kGy to 100 kGy.
- heating can be performed by methods, such as a hot-roll contact system, a hot air system, a near or far-infrared system, for example.
- the heating temperature is usually about 30 ° C. to 70 ° C., preferably about 35 ° C.
- the heating time is usually about 24 hours to 120 hours, preferably about 48 hours to 72 hours.
- the sealant layer 4 is laminated on the surface of the metal layer 3 opposite to the base material layer 1 to obtain a battery packaging material.
- the lead time can be shortened because it is not necessary to perform a long-time aging process that is conventionally required.
- each layer constituting the laminate is improved or stabilized as necessary for film forming property, lamination processing, suitability for final processing (pouching, embossing), etc.
- surface activation treatment such as corona treatment, blast treatment, oxidation treatment, ozone treatment, or the like may be performed.
- the battery packaging material of the present invention is used as a packaging material for sealing and housing battery elements such as a positive electrode, a negative electrode, and an electrolyte.
- a battery element including at least a positive electrode, a negative electrode, and an electrolyte is formed with a flange portion (sealant layer) on the periphery of the battery element with metal terminals connected to each of the positive electrode and the negative electrode protruding outward.
- region which mutually contacts) can be formed, and it coat
- the battery sealed with the battery packaging material of the present invention is provided by heat-sealing and sealing the sealant layers of the flange part.
- the battery packaging material of the present invention is used so that the sealant layer 4 is on the inner side (surface in contact with the battery element).
- the battery packaging material of the present invention can be used for either a primary battery or a secondary battery, but is particularly suitable for a secondary battery.
- the type of the secondary battery to which the battery packaging material of the present invention is applied is not particularly limited.
- a lithium ion battery, a lithium ion polymer battery, a lead live battery, a nickel / hydrogen live battery, a nickel / cadmium live battery Nickel / iron livestock batteries, nickel / zinc livestock batteries, silver oxide / zinc livestock batteries, metal-air batteries, multivalent cation batteries, capacitors, capacitors, and the like are suitable applications for the battery packaging material of the present invention.
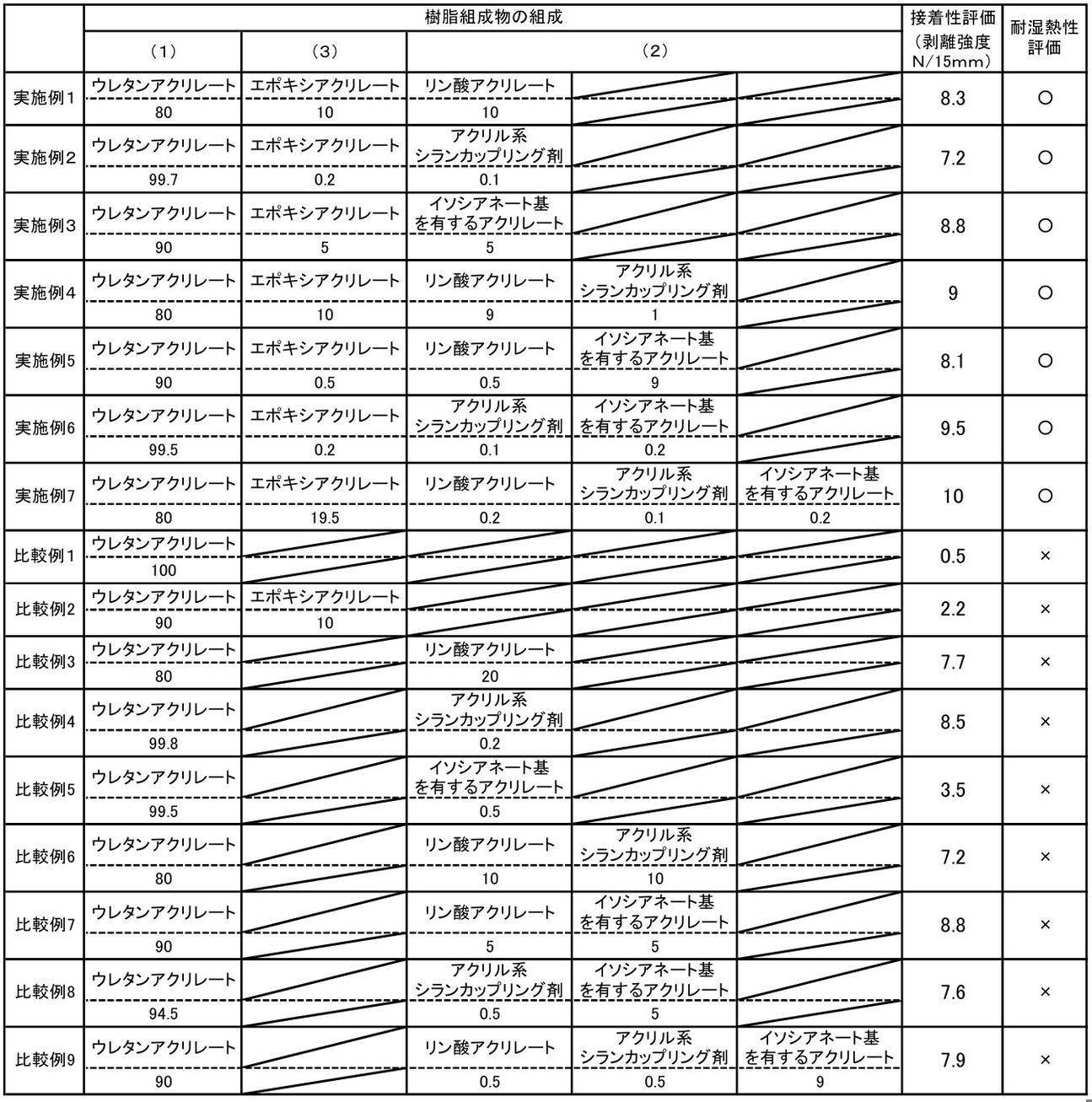
- Example 2 An acrylic silane coupling agent (KBM-5103 manufactured by Shin-Etsu Chemical Co., Ltd.) was used in place of phosphoric acid acrylate, and urethane acrylate, epoxy acrylate, and acrylic silane coupling agent were combined, 99.7: 0.2: A resin composition was obtained in the same manner as in Example 1 except that mixing was performed at a mass ratio (solid content ratio) of 0.1.
