WO2017073473A1 - モリンガエキス - Google Patents
モリンガエキス Download PDFInfo
- Publication number
- WO2017073473A1 WO2017073473A1 PCT/JP2016/081237 JP2016081237W WO2017073473A1 WO 2017073473 A1 WO2017073473 A1 WO 2017073473A1 JP 2016081237 W JP2016081237 W JP 2016081237W WO 2017073473 A1 WO2017073473 A1 WO 2017073473A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- moringa
- extract
- moringa extract
- mass
- parts
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23F—COFFEE; TEA; THEIR SUBSTITUTES; MANUFACTURE, PREPARATION, OR INFUSION THEREOF
- A23F5/00—Coffee; Coffee substitutes; Preparations thereof
- A23F5/46—Coffee flavour; Coffee oil; Flavouring of coffee or coffee extract
- A23F5/465—Flavouring with flavours other than natural coffee flavour or coffee oil
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23G—COCOA; COCOA PRODUCTS, e.g. CHOCOLATE; SUBSTITUTES FOR COCOA OR COCOA PRODUCTS; CONFECTIONERY; CHEWING GUM; ICE-CREAM; PREPARATION THEREOF
- A23G1/00—Cocoa; Cocoa products, e.g. chocolate; Substitutes therefor
- A23G1/30—Cocoa products, e.g. chocolate; Substitutes therefor
- A23G1/32—Cocoa products, e.g. chocolate; Substitutes therefor characterised by the composition containing organic or inorganic compounds
- A23G1/48—Cocoa products, e.g. chocolate; Substitutes therefor characterised by the composition containing organic or inorganic compounds containing plants or parts thereof, e.g. fruits, seeds or extracts
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23G—COCOA; COCOA PRODUCTS, e.g. CHOCOLATE; SUBSTITUTES FOR COCOA OR COCOA PRODUCTS; CONFECTIONERY; CHEWING GUM; ICE-CREAM; PREPARATION THEREOF
- A23G3/00—Sweetmeats; Confectionery; Marzipan; Coated or filled products
- A23G3/34—Sweetmeats, confectionery or marzipan; Processes for the preparation thereof
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23G—COCOA; COCOA PRODUCTS, e.g. CHOCOLATE; SUBSTITUTES FOR COCOA OR COCOA PRODUCTS; CONFECTIONERY; CHEWING GUM; ICE-CREAM; PREPARATION THEREOF
- A23G3/00—Sweetmeats; Confectionery; Marzipan; Coated or filled products
- A23G3/34—Sweetmeats, confectionery or marzipan; Processes for the preparation thereof
- A23G3/36—Sweetmeats, confectionery or marzipan; Processes for the preparation thereof characterised by the composition containing organic or inorganic compounds
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23G—COCOA; COCOA PRODUCTS, e.g. CHOCOLATE; SUBSTITUTES FOR COCOA OR COCOA PRODUCTS; CONFECTIONERY; CHEWING GUM; ICE-CREAM; PREPARATION THEREOF
- A23G3/00—Sweetmeats; Confectionery; Marzipan; Coated or filled products
- A23G3/34—Sweetmeats, confectionery or marzipan; Processes for the preparation thereof
- A23G3/36—Sweetmeats, confectionery or marzipan; Processes for the preparation thereof characterised by the composition containing organic or inorganic compounds
- A23G3/48—Sweetmeats, confectionery or marzipan; Processes for the preparation thereof characterised by the composition containing organic or inorganic compounds containing plants or parts thereof, e.g. fruits, seeds, extracts
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23G—COCOA; COCOA PRODUCTS, e.g. CHOCOLATE; SUBSTITUTES FOR COCOA OR COCOA PRODUCTS; CONFECTIONERY; CHEWING GUM; ICE-CREAM; PREPARATION THEREOF
- A23G4/00—Chewing gum
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23G—COCOA; COCOA PRODUCTS, e.g. CHOCOLATE; SUBSTITUTES FOR COCOA OR COCOA PRODUCTS; CONFECTIONERY; CHEWING GUM; ICE-CREAM; PREPARATION THEREOF
- A23G4/00—Chewing gum
- A23G4/06—Chewing gum characterised by the composition containing organic or inorganic compounds
- A23G4/068—Chewing gum characterised by the composition containing organic or inorganic compounds containing plants or parts thereof, e.g. fruits, seeds, extracts
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23G—COCOA; COCOA PRODUCTS, e.g. CHOCOLATE; SUBSTITUTES FOR COCOA OR COCOA PRODUCTS; CONFECTIONERY; CHEWING GUM; ICE-CREAM; PREPARATION THEREOF
- A23G9/00—Frozen sweets, e.g. ice confectionery, ice-cream; Mixtures therefor
- A23G9/32—Frozen sweets, e.g. ice confectionery, ice-cream; Mixtures therefor characterised by the composition containing organic or inorganic compounds
- A23G9/42—Frozen sweets, e.g. ice confectionery, ice-cream; Mixtures therefor characterised by the composition containing organic or inorganic compounds containing plants or parts thereof, e.g. fruits, seeds, extracts
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L2/00—Non-alcoholic beverages; Dry compositions or concentrates therefor; Preparation or treatment thereof
- A23L2/52—Adding ingredients
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L2/00—Non-alcoholic beverages; Dry compositions or concentrates therefor; Preparation or treatment thereof
- A23L2/52—Adding ingredients
- A23L2/54—Mixing with gases
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/105—Plant extracts, their artificial duplicates or their derivatives
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L9/00—Puddings; Cream substitutes; Preparation or treatment thereof
- A23L9/10—Puddings; Dry powder puddings
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L9/00—Puddings; Cream substitutes; Preparation or treatment thereof
- A23L9/10—Puddings; Dry powder puddings
- A23L9/12—Ready-to-eat liquid or semi-liquid desserts, e.g. puddings, not to be mixed with liquids, e.g. water, milk
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7028—Compounds having saccharide radicals attached to non-saccharide compounds by glycosidic linkages
- A61K31/7034—Compounds having saccharide radicals attached to non-saccharide compounds by glycosidic linkages attached to a carbocyclic compound, e.g. phloridzin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/02—Nutrients, e.g. vitamins, minerals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/06—Antihyperlipidemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/12—Antihypertensives
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L2/00—Non-alcoholic beverages; Dry compositions or concentrates therefor; Preparation or treatment thereof
- A23L2/385—Concentrates of non-alcoholic beverages
- A23L2/39—Dry compositions
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2002/00—Food compositions, function of food ingredients or processes for food or foodstuffs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2236/00—Isolation or extraction methods of medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicine
- A61K2236/30—Extraction of the material
- A61K2236/33—Extraction of the material involving extraction with hydrophilic solvents, e.g. lower alcohols, esters or ketones
- A61K2236/331—Extraction of the material involving extraction with hydrophilic solvents, e.g. lower alcohols, esters or ketones using water, e.g. cold water, infusion, tea, steam distillation or decoction
Definitions
- the present invention relates to a moringa extract, a PPAR activator containing the moringa extract, a composition containing the moringa extract, a production method thereof, and a food and drink containing these.