- KBM-5103 manufactured by Shin-Etsu Chemical Co., Ltd.
- Example 3 An acrylate having an isocyanate group (Karenz AOI manufactured by Showa Denko KK, compound name: 2-isocyanatoethyl acrylate) was used instead of phosphoric acid acrylate, and urethane acrylate, epoxy acrylate, and acrylate having an isocyanate group were : A resin composition was obtained in the same manner as in Example 1 except that mixing was performed at a mass ratio (solid content ratio) of 5: 5.
- Example 4 An acrylic silane coupling agent (KBM-5103 manufactured by Shin-Etsu Chemical Co., Ltd.) is further mixed, and urethane acrylate, epoxy acrylate, phosphoric acid acrylate, and acrylic silane coupling agent are mixed into 80: 10: 9: 1.
- a resin composition was obtained in the same manner as in Example 1 except that the mixing was performed at the mass ratio (solid content ratio).
- Example 5 An acrylate having an isocyanate group (Karenz AOI manufactured by Showa Denko KK, compound name: 2-isocyanatoethyl acrylate) is further mixed, and urethane acrylate, epoxy acrylate, phosphoric acid acrylate, and acrylate having an isocyanate group are A resin composition was obtained in the same manner as in Example 1 except that mixing was performed at a mass ratio (solid content ratio) of 90: 0.5: 0.5: 9.
- Example 6 An acrylic silane coupling agent (KBM-5103 manufactured by Shin-Etsu Chemical Co., Ltd.) is used instead of phosphoric acid acrylate, and urethane acrylate, epoxy acrylate, acrylic silane coupling agent, and acrylate having an isocyanate group are added to 99 Resin composition was obtained in the same manner as in Example 5 except that mixing was performed at a mass ratio (solid content ratio) of .5: 0.2: 0.1: 0.2.
- KBM-5103 manufactured by Shin-Etsu Chemical Co., Ltd.
- Example 7 An acrylate having an isocyanate group (Karenz AOI manufactured by Showa Denko KK, compound name: 2-isocyanatoethyl acrylate) is further mixed, and urethane acrylate, epoxy acrylate, phosphate acrylate, acrylic silane coupling agent, Resin composition in the same manner as in Example 4 except that the acrylate having an isocyanate group was mixed at a mass ratio (solid content ratio) of 80: 19.5: 0.2: 0.1: 0.2. I got a thing.
- Example 3 A resin composition was obtained in the same manner as in Example 1 except that urethane acrylate and phosphoric acid acrylate were mixed at a mass ratio (solid content ratio) of 80:20 without using epoxy acrylate.
- ⁇ Comparative example 4> Resin similar to Example 1 except that urethane acrylate and acrylic silane coupling agent were mixed in a mass ratio (solid content ratio) of 99.8: 0.2 without using epoxy acrylate. A composition was obtained.
- an aluminum foil (thickness 40 ⁇ m) made of soft aluminum ((JIS H4160 A8021H-O)) was used for chemical conversion treatment.
- a treatment solution comprising a chromium compound and phosphoric acid was applied to both surfaces of the metal layer by a roll coating method and baked for 20 seconds under the condition that the film temperature was 180 ° C. or higher.
- the resin compositions obtained in Examples 1 to 7 and Comparative Examples 1 to 9 were respectively applied on one surface of the aluminum foil so as to have a thickness of 4 ⁇ m.
- the stretched nylon film that forms the base material layer was bonded to the top of the layer by the dry laminating method.
- the resin composition was cured by irradiating an electron beam from above the stretched nylon film using an electron beam irradiation apparatus (acceleration voltage 120 kV, irradiation dose 50 kGy).
- an electron beam irradiation apparatus acceleration voltage 120 kV, irradiation dose 50 kGy.
- a sealant layer consisting of two layers, a carboxylic acid-modified polypropylene resin layer (thickness 23 ⁇ m) and a homopolypropylene resin layer (thickness 23 ⁇ m), is laminated, and the metal layer and the sealant layer are adhered by coextrusion processing.
- laminates were obtained using the resin compositions of Examples 1 to 7 and Comparative Examples 1 to 9. The laminate was so that the acid-modified polypropylene resin layer of the sealant layer was on the metal side.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Electrochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Laminated Bodies (AREA)
- Sealing Battery Cases Or Jackets (AREA)
- Adhesives Or Adhesive Processes (AREA)
- Epoxy Resins (AREA)
- Macromonomer-Based Addition Polymer (AREA)
- Electric Double-Layer Capacitors Or The Like (AREA)
Abstract
Description
項1. (1)ウレタン(メタ)アクリレートと、
(2)リン酸基を有する(メタ)アクリレート、(メタ)アクリル系シランカップリング剤、及びイソシアネート基を有する(メタ)アクリレートからなる群から選択された少なくとも1種と、
(3)エポキシ基を有する(メタ)アクリレートと、
を含む樹脂組成物。
項2. 前記樹脂組成物中において、前記(1)ウレタン(メタ)アクリレートと、前記(2)リン酸基を有する(メタ)アクリレート、(メタ)アクリル系シランカップリング剤、及びイソシアネート基を有する(メタ)アクリレートからなる群から選択された少なくとも1種並びに(3)エポキシ基を有する(メタ)アクリレートの合計との質量比((1):(2)+(3))が、99.8:0.2~80:20の範囲にある、項1に記載の樹脂組成物。
項3. 加熱または電離放射線照射により硬化する、項1または2に記載の樹脂組成物。
項4. 項1~3のいずれかに記載の樹脂組成物を硬化してなる、硬化樹脂組成物。
項5. 少なくとも、基材層と、接着層と、金属層と、シーラント層とがこの順に積層された積層体からなり、
前記接着層は、項1~3のいずれかに記載の樹脂組成物の硬化物により形成されてなる、電池用包装材料。
項6. 少なくとも、基材層と、接着層と、金属層と、シーラント層とをこの順に積層して積層体を得る工程を備える電池用包装材料の製造方法であって、
前記工程において、(1)ウレタン(メタ)アクリレートと、(2)リン酸基を有する(メタ)アクリレート、(メタ)アクリル系シランカップリング剤、及びイソシアネート基を有する(メタ)アクリレートからなる群から選択された少なくとも1種と、(3)エポキシ基を有する(メタ)アクリレートとを含む樹脂組成物を硬化させることにより前記接着層を形成する、電池用包装材料の製造方法。
項7. 少なくとも、基材層と、接着層と、金属層と、シーラント層とがこの順に積層された積層体からなる電池用包装材料の前記接着層への、項1~3のいずれかに記載の樹脂組成物の硬化物の使用。
項8. 少なくとも、基材層と、接着層と、金属層と、シーラント層とがこの順に積層された積層体からなり、前記接着層が、項1~3のいずれかに記載の樹脂組成物の硬化物により形成されてなる包装材料の、電池の製造のための使用。
本発明の樹脂組成物は、(1)ウレタン(メタ)アクリレートと、(2)リン酸基を有する(メタ)アクリレート、(メタ)アクリル系シランカップリング剤、及びイソシアネート基を有する(メタ)アクリレートからなる群から選択された少なくとも1種と、(3)エポキシ基を有する(メタ)アクリレートとを含む。なお、本発明において、ウレタン(メタ)アクリレートとは、ウレタンアクリレート及びウレタンメタアクリレートの総称である。(メタ)アクリレートなどの(メタ)を含む化合物なども同様に、名称中に「メタ」を有する化合物と「メタ」を有さない化合物の総称である。
Y=(M÷X)×2
M:ウレタン(メタ)アクリレートの数平均分子量
X:(メタ)アクリロイル基の数
本発明の電池用包装材料は、図1に示されるように、少なくとも、基材層1、上記の樹脂組成物の硬化物により形成された接着層2、金属層3、及びシーラント層4がこの順に積層された積層体からなることを特徴とする。電池用包装材料において、基材層1が最外層となり、シーラント層4は最内層になる。すなわち、電池の組み立て時に、電池用包装材料のシーラント層4が電池の内側となるようにして、電池用包装材料で電池素子を包み込み、電池素子の周縁に位置するシーラント層4同士を熱溶着して電池素子を密封することにより、電池素子が封止される。
[基材層1]
本発明の電池用包装材料において、基材層1は、電池を組み立てた時に、最外層を構成する層である。基材層1を形成する素材は、絶縁性を備えるものであれば特に制限されない。基材層1を形成する素材としては、例えば、ポリエステル樹脂、ポリアミド樹脂、エポキシ樹脂、アクリル樹脂、フッ素樹脂、ポリウレタン樹脂、珪素樹脂、フェノール樹脂、及びこれらの混合物や共重合物などの樹脂フィルムが挙げられる。
本発明の電池用包装材料において、金属層3は、電池用包装材料の強度向上の他、電池内部に水蒸気、酸素、光などが侵入することを防止するためのバリア層として機能する層である。金属層3を構成する金属としては、特に限定されないが、例えば、アルミニウム、ステンレス、チタンなどが挙げられ、好ましくはアルミニウムが挙げられる。金属層3は、金属箔や金属蒸着などにより形成することができ、金属箔により形成することが好ましく、アルミニウム箔により形成することがさらに好ましい。電池用包装材料の製造時に、金属層3にしわやピンホールが発生することを防止する観点からは、例えば、焼きなまし処理済みのアルミニウム(JIS A803P-O、JIS A8079P-O)など軟質アルミニウム箔により形成することが特に好ましい。
本発明の電池用包装材料において、接着層2は、基材層1と金属層3とを接着するために、基材層1と金属層3との間に設けられる層である。
本発明の電池用包装材料において、シーラント層4は、電池を組み立てた時に、本発明の電池用包装材料の最内層を構成する層である。電池の組み立て時に、シーラント層4の表面同士を互いに接触させ、接触した部分を熱溶着して電池素子を密封することができる。
本発明の電池用包装材料においては、金属層3とシーラント層4とを強固に接着させることなどを目的として、図2に示されるように、金属層3とシーラント層4との間に接着層5をさらに設けてもよい。