- Moringa Plants belonging to the genus Wasabi (generally referred to as “moringa” in the present specification) are plants that are widely used as medicinal plants in India and Southeast Asia, and have various useful physiological functions such as antioxidant and anti-inflammatory effects. Has been found. Moringa contains many minerals, amino acids, benzylglucosinolates (BGLs) and the like as active ingredients for these effects. Therefore, in recent years, dry pulverized products of moringa leaves or roots, and extract powders extracted from these with hot water or hydrous alcohol, etc., have begun to be sold as raw materials for functional foods and are attracting attention. (See Patent Documents 1 and 2 and Non-Patent Document 1)
- PPAR peroxisome proliferator activated receptor
- This PPAR has three subtypes of ⁇ , ⁇ / ⁇ , and ⁇ , and is involved in lipid metabolism, sugar metabolism, cell proliferation and differentiation.
- ⁇ is mainly expressed in the liver to burn fat
- ⁇ / ⁇ is expressed in various tissues including muscles to improve exercise ability
- ⁇ is expressed in white adipose tissue and macrophages. Their involvement in differentiation induction and fat synthesis has been reported. Therefore, PPAR activation is expected to lead to the prevention and amelioration effects of various diseases such as insulin resistance, diabetes, obesity, and hypertension, and development of activators is expected (Non-patent Documents 2 and 3).
- PPAR activators made by chemical synthesis such as ibuprofen, leukotriene B4, indomethacin, fibrate compounds and the like have been developed and known as substances that activate PPAR.
- PPAR activator derived from a natural component curcumin contained in turmeric, monoacylglycerol which is a kind of fats and oils, catechins contained in tea and the like have been reported (see Patent Document 3).
- Japanese Patent No. 4032393 JP 2008-237117 A Japanese Patent No. 4719372 Japanese Patent Application Laid-Open No. 7-304685 Special table 2012-530769 gazette
- moringa extract which is an extract of moringa
- the content of the moringa extract is reduced due to a part of the active ingredient being decomposed during the extraction process, and the useful physiological function may not be exhibited.
- benzyl glucosinolate is easily decomposed during extract extraction, and further improvement is required.
- Moringa is no exception.
- the moringa extract obtained by extracting from the moringa also contains alkaloids. It seems safe if ordinary people take the normal amount orally, but if pregnant women take it, there is a possibility of uterine contraction or miscarriage. Therefore, further improvement is required for the safety of Moringa extract.
- the first object of the present invention is to provide a moringa extract having useful physiological functions and high safety.
- the second object of the present invention is to provide a natural component-derived PPAR activator that is excellent in PPAR activation.
- benzyl glucosinolate is difficult to prepare an extract that is stabilized in quality, such as being easily decomposed when manufactured from a plant extract.
- Benzylglucosinolate has been confirmed to be contained not only in Brassicaceae but also in Moringa, which is a member of the Brassicaceae family. It is difficult to provide a stable quality.
- the third object of the present invention is to provide a benzylglucosinolate-containing composition that suppresses the decomposition of benzylglucosinolate and is excellent in stability.
- the present invention for solving the first problem is: [1] Moringa extract, wherein the content of benzylglucosinolate is 6% by mass or more in terms of the dry solid content of the extract, and substantially free of alkaloids, [2] A method for producing a moringa extract, comprising a step of extracting moringa pretreated at 80 ° C. or higher in a solvent at 10 to 50 ° C. and, [3] The present invention relates to a food or drink containing the moringa extract according to [1].
- the present invention for solving the second problem is: [1] A peroxisome proliferator-activated receptor (PPAR) activator comprising Moringa extract, [2] A food or drink comprising the PPAR activator according to [1], and [3]
- the present invention relates to a method for producing a PPAR activator containing a moringa extract, comprising a step of extracting moringa pretreated at 80 ° C. or higher in a solvent at 10 to 50 ° C.
- the present invention for solving the third problem is: [1] A benzylglucosinolate-containing composition containing a moringa extract and an excipient, wherein the content of the excipient is 65 parts by mass or more with respect to 100 parts by mass of the dry solid content of the moringa extract A composition, [2] A food or drink comprising the composition according to [1], and, [3] A method for producing a benzylglucosinolate-containing composition comprising a step of mixing a moringa extract and an excipient, wherein the amount of the excipient is 100 parts by mass of the dry solid content of the moringa extract The production method is 65 to 1000 parts by mass based on the mass.
- a moringa extract having a useful physiological function and high safety can be provided.
- benzylglucosinolate-containing composition that suppresses the decomposition of benzylglucosinolate and is excellent in stability.
- the moringa extract of this embodiment contains benzyl glucosinolate.
- Benzylglucosinolate is known to have useful physiological functions such as anti-fatigue, antioxidant, nutritional tonic, and hormone regulation. These functions can be exhibited by containing a predetermined amount or more of benzyl glucosinolate in the moringa extract of this embodiment.
- Fatigue is caused by pure exercise and social activities, environmental factors such as temperature and humidity, and appears as symptom of slow movements, decreased amount of action, distraction of attention, decreased stimulus responsiveness, etc. There is skin fatigue that appears as skin sagging and wrinkles.
- the fatigue in anti-fatigue referred to in this embodiment includes any fatigue, and is preferably anti-fatigue against the former fatigue.
- the DPPH radical scavenging activity of the moringa extract of this embodiment has an IC50 value of preferably 600 ⁇ g / ml or less, more preferably 300 ⁇ g / ml or less when measured by the method described in the Examples section below. Preferably, it is 100 ⁇ g / ml or less.
- the content of benzylglucosinolate in the moringa extract is 6% by mass or more, preferably 10% by mass or more, and more preferably 15% by mass or more in terms of dry extract solid content, from the viewpoint of exhibiting useful physiological functions.
- the upper limit is not particularly limited, but can be, for example, 50% by mass or less.
- the content of benzylglucosinolate in the moringa extract refers to the amount analyzed by the method of Zhang et al. (Reference: Zhang, Y., Wade, KL, Prestera, T., and and Talalay, P. Anal. Biochem. 239: 160-167, 1996). That is, a reaction product (1,3-benzenedithiol-2-thione) generated when moringa extract is treated with myrosinase and the treated product is reacted with 1,2-benzenedithiol in a weak alkaline buffer solution. This is a method of analyzing and quantifying by reverse phase high performance liquid chromatography (detection: UV 365 nm).
- the moringa extract of this embodiment is substantially free of alkaloids, and preferably does not contain any alkaloids.
- substantially free of alkaloid means that a dry powder of Moringa extract is used as a sample, 100 mg of sample is dissolved in 100 mL of methanol, and 100 ⁇ L is dropped on a thin layer plate (the amount of sample dropped at this time) Is approximately 100 micrograms), it is determined that the substance is substantially not contained when it develops with a developing solvent and then does not develop color when sprayed with the Dragendorf reagent.
- the detection limit of alkaloids is 1 microgram, and the detection limit in the sample is about 1%.
- alkaloids to be detected include spirotine.
- the moringa extract of this embodiment can contain a free amino acid, and can further contain, for example, one or more amino acids selected from the group consisting of arginine, glutamic acid, alanine, methionine, and cysteine.
- the content of free amino acid in the moringa extract is preferably 0.1% by mass or more, more preferably 0.5% by mass or more in terms of dry solid content of the extract from the viewpoint of health promotion, and the upper limit is particularly limited. Although it is not a thing, it can be 2.0 mass% or less, for example.
- the content when two or more free amino acids are included refers to the total amount.
- the moringa extract of this embodiment is obtained by extraction from moringa using a solvent.
- Moringa used for extraction is not particularly limited, and examples thereof include Moringa oleifera, Moringa concanensis, Moringa drohardi and the like. Of these, Moringa oleifera is preferred from the viewpoint of widespread cultivation and easy collection. Moringa oleifera is a small deciduous tree that is native to India and has other names such as horseradish tree, Bennut, Malungai (Tagalog), and Sanjanaa (Hindu).