本発明の電池用包装材料の製造方法は、所定の組成の各層を積層させた上記の積層体が得られる限り、特に制限されないが、例えば、以下の方法を例示することができる。
本発明の電池用包装材料は、正極、負極、電解質などの電池素子を密封して収容するための包装材料として使用される。
アクリロイル基を2つ有するウレタンアクリレート(ダイセル・サイテック社製のEBECRYL9227EA、重量平均分子量6000)と、エポキシアクリレート(ダイセル・サイテック社製のEBECRYL3703)と、リン酸アクリレート(共栄社化学株式会社製のライトアクリレート P-1A(N))とを、80:10:10の質量比(固形分比)で混合して樹脂組成物を得た。
リン酸アクリレートの代わりにアクリル系シランカップリング剤(信越化学社製のKBM-5103)を用い、ウレタンアクリレートと、エポキシアクリレートと、アクリル系シランカップリング剤とを、99.7:0.2:0.1の質量比(固形分比)で混合したこと以外は、実施例1と同様にして、樹脂組成物を得た。
リン酸アクリレートの代わりにイソシアネート基を有するアクリレート(昭和電工社製のカレンズAOI、化合物名:2-イソシアナトエチルアクリレート)を用い、ウレタンアクリレートと、エポキシアクリレートと、イソシアネート基を有するアクリレートとを、90:5:5の質量比(固形分比)で混合したこと以外は、実施例1と同様にして、樹脂組成物を得た。
アクリル系シランカップリング剤(信越化学社製のKBM-5103)をさらに混合し、ウレタンアクリレートと、エポキシアクリレートと、リン酸アクリレートと、アクリル系シランカップリング剤とを、80:10:9:1の質量比(固形分比)で混合したこと以外は、実施例1と同様にして、樹脂組成物を得た。
イソシアネート基を有するアクリレート(昭和電工社製のカレンズAOI、化合物名:2-イソシアナトエチルアクリレート)をさらに混合し、ウレタンアクリレートと、エポキシアクリレートと、リン酸アクリレートと、イソシアネート基を有するアクリレートとを、90:0.5:0.5:9の質量比(固形分比)で混合したこと以外は、実施例1と同様にして、樹脂組成物を得た。
リン酸アクリレートの代わりにアクリル系シランカップリング剤(信越化学社製のKBM-5103)を用い、ウレタンアクリレートと、エポキシアクリレートと、アクリル系シランカップリング剤と、イソシアネート基を有するアクリレートとを、99.5:0.2:0.1:0.2の質量比(固形分比)で混合したこと以外は、実施例5と同様にして、樹脂組成物を得た。
イソシアネート基を有するアクリレート(昭和電工社製のカレンズAOI、化合物名:2-イソシアナトエチルアクリレート)をさらに混合し、ウレタンアクリレートと、エポキシアクリレートと、リン酸アクリレートと、アクリル系シランカップリング剤と、イソシアネート基を有するアクリレートとを、80:19.5:0.2:0.1:0.2の質量比(固形分比)で混合したこと以外は、実施例4と同様にして、樹脂組成物を得た。
エポキシアクリレートとリン酸アクリレートを用いなかったこと以外は、実施例1と同様にして、樹脂組成物を得た。
リン酸アクリレートを用いず、ウレタンアクリレートと、エポキシアクリレートとを、90:10の質量比(固形分比)で混合したこと以外は、実施例1と同様にして、樹脂組成物を得た。
エポキシアクリレートを用いず、ウレタンアクリレートと、リン酸アクリレートとを、80:20の質量比(固形分比)で混合したこと以外は、実施例1と同様にして、樹脂組成物を得た。
エポキシアクリレートを用いず、ウレタンアクリレートと、アクリル系シランカップリング剤とを、99.8:0.2の質量比(固形分比)で混合したこと以外は、実施例1と同様にして、樹脂組成物を得た。
エポキシアクリレートを用いず、ウレタンアクリレートと、イソシアネート基を有するアクリレートとを、99.5:0.5の質量比(固形分比)で混合したこと以外は、実施例1と同様にして、樹脂組成物を得た。
エポキシアクリレートを用いず、ウレタンアクリレートと、リン酸アクリレートと、アクリル系シランカップリング剤とを、80:10:10の質量比(固形分比)で混合したこと以外は、実施例4と同様にして、樹脂組成物を得た。
エポキシアクリレートを用いず、ウレタンアクリレートと、リン酸アクリレートと、イソシアネート基を有するアクリレートとを、90:5:5の質量比(固形分比)で混合したこと以外は、実施例5と同様にして、樹脂組成物を得た。
エポキシアクリレートを用いず、ウレタンアクリレートと、アクリル系シランカップリング剤と、イソシアネート基を有するアクリレートとを、94.5:0.5:5の質量比(固形分比)で混合したこと以外は、実施例6と同様にして、樹脂組成物を得た。
エポキシアクリレートを用いず、ウレタンアクリレートと、リン酸アクリレートと、アクリル系シランカップリング剤と、イソシアネート基を有するアクリレートとを、90:0.5:0.5:9の質量比(固形分比)で混合したこと以外は、実施例7と同様にして、樹脂組成物を得た。
金属層を形成するアルミニウム箔として、軟質アルミニウム((JIS H4160 A8021H-O)からなるアルミニウム箔(厚さ40μm)の両面を化成処理したものを用いた。アルミニウム箔の化成処理は、フェノール樹脂、フッ化クロム化合物、及びリン酸からなる処理液をロールコート法により金属層の両面に塗布し、皮膜温度が180℃以上となる条件で20秒間焼付けすることにより行った。次に、化成処理を行ったアルミニウム箔の一方の表面上に、実施例1~7及び比較例1~9で得られた樹脂組成物を、4μmの厚みとなるようにして、それぞれ塗布した。次に、樹脂組成物の層の上から基材層を形成する延伸ナイロンフィルムをドライラミネート法により貼り合わせた。なお、ドライラミネート法における乾燥温度は60℃とした。次に、延伸ナイロンフィルムの上から、電子線照射装置(加速電圧120kV、照射線量50kGy)を用いて電子線を照射し、樹脂組成物を硬化させた。次に、金属層の上から、カルボン酸変性ポリプロピレン系樹脂層(厚さ23μm)とホモポリプロピレン樹脂層(厚さ23μm)の2層からなるシーラント層を積層し、共押出加工により金属層とシーラント層とを接着させ、実施例1~7及び比較例1~9の樹脂組成物を用いた積層体を得た。なお、シーラント層の酸変性ポリプロピレン系樹脂層が金属側となるように積層させた。
実施例1~7及び比較例1~9の樹脂組成物を用いて得られた上記の積層体について、それぞれ15mm巾の短冊状に切り取り試験片を得た。次に、得られた試験片の延伸ナイロンフィルムとアルミニウム箔とを引張り機(島津製作所製の商品名AGS-50D)を用いて、50mm/分の速度で引張り、積層体の剥離強度(N/15mm)を測定した。結果を表1に示す。
実施例1~7及び比較例1~9の樹脂組成物を用いて得られた上記の積層体について、それぞれ80mm×120mmのサイズに裁断して試験片を得た。次に、30mm×50mm口径のメス型の成形金型とこれに対応したオス型の成形金型を用いて、得られた試験片に6.0mmの深さの凹部が形成されるように冷間成形した。次に、成形した積層体を温度65℃、湿度90%の環境下において、48時間静置し、目視にて積層体の層間の剥がれを評価した。積層体の層間に剥がれが無かったものを耐湿熱性が良好(○)、剥がれがあったものを耐湿熱性が悪い(×)と評価した。結果を表1に示す。
2…接着層
3…金属層
4…シーラント層
5…接着層
Claims (8)
- (1)ウレタン(メタ)アクリレートと、
(2)リン酸基を有する(メタ)アクリレート、(メタ)アクリル系シランカップリング剤、及びイソシアネート基を有する(メタ)アクリレートからなる群から選択された少なくとも1種と、
(3)エポキシ基を有する(メタ)アクリレートと、
を含む樹脂組成物。 - 前記樹脂組成物中において、前記(1)ウレタン(メタ)アクリレートと、前記(2)リン酸基を有する(メタ)アクリレート、(メタ)アクリル系シランカップリング剤、及びイソシアネート基を有する(メタ)アクリレートからなる群から選択された少なくとも1種並びに(3)エポキシ基を有する(メタ)アクリレートの合計との質量比((1):(2)+(3))が、99.8:0.2~80:20の範囲にある、請求項1に記載の樹脂組成物。
- 加熱または電離放射線照射により硬化する、請求項1または2に記載の樹脂組成物。
- 請求項1~3のいずれかに記載の樹脂組成物を硬化してなる、硬化樹脂組成物。
- 少なくとも、基材層と、接着層と、金属層と、シーラント層とがこの順に積層された積層体からなり、
前記接着層は、請求項1~3のいずれかに記載の樹脂組成物の硬化物により形成されてなる、電池用包装材料。 - 少なくとも、基材層と、接着層と、金属層と、シーラント層とをこの順に積層して積層体を得る工程を備える電池用包装材料の製造方法であって、
前記工程において、(1)ウレタン(メタ)アクリレートと、(2)リン酸基を有する(メタ)アクリレート、(メタ)アクリル系シランカップリング剤、及びイソシアネート基を有する(メタ)アクリレートからなる群から選択された少なくとも1種と、(3)エポキシ基を有する(メタ)アクリレートとを含む樹脂組成物を硬化させることにより前記接着層を形成する、電池用包装材料の製造方法。 - 少なくとも、基材層と、接着層と、金属層と、シーラント層とがこの順に積層された積層体からなる電池用包装材料の前記接着層への、請求項1~3のいずれかに記載の樹脂組成物の硬化物の使用。
- 少なくとも、基材層と、接着層と、金属層と、シーラント層とがこの順に積層された積層体からなり、前記接着層が、請求項1~3のいずれかに記載の樹脂組成物の硬化物により形成されてなる包装材料の、電池の製造のための使用。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2014556849A JP5704289B1 (ja) | 2013-06-24 | 2014-06-24 | 樹脂組成物 |
| KR1020157033182A KR20160022815A (ko) | 2013-06-24 | 2014-06-24 | 수지 조성물 |
| CN201480028631.2A CN105229106B (zh) | 2013-06-24 | 2014-06-24 | 树脂组合物 |
| EP14817680.3A EP3015523B1 (en) | 2013-06-24 | 2014-06-24 | Resin composition |
| US14/899,332 US10005937B2 (en) | 2013-06-24 | 2014-06-24 | Resin composition |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013-131369 | 2013-06-24 | ||
| JP2013131369 | 2013-06-24 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014208518A1 true WO2014208518A1 (ja) | 2014-12-31 |
Family
ID=52141852
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2014/066616 Ceased WO2014208518A1 (ja) | 2013-06-24 | 2014-06-24 | 樹脂組成物 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US10005937B2 (ja) |
| EP (1) | EP3015523B1 (ja) |
| JP (1) | JP5704289B1 (ja) |
| KR (1) | KR20160022815A (ja) |
| CN (1) | CN105229106B (ja) |
| WO (1) | WO2014208518A1 (ja) |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2017228478A (ja) * | 2016-06-24 | 2017-12-28 | 昭和電工パッケージング株式会社 | 蓄電デバイス用外装材及び蓄電デバイス |
| JP2018055976A (ja) * | 2016-09-29 | 2018-04-05 | 昭和電工パッケージング株式会社 | 包装材及びその製造方法 |
| WO2018066196A1 (ja) * | 2016-10-03 | 2018-04-12 | 昭和電工パッケージング株式会社 | 包装材の製造方法 |
| JP2018062101A (ja) * | 2016-10-12 | 2018-04-19 | 昭和電工パッケージング株式会社 | 包装材の製造方法 |
| CN108137982A (zh) * | 2015-10-13 | 2018-06-08 | 横滨橡胶株式会社 | 密封材料用水系底漆组合物 |
| WO2019097887A1 (ja) * | 2017-11-16 | 2019-05-23 | 昭和電工株式会社 | 接着剤、積層体、電池外装用包装材、電池ケース、及び電池ケースの製造方法 |
| JPWO2020203213A1 (ja) * | 2019-03-29 | 2020-10-08 | ||
| JP2020168861A (ja) * | 2016-10-03 | 2020-10-15 | 昭和電工パッケージング株式会社 | 包装材の製造方法 |