- any of leaves, stems, sheaths (fruits), and seeds can be used. These parts may be used as they are, or may be used after drying, but are preferably used after drying from the viewpoint of storage stability as a raw material and yield in producing the extract.
- pulp or seeds it is preferable to use pulp or seeds, and more preferable to use seeds, from the viewpoint of relatively few sites of alkaloids.
- seed it may be used after removing the seed coat, or may be used after pulverized into powder. From the viewpoint of easily producing an extract, it is preferable to use a powdery material obtained by pulverizing seeds with a skin.
- a moringa extract that is substantially free of alkaloids can be obtained even if it is a leaf or stem part having a large amount of alkaloids, according to the production method of the embodiment described below. Also from this point, the manufacturing method of the aspect mentioned later has technical significance.
- the dried moringa powder or the like is performed at 80 ° C. or higher, preferably 80 to 170 ° C., for example, preferably 80 to 95 ° C., more preferably 85 to 90 ° C.
- the treatment include treatment in a liquid for 1 to 10 minutes, more preferably 3 to 5 minutes.
- pretreatment for a relatively short time is effective in this manner, and when the treatment is performed for more than 10 minutes, benzylglucosinolate is decomposed due to poor thermal stability.
- pretreatment within 10 minutes is preferable from the viewpoint of suppressing alkaloid extraction.
- the pretreatment of this embodiment is not particularly limited, and examples include boiled water, oil-like, roasting, and when using a medium, water, ethanol, animal and vegetable oils or mixtures thereof, and the like are safe. From the viewpoint of water, water is preferable.
- Such pretreatment can denature proteins that are considered to be benzyl glucosinolate degradation promoting components, deactivate the degradation accelerating activity, and increase the content of benzyl glucosinolate in Moringa extract It is estimated to be.
- hot water extraction was conventionally performed, extraction for a long time exceeding 10 minutes is required for sufficient extraction.
- problems such as decomposition of benzylglucosinolate by heat and extraction of a large amount of alkaloids.
- the pretreatment of this embodiment the low-temperature extraction described later is performed after the pretreatment within 10 minutes, so that the activity of the proteins can be deactivated without causing such inconvenience. Presumed.
- proteins that are considered to be benzylglucosinolate degradation promoting components include myrosinase.
- the pretreatment is not limited to this embodiment, and a known treatment embodiment for inactivating the activity of these proteins can be employed.
- a solvent used for extraction water or an organic solvent containing water is used.
- the organic solvent include lower alcohols (monohydric or polyhydric alcohols having 1 to 4 carbon atoms such as methanol, ethanol, propanol, propylene glycol, butylene glycol, and glycerin) that can be mixed with water, acetone, and the like. It is done. These organic solvents may be used after being mixed with water in advance, or two or more kinds may be used by mixing with water. From the viewpoint of safety, it is preferable to extract only with water.
- the amount of the solvent used for the extraction is not particularly limited, and can be, for example, 200 to 3000 parts by mass with respect to 100 parts by mass of Moringa used for extraction.
- the temperature of the solvent at the time of extraction is 10 to 50 ° C., preferably 20 to 40 ° C. from the viewpoint of reducing production efficiency and alkaloid content in the moringa extract.
- the resulting moringa extract contains a considerable amount of alkaloid, but surprisingly, by extracting at a lower temperature, the alkaloid content is less. I found out that Although the mechanism by which the content of alkaloid is reduced is not clear, it is presumed that the extraction of alkaloid is suppressed under low temperature conditions.
- the extraction time is not particularly limited, but can be, for example, 30 to 150 minutes from the viewpoint of production efficiency.
- Extraction can be performed with stirring or stationary. Further, when the pretreatment is performed using the solvent used for extraction, the pretreatment and extraction can be performed continuously. For example, after the moringa is treated at 80 ° C. or higher for 1 to 10 minutes, the temperature is adjusted to 10 to 50 ° C. and the extract is extracted.
- the extract After extraction, treatment such as filtration or centrifugation is performed to remove the residue, and then the extraction solvent can be removed by decompression or the like.
- the extract when used as a powder, the extract can be dried with a spray dryer or the like as necessary.
- moringa extract of this embodiment thus obtained contains a large amount of benzyl glucosinolate, it can exhibit useful physiological functions. Moreover, since the moringa extract of this aspect does not contain an alkaloid substantially, even if a pregnant woman ingests, there is no problem and it is a highly safe thing.
- the moringa extract of this embodiment may be either liquid or solid, but is preferably a solid such as powder from the viewpoint of easy transportation.
- moringa extract-containing composition containing a moringa extract and an excipient is preferable from the viewpoint of preventing solidification of the moringa extract and from the viewpoint of stabilizing the quality by suppressing the decomposition of benzylglucosinolate.
- a method for stabilizing the moringa extract a method of blending an excipient with the moringa extract is preferable.
- excipient examples include lactose, starch, cyclodextrin, galactomannan, dextrin, maltodextrin, etc., and from the viewpoints of actual use and allergenicity, preferably cyclodextrin, dextrin, or maltodextrin, more preferably Is dextrin or maltodextrin.
- the content of the excipient in the moringa extract-containing composition of the present embodiment is not particularly limited, but is preferably 65 from the viewpoint of stability with respect to 100 parts by mass of the dry solid content of the moringa extract. It is at least 100 parts by mass, more preferably at least 100 parts by mass, and from the viewpoint of reducing the amount of excipients used and reducing costs, it is preferably at most 1000 parts by mass, more preferably at most 500 parts by mass.
- the addition timing of the excipient is not particularly limited, but it may be after extraction, after removal of insoluble residue during extract production, before powdering, etc. From the viewpoint of improving filterability, removal of insoluble residue Addition after or before pulverization is preferred, and addition after removal of the insoluble residue is more preferred.
- the moringa extract of this aspect can be blended in various foods and drinks.
- beverages such as soft drinks, carbonated drinks, nutrition drinks, fruit drinks, and lactic acid drinks, concentrated concentrates of these drinks, and powders for adjustment may be used.
- this moringa extract can be added to frozen confectionery such as ice cream, sorbet, shaved ice, and noodles such as buckwheat, udon, harusame, gyoza peel, cucumber peel, Chinese noodles, and instant noodles.
- this moringa extract can also be added to confectionery such as candy, candy, gum, chocolate, tablet candy, gummy candy, snack confectionery, biscuits, jelly, pudding, jam, cream, baked confectionery and the like.
- Moringa extract is added to fishery and livestock processed foods such as kamaboko, ham and sausage, dairy products such as processed milk and fermented milk, salad oil, tempura oil, margarine, mayonnaise, shortening, whipped cream and dressing, etc. It can also be added to seasonings such as oils and processed foods, sauces and sauces, soups, stews, salads, prepared dishes and pickles.
- seasonings such as oils and processed foods, sauces and sauces, soups, stews, salads, prepared dishes and pickles.
- various forms of health and nutritional supplements such as tablets, capsules and drinks, and other oral medicines such as mouth fresheners, dentifrices, mouthwashes, etc. It can be used by adding to external products, emollient cream, emollient lotion and the like.
- the blending amount of the moringa extract of this embodiment is not particularly limited, but it can be blended in food and drink so as to be 0.01 to 80% by mass in terms of dry extract solid content, for example.