| JP2023045785A (ja) * | 2021-09-22 | 2023-04-03 | 日本ユピカ株式会社 | 金属密着性に優れる電気電子部品用結晶性ラジカル重合性組成物、当該組成物を使用した電気電子部品、及び当該電気電子部品の製造方法 |
| JP2023093511A (ja) * | 2022-02-14 | 2023-07-04 | 株式会社レゾナック・パッケージング | 蓄電デバイス用外装材 |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR102270874B1 (ko) | 2018-03-09 | 2021-07-01 | 주식회사 엘지에너지솔루션 | 리튬 이차 전지용 파우치 외장재 및 이를 포함하는 리튬 이차 전지 |
| KR102318161B1 (ko) * | 2021-04-01 | 2021-10-26 | 동우 화인켐 주식회사 | 접착제 조성물 및 전지용 포장재료 |
| CN113462345A (zh) * | 2021-07-13 | 2021-10-01 | 天能电池集团股份有限公司 | 一种紫外光固化色胶及其制备方法 |
| KR20230124118A (ko) * | 2022-02-17 | 2023-08-25 | 동우 화인켐 주식회사 | 접착제 조성물 및 전지용 포장재 |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS58204060A (ja) * | 1982-05-25 | 1983-11-28 | Asahi Chem Ind Co Ltd | 感光性コ−チング材 |
| JPH0657232A (ja) | 1992-08-05 | 1994-03-01 | Dainippon Printing Co Ltd | 接着剤組成物 |
| JPH1036809A (ja) | 1996-07-22 | 1998-02-10 | Toyo Mooton Kk | 電離放射線硬化型接着剤組成物 |
| JPH1046109A (ja) * | 1996-08-07 | 1998-02-17 | Sumitomo Chem Co Ltd | ディジタルビデオディスクの保護コート兼接着用樹脂組成物 |
| JPH10287718A (ja) * | 1997-02-13 | 1998-10-27 | Jsr Corp | 光硬化型樹脂組成物 |
| JP2002114949A (ja) * | 2000-10-05 | 2002-04-16 | Nippon Kayaku Co Ltd | 光ディスク用接着剤組成物、硬化物および物品 |
| JP2006104446A (ja) * | 2004-08-19 | 2006-04-20 | Nippon Kayaku Co Ltd | 光ディスク用接着剤組成物、硬化物および物品 |
| JP2009114352A (ja) * | 2007-11-07 | 2009-05-28 | Bridgestone Corp | 接着性樹脂組成物及びそれを用いた接着材料 |
| JP2012506929A (ja) * | 2008-10-31 | 2012-03-22 | ビーエーエスエフ ソシエタス・ヨーロピア | 接着促進剤としての(メタ)アクリルホスホン酸エステル |
Family Cites Families (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2733180B2 (ja) * | 1993-02-09 | 1998-03-30 | 東洋製罐株式会社 | フレキシブル包装用ラミネート、その製造方法及びそれに用いる接着剤 |
| US6440519B1 (en) | 1997-02-13 | 2002-08-27 | Dsm N.V. | Photocurable adhesive for optical disk |
| JP3734707B2 (ja) * | 1998-05-28 | 2006-01-11 | 大日本インキ化学工業株式会社 | 紫外線硬化型組成物、光ディスク及び光ディスクの製造方法 |
| US7052756B2 (en) * | 2001-05-17 | 2006-05-30 | Nippon Kayaku Kabushiki Kaisha | Adhesive for optical disk and optical disk |
| DE60237501D1 (de) | 2001-06-20 | 2010-10-14 | Dainippon Printing Co Ltd | Batterieverpackungsmaterial |
| US20030211299A1 (en) * | 2001-09-27 | 2003-11-13 | 3M Innovative Properties Company | Adhesion-enhancing surfaces for marking materials |
| EP1616710A1 (de) * | 2004-07-01 | 2006-01-18 | Alcan Technology & Management Ltd. | Verfahren zur Herstellung eines Verpackungsmaterials |
| ATE357333T1 (de) * | 2004-07-01 | 2007-04-15 | Alcan Tech & Man Ltd | Verfahren zur herstellung eines verpackungsmaterials |
| US20080152857A1 (en) | 2004-08-19 | 2008-06-26 | Nippon Kayaku Kabushiki Kaisha | Adhesive Compositon for Optical Disk, Cured Product and Article |
| DE102005057245A1 (de) * | 2005-11-29 | 2007-06-06 | Bayer Materialscience Ag | Lackierte Folien |
| KR100929593B1 (ko) * | 2007-09-20 | 2009-12-03 | 제일모직주식회사 | 이방 도전성 접착 조성물 및 그를 포함하는 이방 도전성 필름 |
| WO2009139318A1 (ja) * | 2008-05-13 | 2009-11-19 | 三菱瓦斯化学株式会社 | 曲げ加工可能なポリカーボネート樹脂積層体および光透過型電磁波シールド積層体並びにそれらの製造方法 |