- the content of benzyl glucosinolate in the food and drink of this embodiment can be 0.0006 to 40.0% by mass.
- the food / beverage products of this aspect are a thing which does not contain the alkaloid derived from the moringa extract mix
- the food and drink containing the moringa extract of this aspect contains many benzyl glucosinolates, it can exhibit a useful physiological function. Moreover, since the food / beverage products containing the moringa extract of this aspect do not contain the algaloid derived from the moringa extract mix
- Moringa has been eaten for many years mainly in India and Southeast Asia, and its safety has been confirmed, and even if added to food, it does not impair the unique flavor of the food. Can be taken on a daily basis for a long time.
- the PPAR activator of this embodiment includes a moringa extract.
- a moringa extract a commercially available moringa extract may be used, or it may be extracted from any part of the moringa using a solvent.
- an extract extracted from Moringa seeds also referred to as “seed extract”
- Examples of a method for extracting an extract from moringa seeds include the following embodiments.
- Moringa used for extraction is not particularly limited, and examples thereof include Moringa oleifera, Moringa concanensis, Moringa drohardi and the like. Of these, Moringa oleifera is preferred from the viewpoint of widespread cultivation and easy collection. Moringa oleifera is a small deciduous tree that is native to India and has other names such as horseradish tree, Bennut, Malungai (Tagalog), and Sanjanaa (Hindu).
- Moringa seeds used for extraction may be used raw or after drying, but they should be used after drying from the viewpoint of storage stability as raw materials and yield during extract production. Is preferred. It may be used after removing the hull of the seed, or may be used after being pulverized into powder, but from the viewpoint of easily producing the extract, the powder obtained by crushing the seed with the hull attached It is preferable to use the body.
- Moringa seeds subjected to extraction can be optionally pretreated before extraction in order to increase the content of functional components such as benzylglucosinolate in the seed extract.
- the use of a seed extract containing a large amount of functional components such as benzylglucosinolate as a PPAR activator is preferable from the viewpoint of imparting various useful physiological functions such as antioxidant and anti-inflammatory effects to the PPAR activator.
- This pretreatment is a treatment for denaturing or deactivating components that promote decomposition of benzylglucosinolate or the like during or after extraction of the extract.
- the dried moringa seed powder or the like is used as an aspect of this pretreatment.
- the dried moringa powder or the like is heated to 80 ° C. or higher, preferably 80 to 170 ° C.
- a method of treating in a liquid preferably at 80 to 95 ° C., more preferably at 85 to 90 ° C., preferably for 1 to 10 minutes, more preferably for 3 to 5 minutes.
- the pretreatment of this embodiment is not particularly limited, but includes boiled water, oil-like, roasting, etc., and when using a medium, water, ethanol, animal and vegetable oils or mixtures thereof, and the like, Water is preferable from the viewpoint of safety.
- the solvent used for extraction water or an organic solvent containing water can be used.
- the organic solvent include lower alcohols (monohydric or polyhydric alcohols having 1 to 4 carbon atoms such as methanol, ethanol, propanol, propylene glycol, butylene glycol, and glycerin) that can be mixed with water, acetone, and the like. It is done. These organic solvents may be used after being mixed with water in advance, or two or more kinds may be used by mixing with water. From the viewpoint of safety, it is preferable to extract only with water.
- the amount of the solvent used for the extraction is not particularly limited, and can be, for example, 200 to 3000 parts by mass with respect to 100 parts by mass of moringa seeds used for extraction.
- the temperature of the solvent at the time of extraction is not particularly limited, and can be, for example, 10 to 95 ° C. From the viewpoint of reducing the alkaloid content in the seed extract, extraction at a relatively low temperature is preferable. For example, 10 to 50 ° C. is preferable, and 20 to 40 ° C. is more preferable.
- alkaloids include spirotine. It is preferable to use a seed extract having a low alkaloid content as a PPAR activator because the PPAR activator can be safely ingested for a longer period of time. That is, a PPAR activator that substantially does not contain alkaloid derived from seed extract or does not contain at all is preferable.
- the extraction time is not particularly limited, but can be, for example, 30 to 150 minutes from the viewpoint of production efficiency.
- Extraction can be performed with stirring or stationary. Further, when the pretreatment is performed using the solvent used for extraction, the pretreatment and extraction can be performed continuously. For example, after the moringa is treated at 80 ° C. or higher for 1 to 10 minutes, the temperature is adjusted to 10 to 50 ° C. and the extract is extracted.
- the seed extract After extraction, a treatment such as filtration or centrifugation is performed to remove the residue, and then the extraction solvent is removed by decompression or the like to obtain a seed extract.
- a treatment such as filtration or centrifugation is performed to remove the residue, and then the extraction solvent is removed by decompression or the like to obtain a seed extract.
- the seed extract when used as a powder, the seed extract can be dried with a freeze dryer or a spray dryer, if necessary.
- the seed extract may be either liquid or solid, but a solid form such as powder is preferable from the viewpoint of easy transportation.
- the content of the moringa extract in the PPAR activator is not particularly limited, but can be, for example, 5 to 100% by mass.
- the PPAR activator can contain arbitrary additives such as excipients and lubricants.
- excipient examples include lactose, starch, cyclodextrin, galactomannan, dextrin, maltodextrin, etc., and from the viewpoints of actual use and allergenicity, preferably cyclodextrin, dextrin, or maltodextrin, more preferably Is dextrin or maltodextrin.
- the content of the excipient in the PPAR activator is not particularly limited, but is preferably 65 parts by mass or more from the viewpoint of stability with respect to 100 parts by mass of the dry solid content of the moringa extract. More preferably, it is 100 parts by mass or more, and preferably 1000 parts by mass or less, more preferably 500 parts by mass or less, from the viewpoint of reducing the amount of excipients used and reducing the cost.
- the timing of addition of the excipient is not particularly limited, and examples include moringa extract extraction, after removal of insoluble residue during the manufacture of the extract, and before powdering. From the viewpoint of improving filterability, it is insoluble. Addition after removal of the residue or before powdering is preferred, and addition after removal of the insoluble residue is more preferred.
- the activated PPAR may be any of the three subtypes of ⁇ , ⁇ / ⁇ , and ⁇ , but PPAR ⁇ / ⁇ is preferred, and all are more preferably activated.
- the PPAR activator regulates and induces fatty acid metabolism such as fatty acid uptake, transport, oxidation, and uncoupling protein in skeletal muscle, brain, liver, heart muscle, small intestine, spleen, and adipose tissue.
- the subject of this embodiment is effective in mammals having PPAR, and examples thereof include humans, mice, rats, rabbits, dogs, cats, cows, horses, pigs, monkeys, and the like.
- the PPAR activator of this aspect is, for example, recovery of physical and eye strain, improvement of whole body and muscle endurance, prevention and improvement of hypertension, prevention and improvement of hyperlipidemia, prevention and improvement of obesity, prevention of diabetes
- PPAR activator of this aspect is, for example, recovery of physical and eye strain, improvement of whole body and muscle endurance, prevention and improvement of hypertension, prevention and improvement of hyperlipidemia, prevention and improvement of obesity, prevention of diabetes
- it can be incorporated into various foods and drinks.
- Examples of foods and beverages include beverages such as soft drinks, carbonated drinks, nutritional drinks, fruit drinks, and lactic acid drinks, concentrated concentrates and powders for adjustment of these drinks, ice cream, sorbet, frozen confectionery such as shaved ice, soba, udon , Harusame, Gyoza skin, Suimai skin, Chinese noodles, instant noodles and other noodles, rice cake, candy, gum, chocolate, tablet candy, gummy candy, snack confectionery, biscuits, jelly, pudding, jam, cream, baked confectionery, etc.
- beverages such as soft drinks, carbonated drinks, nutritional drinks, fruit drinks, and lactic acid drinks, concentrated concentrates and powders for adjustment of these drinks, ice cream, sorbet, frozen confectionery such as shaved ice, soba, udon , Harusame, Gyoza skin, Suimai skin, Chinese noodles, instant noodles and other noodles, rice cake, candy, gum, chocolate, tablet candy, gummy candy, snack confectionery, biscuits, jelly, pudding, jam, cream, baked confection
- Processed fishery and livestock products such as confectionery, kamaboko, ham, sausage, dairy products such as processed milk and fermented milk, salad oil, tempura oil, margarine, mayonnaise, shortening, whipped cream, dressing, etc. , Seasonings such as sauce, soup, stew, salad, side dish, pickles, tablets , Capsules, fine granules (including powders), liquids (including syrups), and other forms of health and nutritional supplements, supplements, other mouth refreshers, mouth odors, etc. Agents, pet food and drinks, and the like.
- the blending amount of the PPAR activator is not particularly limited, but can be blended in the food and drink, for example, to be 0.0001 to 80% by mass in terms of dry extract solid content.
- the benzylglucosinolate-containing composition of this embodiment includes a moringa extract and an excipient. Although the mechanism by which the degradation of benzylglucosinolate in Moringa extract can be suppressed by including an excipient is not clear, it is presumed that the hydrolysis of benzylglucosinolate by the excipient is delayed.
- Moringa extract contained in the composition of this embodiment contains benzyl glucosinolate.
- the content of benzylglucosinolate in the moringa extract is preferably 6% by mass or more, more preferably 10% by mass or more, more preferably 15% by mass or more in terms of dry extract solid content, from the viewpoint of exhibiting useful physiological functions.
- the upper limit is not particularly limited, but can be, for example, 50% by mass or less.
- the moringa extract is preferably substantially free of alkaloids, more preferably completely free of alkaloids.
- the moringa extract can contain free amino acids, and can further contain one or more amino acids selected from the group consisting of arginine, glutamic acid, alanine, methionine, and cysteine, for example.
- the content of free amino acid in the moringa extract is preferably 0.1% by mass or more, more preferably 0.5% by mass or more in terms of dry solid content of the extract from the viewpoint of health promotion, and the upper limit is particularly limited. Although it is not a thing, it can be 2.0 mass% or less, for example.
- the content when two or more free amino acids are included refers to the total amount.
- Moringa extract may be extracted from Moringa using a solvent, or a commercially available product may be used. Examples of commercially available products include Moringa Extract 5% (manufactured by Apollo Ingredient, India). Some aspects of extracting from moringa are described below.
- Moringa used for extraction is not particularly limited, and examples thereof include Moringa oleifera, Moringa concanensis, Moringa drohardi and the like. Of these, Moringa oleifera is preferred from the viewpoint of widespread cultivation and easy collection. Moringa oleifera is a small deciduous tree that is native to India and has other names such as horseradish tree, Bennut, Malungai (Tagalog), and Sanjanaa (Hindu).
- any of leaves, stems, sheaths (fruits), and seeds can be used. These parts may be used as they are, or may be used after drying, but are preferably used after drying from the viewpoint of storage stability as a raw material and yield in producing the extract.
- pulp or seeds In view of the distribution amount of alkaloids by region, it is preferable to use pulp or seeds, and more preferable to use seeds, from the viewpoint of relatively few alkaloids.
- seed When seed is used, it may be used after removing the seed coat, or may be used after pulverized into powder. From the viewpoint of easily producing an extract, it is preferable to use a powdery material obtained by pulverizing seeds with a skin.
- a moringa extract that is substantially free of alkaloids can be obtained even if it is a leaf or stem part having a large amount of alkaloids, according to the production method of the embodiment described below.
- the dried moringa powder or the like is performed at 80 ° C. or higher, preferably 80 to 170 ° C., for example, preferably 80 to 95 ° C., more preferably 85 to 90 ° C.
- a method of treating in a liquid preferably for 1 to 10 minutes, more preferably for 3 to 5 minutes can be mentioned.
- pretreatment for a relatively short time is effective in this way.
- pretreatment within 10 minutes is preferable from the viewpoint of suppressing alkaloid extraction.
- the pretreatment of this embodiment is not particularly limited, and examples include boiled water, oil-like, roasting, and when using a medium, water, ethanol, animal and vegetable oils or mixtures thereof, and the like are safe. From the viewpoint of water, water is preferable.
- a solvent used for extraction water or an organic solvent containing water is used.
- the organic solvent include lower alcohols (monohydric or polyhydric alcohols having 1 to 4 carbon atoms such as methanol, ethanol, propanol, propylene glycol, butylene glycol, and glycerin) that can be mixed with water, acetone, and the like. It is done. These organic solvents may be used after being mixed with water in advance, or two or more kinds may be used by mixing with water. From the viewpoint of safety, it is preferable to extract only with water.
- the amount of the solvent used for the extraction is not particularly limited, and can be, for example, 200 to 3000 parts by mass with respect to 100 parts by mass of Moringa used for extraction.
- the temperature of the solvent at the time of extraction is not particularly limited, and can be, for example, 10 to 95 ° C., and preferably 10 to 50 ° C. from the viewpoint of reducing the production efficiency and the alkaloid content in the moringa extract. 20 to 40 ° C. is more preferable.
- the extraction time is not particularly limited, but can be, for example, 30 to 150 minutes from the viewpoint of production efficiency.
- Extraction can be carried out with stirring or at rest. Further, when the pretreatment is performed using the solvent used for extraction, the pretreatment and extraction can be performed continuously. For example, after the moringa is treated at 80 ° C. or higher for 1 to 10 minutes, the temperature is adjusted to 10 to 50 ° C. and the extract is extracted.
- the extract After extraction, treatment such as filtration or centrifugation is performed to remove the residue, and then the extraction solvent can be removed by decompression or the like.
- the extract when used as a powder, the extract can be dried with a spray dryer or the like as necessary.
- the moringa extract may be either liquid or solid, but the moringa extract used in the composition of this embodiment is preferably a solid form such as a powder.
- the excipient contained in the benzylglucosinolate-containing composition of the present embodiment can include, for example, one or more selected from the group consisting of maltodextrin, galactomannan, cyclodextrin, dextrin, starch, and lactose. From the viewpoint of actual use and allergenicity, it is preferably at least one selected from the group consisting of maltodextrin, galactomannan, and cyclodextrin, and more preferably maltodextrin.
- the content of the excipient in the benzylglucosinolate-containing composition of the present embodiment is 65 masses from the viewpoint of stability with respect to 100 parts by mass of the dry solid content of the moringa extract in the benzylglucosinolate-containing composition. Part or more, preferably 100 parts by weight or more. Also, from the viewpoint of reducing the amount of excipients used and reducing the cost, 1000 parts by weight or less is preferable, and 500 parts by weight or less is more preferable. Therefore, the content of the excipient of this embodiment is 65 to 1000 parts by weight, 65 to 500 parts by weight, 65 to 100 parts by weight, 100 to 1000 parts by weight, 100 to 500 parts by weight, 500 to 1000 parts by weight, etc. A suitable range is mentioned.