| EP3101709B1 (en) * | 2009-08-07 | 2019-01-02 | Dai Nippon Printing Co., Ltd. | Packaging material for electrochemical cells |
| CN101781486B (zh) | 2010-03-11 | 2012-03-21 | 苏州市明大高分子科技材料有限公司 | 紫外光固化阻燃涂料及其制备方法 |
| US20110300367A1 (en) | 2010-06-07 | 2011-12-08 | Ching-Kee Chien | Optical Fiber With Photoacid Coating |
| US20150166751A1 (en) * | 2012-07-06 | 2015-06-18 | Mitsui Chemicals, Inc. | Laminate |
| JP5267718B1 (ja) * | 2012-11-01 | 2013-08-21 | 東洋インキScホールディングス株式会社 | 電池用包装材用ポリウレタン接着剤、電池用包装材、電池用容器および電池 |
| JP5578269B1 (ja) * | 2013-02-25 | 2014-08-27 | 東洋インキScホールディングス株式会社 | 電池用包装材用ポリウレタン接着剤、電池用包装材、電池用容器および電池 |
-
2014
- 2014-06-24 CN CN201480028631.2A patent/CN105229106B/zh active Active
- 2014-06-24 EP EP14817680.3A patent/EP3015523B1/en active Active
- 2014-06-24 WO PCT/JP2014/066616 patent/WO2014208518A1/ja not_active Ceased
- 2014-06-24 KR KR1020157033182A patent/KR20160022815A/ko not_active Withdrawn
- 2014-06-24 US US14/899,332 patent/US10005937B2/en active Active
- 2014-06-24 JP JP2014556849A patent/JP5704289B1/ja active Active
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS58204060A (ja) * | 1982-05-25 | 1983-11-28 | Asahi Chem Ind Co Ltd | 感光性コ−チング材 |
| JPH0657232A (ja) | 1992-08-05 | 1994-03-01 | Dainippon Printing Co Ltd | 接着剤組成物 |
| JPH1036809A (ja) | 1996-07-22 | 1998-02-10 | Toyo Mooton Kk | 電離放射線硬化型接着剤組成物 |
| JPH1046109A (ja) * | 1996-08-07 | 1998-02-17 | Sumitomo Chem Co Ltd | ディジタルビデオディスクの保護コート兼接着用樹脂組成物 |
| JPH10287718A (ja) * | 1997-02-13 | 1998-10-27 | Jsr Corp | 光硬化型樹脂組成物 |
| JP2002114949A (ja) * | 2000-10-05 | 2002-04-16 | Nippon Kayaku Co Ltd | 光ディスク用接着剤組成物、硬化物および物品 |
| JP2006104446A (ja) * | 2004-08-19 | 2006-04-20 | Nippon Kayaku Co Ltd | 光ディスク用接着剤組成物、硬化物および物品 |
| JP2009114352A (ja) * | 2007-11-07 | 2009-05-28 | Bridgestone Corp | 接着性樹脂組成物及びそれを用いた接着材料 |
| JP2012506929A (ja) * | 2008-10-31 | 2012-03-22 | ビーエーエスエフ ソシエタス・ヨーロピア | 接着促進剤としての(メタ)アクリルホスホン酸エステル |
Cited By (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN108137982A (zh) * | 2015-10-13 | 2018-06-08 | 横滨橡胶株式会社 | 密封材料用水系底漆组合物 |
| CN108137982B (zh) * | 2015-10-13 | 2020-07-14 | 横滨橡胶株式会社 | 密封材料用水系底漆组合物 |
| WO2017221553A1 (ja) * | 2016-06-24 | 2017-12-28 | 昭和電工パッケージング株式会社 | 蓄電デバイス用外装材及び蓄電デバイス |
| US11731398B2 (en) | 2016-06-24 | 2023-08-22 | Showa Denko Packaing Co., Ltd. | Exterior material for power storage device and power storage device |
| JP2017228478A (ja) * | 2016-06-24 | 2017-12-28 | 昭和電工パッケージング株式会社 | 蓄電デバイス用外装材及び蓄電デバイス |
| JP2018055976A (ja) * | 2016-09-29 | 2018-04-05 | 昭和電工パッケージング株式会社 | 包装材及びその製造方法 |
| WO2018061375A1 (ja) * | 2016-09-29 | 2018-04-05 | 昭和電工パッケージング株式会社 | 包装材及びその製造方法 |
| JP2018058230A (ja) * | 2016-10-03 | 2018-04-12 | 昭和電工パッケージング株式会社 | 包装材の製造方法 |
| JP2020168861A (ja) * | 2016-10-03 | 2020-10-15 | 昭和電工パッケージング株式会社 | 包装材の製造方法 |
| JP7303160B2 (ja) | 2016-10-03 | 2023-07-04 | 株式会社レゾナック・パッケージング | 蓄電デバイス用外装材の製造方法 |
| WO2018066196A1 (ja) * | 2016-10-03 | 2018-04-12 | 昭和電工パッケージング株式会社 | 包装材の製造方法 |