- the addition timing of the excipient is not particularly limited, but it may be after extraction, after removal of insoluble residue during extract production, before powdering, etc. From the viewpoint of improving filterability, removal of insoluble residue Addition after or before pulverization is preferred, and addition after removal of the insoluble residue is more preferred.
- the method for producing a benzylglucosinolate-containing composition of the present embodiment includes, for example, a method for producing a benzylglucosinolate-containing composition comprising a step of mixing a moringa extract and an excipient, And the like, and the like, and the like.
- the benzylglucosinolate-containing composition of this embodiment suppresses the decomposition of benzylglucosinolate and is excellent in stability. Therefore, the benzyl glucosinolate-containing composition of this embodiment contains a large amount of benzyl glucosinolate and can exhibit useful physiological functions.
- the benzyl glucosinolate-containing composition of this embodiment can be blended in various foods and drinks.
- beverages such as soft drinks, carbonated drinks, nutrition drinks, fruit drinks, and lactic acid drinks, concentrated concentrates of these drinks, and powders for adjustment may be used.
- this benzylglucosinolate-containing composition can be added to frozen desserts such as ice cream, sorbet, shaved ice, and other noodles such as buckwheat, udon, harusame, gyoza peel, cucumber peel, Chinese noodles, and instant noodles. it can.
- this benzylglucosinolate-containing composition can also be added to confectionery such as candy, candy, gum, chocolate, tablet confectionery, gummy candy, snack confectionery, biscuits, jelly, pudding, jam, cream, baked confectionery, etc. .
- benzylglucosinolate-containing compositions can be added to fishery and livestock processed foods such as kamaboko, ham and sausage, and dairy products such as processed milk and fermented milk, salad oil, tempura oil, margarine, mayonnaise, shortening, It can also be added to fats and oils such as whipped cream and dressings, seasonings such as sauces and sauces, soups, stews, salads, prepared dishes and pickles.
- various forms of health and nutritional supplements such as tablets, capsules and drinks, and other oral medicines such as mouth fresheners, dentifrices, mouthwashes, etc. It can be used by adding to external products, emollient cream, emollient lotion and the like.
- the blending amount of the benzylglucosinolate-containing composition of the present embodiment is not particularly limited, but it can be blended in the food and drink, for example, to be 0.01 to 80% by mass. Thereby, the content of benzyl glucosinolate in the food and drink of this embodiment can be 0.0006 to 40.0% by mass.
- the food / beverage products of this aspect contain the benzyl glucosinolate containing composition which contains many benzyl glucosinolates as above-mentioned, it can exhibit a useful physiological function.
- Moringa seeds were pulverized with a mill to obtain a pulverized seed. After adding 500 g of deionized water (90 ° C.) to 100 g of the seed pulverized product and stirring for 5 minutes, 1500 g of deionized water (10 ° C.) was added to 35 ° C. and stirred for 2 hours. It filtered with the back paper, and the filtrate was concentrate
- Moringa extract of Example 1-2 15 g was obtained in the same manner as Example 1-1 except that the dried moringa leaves were pulverized with a mill and the obtained pulverized leaves were used. Moringa stalk was pulverized with a hammer mill, and 10 g of moringa extract of Example 1-3 was obtained in the same manner as Example 1-1 except that the obtained pulverized stalk was used. Moringa extract of Example 1-4 was obtained in the same manner as Example 1-1 except that the moringa pod was cut into about 1 cm, freeze-dried, pulverized with a mill, and the obtained dried pulverized pod was used. 10 g was obtained.
- Example 1-7 1500 g of deionized water (35 ° C.) was added to 100 g of moringa seed pulverized product pulverized by a mill and steamed at 100 ° C. for 5 minutes, and stirred for 2 hours. Subsequent steps were carried out in the same manner as in Example 1-1 to obtain 7 g of Moringa extract of Example 1-7.
- Moringa stalk was pulverized with a hammer mill, and 20 g of the moringa extract of Comparative Example 1-5 was obtained in the same manner as Comparative Example 1-1 except that the obtained pulverized stalk was used.
- the moringa extract of Comparative Example 1-7 was prepared in the same manner as in Comparative Example 1-1 except that the moringa pod was cut into about 1 cm, freeze-dried, pulverized with a mill, and the resulting dried pulverized pod was used. 18g was obtained.
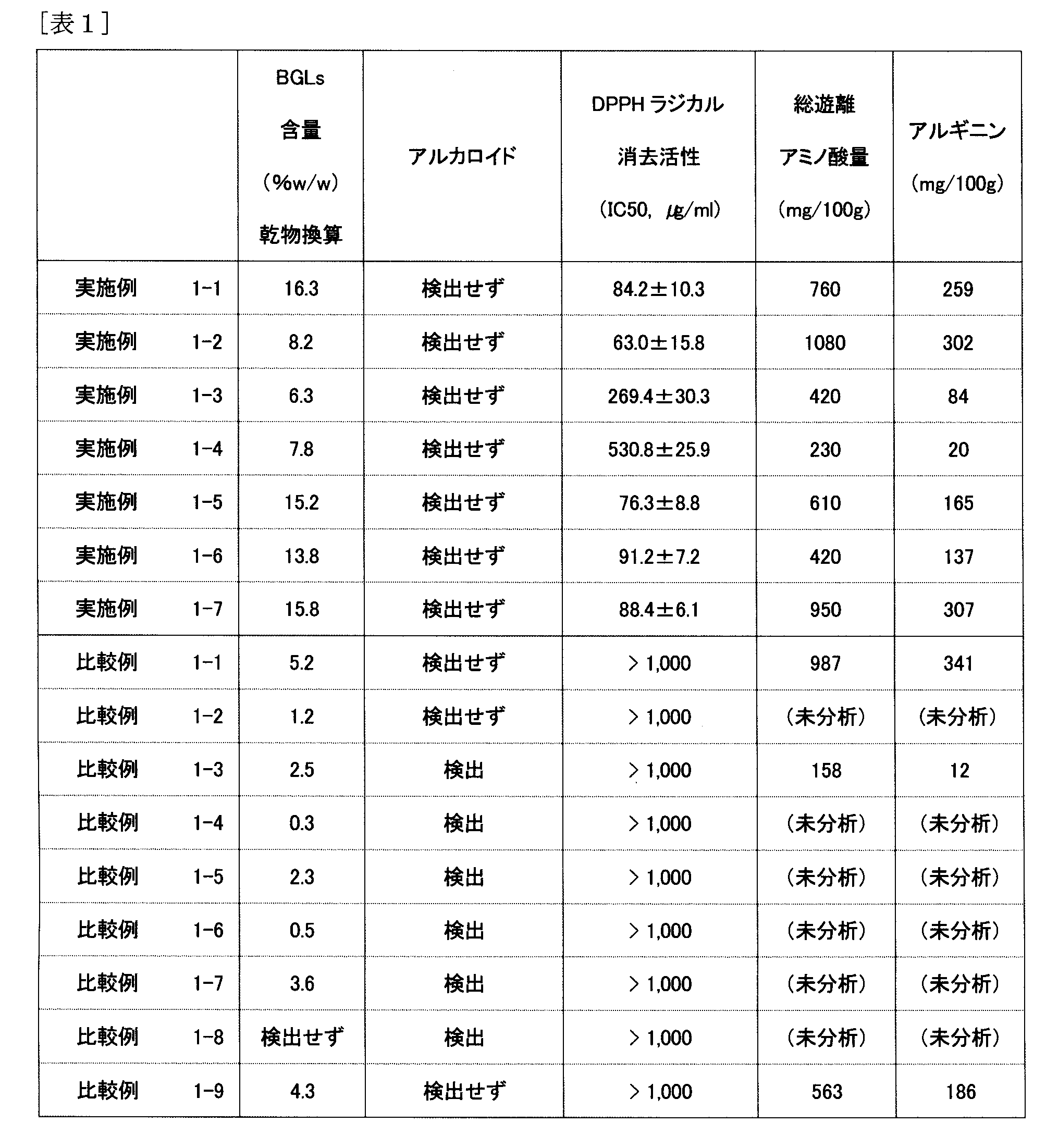
- Benzylglucosinolate content (BGLs content, in terms of extract dry solid content) of the Moringa extract of each Example and Comparative Example was analyzed based on the following conditions. The results are shown in Table 1. Moringa extract aqueous solutions (concentration: 0.2% (w / v)) of each Example and Comparative Example were prepared. Myrosinase (manufactured by Sigma) was added to these sample solutions and reacted at 30 ° C. for 16 hours. The reaction solution is diluted in a phosphate buffer solution (pH 8.5), 1,2-benzenedithiol is added, the mixture is treated at 65 ° C.
- BGLs content in terms of extract dry solid content
- DPPH radical scavenging activity was analyzed with reference to the method of Rao et al. (Reference: Austin J Nutr Metab-Volume 1 Issue 1-2014). . That is, 100 mg of each sample (dry powder) was dissolved in 100 ml of deionized water, diluted stepwise, and adjusted to a concentration of 0.5, 0.25, 0.125, 0.6125 mg / ml. .
- Moringa extracts of Examples 1-1 to 1-7 had a high content of benzyl glucosinolate, were excellent in DPPH radical scavenging activity, and an antioxidant action was observed.
- the Moringa extracts of Comparative Examples 1-1 to 1-9 extracted with hot water at 90 ° C. for 2 hours or with ethanol at 55 ° C. for 2 hours have a benzylglucosinolate content of Example 1-1 to Less than 1-7, and no DPPH radical scavenging activity was observed.
- alkaloids were not detected in any of Moringa extracts of Examples 1-1 to 1-7, and there was no problem in safety as shown in safety evaluations 1 and 2 below.
- Example 1-1 and Comparative Example 1-3 Using the obtained extract powders of Example 1-1 and Comparative Example 1-3, the safety of abortion was confirmed by animal experiments using rats. Specifically, female albino rats aged 8 to 10 weeks (weight range of 160 to 200 g per animal) were used, and after being habituated to the breeding environment for a predetermined period, they were paired with male rats. Rats in which sperm were confirmed by intravaginal observation after pairing were copied to another gauge, and Example 1-1 and Comparative Example 1-3 were forcibly administered at 200 mg / kg body weight per day for 10 days. The status of the intrauterine fetus was confirmed. As a control, a test group allowed to eat and drink freely after pairing was set. Seven rats were used in each test group.
- Example 1-1 Soft Drink (Composition) (Parts by Mass) Moringa extract of Example 1-1 0.5 Fructose glucose liquid sugar 11.0 Citric acid 0.2 Trisodium citrate 0.06 L-ascorbic acid 0.01 Fragrance 0.2 Dye 0.1 Water 87.93 Total amount 100.00
- Example 1-2 Carbonated beverage (composition) (parts by mass) Moringa extract of Example 1-1 0.5 Granulated sugar 8.0 Concentrated lemon juice 1.0 L-ascorbic acid 0.01 Citric acid 0.10 Sodium citrate 0.04 Coloring agent 0.05 Fragrance 0.15 Carbonated water 90.15 Total amount 100.00
- Example 1-3 Candy (composition) (parts by mass) Moringa extract of Example 1-1 1.0 Sugar 47.0 Minamata 49.0 Fragrance 1.0 Water 2.0 Total amount 100.0
- Example 1-4 Lozenge (composition) (parts by weight) Moringa extract of Example 1-1 0.5 Gum arabic 6.0 Glucose 72.0 Potassium phosphate 0.2 Potassium phosphate 0.1 Lactose 17.0 Fragrance 0.1 Magnesium stearate 4.1 Total amount 100.0
- Formulation Example 1-5 Tablet (tablet): (Composition) (Mass) Moringa extract of Example 1-1 80.0 Crystal Cellulose 10.0 Reduced maltose starch syrup powder 6.0 Calcium stearate 2.0 Shellac 2.0 Total amount 100.0
- Example 1-6 Powdered Soft Drink (Composition) (Mass) Moringa extract of Example 1-1 5.0 Dextrin 65.0 Raspberry juice powder 15.0 Black tea extract powder 5.0 Oligosaccharide 5.0 Cyclic oligosaccharide 2.5 Fragrance 0.9 Citric acid 1.0 Trisodium citrate 0.3 Stevia extract 0.3 Total amount 100.0
- Formulation Example 1-7 Gummy candy (composition) (parts by mass) Moringa extract of Example 1-1 10.0 Sugar 30.0 Minamata 32.0 Reduced water tank 12.0 Gum arabic 5.0 Ion exchange water 5.0 Gelatin 5.0 Coloring agent 0.2 Fragrance 0.8 Total amount 100.0
- Formulation Example 1-8 Gum (composition) (parts by weight) Moringa extract of Example 1-1 0.05 Gum base 20.0 Calcium carbonate 2.0 Stevia extract 0.1 Lactose 76.85 Fragrance 1.0 Total amount 100.00
- Example 1-9 Caramel (composition) (parts by weight) Moringa extract of Example 1-1 5.0 Granulated sugar 27.0 Minamata 20.0 Milk powder 40.0 Hardened oil 4.0 Salt 0.6 Fragrance 0.2 Water 3.2 Total amount 100.0
- Example 1-10 Jelly (Coffee Jelly) (Composition) (Mass) Moringa extract of Example 1-1 1.0 Granulated sugar 15.0 Gelatin 1.0 Coffee extract 5.0 Water 78.0 Total amount 100.0
- Example 1-11 Ice cream (composition) (parts by mass) Moringa extract of Example 1-1 1.0 Fresh cream (45% fat) 33.8 Nonfat dry milk 11.0 Granulated sugar 14.8 Sweetened egg yolk 0.3 Vanilla extract 0.1 Water 39.0 Total amount 100.0
- Formulation Example 1-12 Custard pudding (composition) (parts by mass) Moringa extract of Example 1-1 0.1 Milk 47.5 Whole egg 31.9 Super white sugar 17.1 Water 3.4 Total amount 100.0
- Example 1-13 Dentifrice (Composition) (parts by weight) Moringa extract of Example 1-1 0.05 Dicalcium phosphate 42.0 Glycerin 18.0 Carrageenan 0.9 Sodium lauryl sulfate 1.2 Saccharin sodium 0.09 Butyl paraoxybenzoate 0.005 Fragrance 1.0 Water 36.755 Total amount 100.00
- Formulation Example 1-14 Mouthwash (composition) (parts by weight) Moringa extract of Example 1-1 0.05 Sodium lauryl sulfate 0.8 Glycerin 7.0 Sorbitol 5.0 Ethyl alcohol 15.0 l-Menthol 0.05 Perfume 0.04 Saccharin sodium 0.1 Water 71.96 Total amount 100.00
- Emollient cream (composition) (parts by mass) Moringa extract of Example 1-1 0.1 Beeswax 2.0 Stearyl alcohol 5.0 Stearic acid 8.0 Squalane 10.0 Self-emulsifying propylene glycol monostearate 3.0 Polyoxyethylene cetyl ether (20EO) 1.0 Fragrance 0.5 Antioxidant Trace amount of preservative Trace amount of propylene glycol 4.8 Glycerin 3.0 Sodium hyaluronate 0.1 Triethanolamine 1.0 Purified water 61.5 Total amount 100.0
- Emollient lotion (composition) (parts by weight) Moringa extract of Example 1-1 0.1 Stearic acid 2.0 Cetanol 1.5 Vaseline 3.0 Lanolin alcohol 2.0 Liquid paraffin 10.0 Polyoxyethylene monooleate (10EO) 2.0 Fragrance 0.5 Antioxidant Trace amount of preservative Trace amount of propylene glycol 4.8 Glycerin 3.0 Sodium hyaluronate 0.1 Triethanolamine 1.0 Purified water 70.0 Total amount 100.0
- Preparation of PPAR activator Moringa seeds were pulverized in a mill to obtain a seed pulverized product. After adding 500 g of deionized water (90 ° C.) to 100 g of the seed pulverized product and stirring for 5 minutes, 1500 g of deionized water (10 ° C.) was added to 35 ° C., and the extract was extracted while stirring for 2 hours. . It filtered with the back paper, and the filtrate was concentrate
- Test Examples Examples 2-1 and 2-2 BAEC (bovine vascular endothelial cells) was implanted into the culture plate and cultured overnight (about 15 hours) at 37 ° C. and 5% CO 2 .