| JP2018062101A (ja) * | 2016-10-12 | 2018-04-19 | 昭和電工パッケージング株式会社 | 包装材の製造方法 |
| WO2019097887A1 (ja) * | 2017-11-16 | 2019-05-23 | 昭和電工株式会社 | 接着剤、積層体、電池外装用包装材、電池ケース、及び電池ケースの製造方法 |
| JP6526368B1 (ja) * | 2017-11-16 | 2019-06-05 | 昭和電工株式会社 | 接着剤、積層体、電池外装用包装材、電池ケース、及び電池ケースの製造方法 |
| US10766229B2 (en) | 2017-11-16 | 2020-09-08 | Showa Denko K.K. | Adhesive, laminate, packaging material for battery casing, battery case, and method of producing battery case |
| JPWO2020203213A1 (ja) * | 2019-03-29 | 2020-10-08 | ||
| JP7428178B2 (ja) | 2019-03-29 | 2024-02-06 | 株式会社レゾナック | 接着剤用組成物、蓄電装置用外装材及びその製造方法 |
| JP2023045785A (ja) * | 2021-09-22 | 2023-04-03 | 日本ユピカ株式会社 | 金属密着性に優れる電気電子部品用結晶性ラジカル重合性組成物、当該組成物を使用した電気電子部品、及び当該電気電子部品の製造方法 |
| JP7836650B2 (ja) | 2021-09-22 | 2026-03-27 | 三菱ガス化学ネクスト株式会社 | 金属密着性に優れる電気電子部品用結晶性ラジカル重合性組成物、当該組成物を使用した電気電子部品、及び当該電気電子部品の製造方法 |
| JP2023093511A (ja) * | 2022-02-14 | 2023-07-04 | 株式会社レゾナック・パッケージング | 蓄電デバイス用外装材 |
Also Published As
| Publication number | Publication date |
|---|---|
| US10005937B2 (en) | 2018-06-26 |
| EP3015523A1 (en) | 2016-05-04 |
| JP5704289B1 (ja) | 2015-04-22 |
| CN105229106B (zh) | 2018-07-10 |
| EP3015523A4 (en) | 2016-11-16 |
| US20160145477A1 (en) | 2016-05-26 |
| CN105229106A (zh) | 2016-01-06 |
| KR20160022815A (ko) | 2016-03-02 |
| JPWO2014208518A1 (ja) | 2017-02-23 |
| EP3015523B1 (en) | 2018-11-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5704289B1 (ja) | 樹脂組成物 | |
| KR102230063B1 (ko) | 전지용 포장 재료 | |
| KR102220457B1 (ko) | 전지용 포장 재료 | |
| JP2020107608A (ja) | 電池用包装材料 | |
| JP6566133B2 (ja) | 電池用包装材料、その製造方法、電池、及びポリエステルフィルム | |
| JP2020098794A (ja) | 電池用包装材料 | |
| JP6686587B2 (ja) | 電池用包装材料、その製造方法及び電池 | |
| JP6679918B2 (ja) | 電池用包装材料 | |
| JP7314970B2 (ja) | 電池用包装材料、その製造方法及び電池 | |
| WO2017209218A1 (ja) | 電池用包装材料、その製造方法、電池、及びポリエステルフィルム | |
| JP2017069203A (ja) | 電池用包装材料及び電池 | |
| JP7225832B2 (ja) | 蓄電デバイス用外装材、蓄電デバイス用外装材の製造方法、及び蓄電デバイス | |
| JP2016184546A (ja) | 金属端子用接着性フィルム | |
| JP2016171078A (ja) | 電池用包装材料 | |
| JP2017139121A (ja) | 金属端子用接着性フィルム | |
| WO2016159190A1 (ja) | 電池用包装材料、その製造方法及び電池 | |
| JP2019212433A (ja) | 電池用包装材料、その製造方法、電池用包装材料の巻取体、電池 | |
| WO2019078284A1 (ja) | 電池用包装材料及び電池 | |
| JP6736837B2 (ja) | 電池用包装材料 | |
| JP6003953B2 (ja) | 電池用包装材料 | |
| JP6686634B2 (ja) | 電池用包装材料、その製造方法及び電池 | |
| JP7234794B2 (ja) | 蓄電デバイス用外装材、その製造方法、蓄電デバイス、及びポリアミドフィルム | |
| JP6245328B2 (ja) | 電池用包装材料 | |
| JP7225833B2 (ja) | 蓄電デバイス用外装材、蓄電デバイス用外装材の製造方法、及び蓄電デバイス | |
| JP6672675B2 (ja) | 電池用包装材料 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201480028631.2 Country of ref document: CN |
|
| ENP | Entry into the national phase |
Ref document number: 2014556849 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14817680 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 20157033182 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14899332 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2014817680 Country of ref document: EP |