- DMEM High Glucose
- SIGMA Fetal Bovine Serum
- TransIT-LT1 (Mirus)
- a reporter vector (tK-PPREx3-LUC) having a PPAR responsive element (PPRE)
- PPRE PPAR responsive element
- an expression vector of human PPAR ⁇ , ⁇ / ⁇ , and ⁇ GS-hPPAR ⁇ , pCMX-, respectively
- hNUCI PPAR ⁇ , pCMX-hPPARg1
- ⁇ -galactosidase expression vector pSV- ⁇ -galactosidase
- Example 2-1 0.004% by mass
- Example 2-2 0.006% by mass
- a control group was prepared by adding 25 ⁇ l of distilled water alone and culturing. After completion of the culture, the cells were collected, and the luciferase activity of the cell extract was measured using Luciferase Assay Reagent (Progma). The gene transfer efficiency was corrected by the enzyme activity of ⁇ -galactosidase.
- the result is shown in the graph of FIG.
- each PPAR is activated, in particular, activation of PPAR ⁇ / ⁇ involved in lipid metabolism of skeletal muscle and differentiation of adipocytes It was found that the activation of PPAR ⁇ involved in induction and fat synthesis was remarkable.
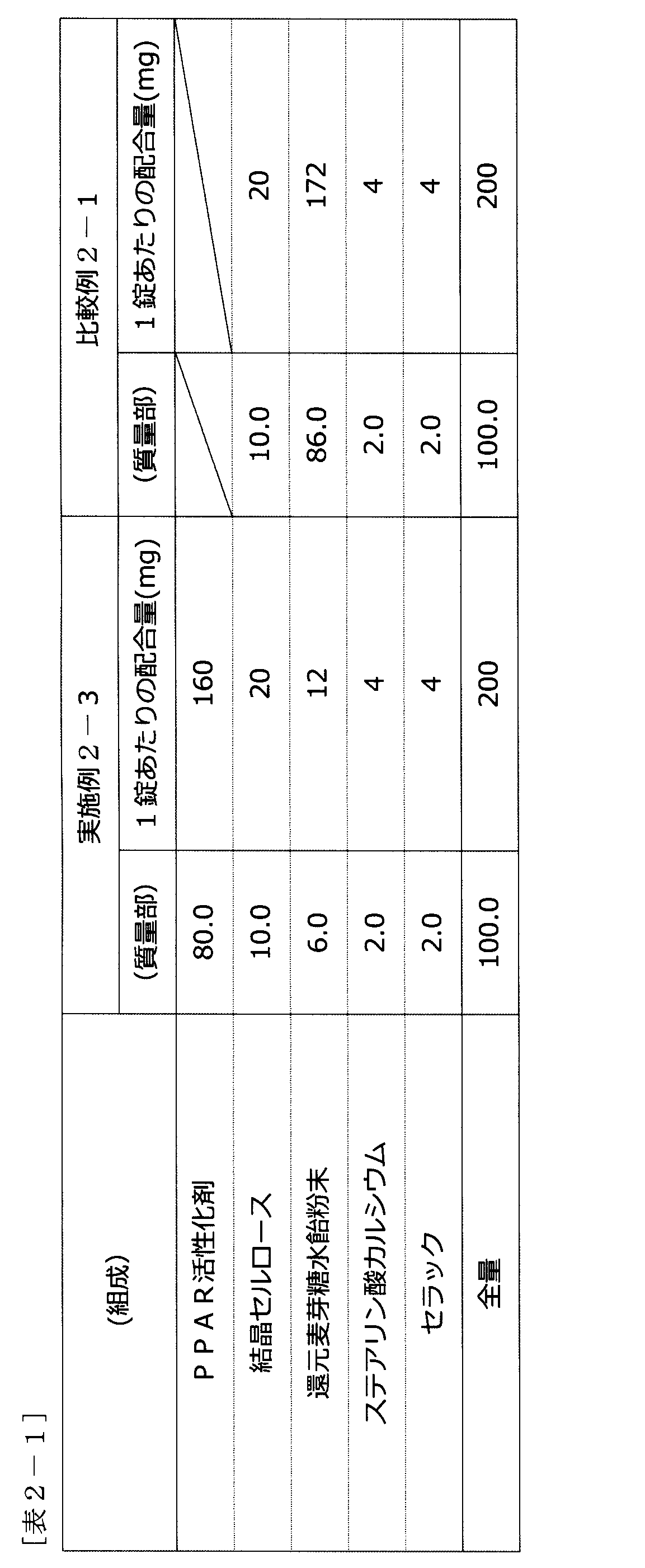
- Tablets using the PPAR activator prepared as described above were prepared with the composition shown in Table 2-1, and used as tablets of Example 2-3. Also, a tablet containing no PPAR activator was prepared and used as the tablet of Comparative Example 2-1. Volunteer tests using these were performed as follows.
- Example 2-3 and Comparative Example 2-1 were ingested 2 tablets a day for 4 consecutive weeks without knowing which volunteers had ingested. After ingestion, after a washout period of one month, tablets different from those ingested earlier were ingested 2 tablets a day for 4 consecutive weeks.
- ⁇ Evaluation method The evaluation method was as follows. 1. Feeling of general fatigue: As a subjective evaluation of volunteers after ingestion, regarding the change before ingestion and after ingestion for 4 weeks, ⁇ was effective, and x was not effective or worsened, The number of people marked with ⁇ is shown in Table 2-2 (when ingesting Example 2-3) and Table 2-3 (when ingesting Comparative Example 2-1). 2. Eye fatigue: The same method as for general fatigue. 3. 3-minute walking distance: Evaluated as an index of endurance throughout the body. In other words, we used a 400-meter track at an athletic stadium and measured how many meters volunteers walked in 3 minutes.
- Table 2-2 (when ingesting Example 2-3) and Table 2-3 (comparative example) are shown to be effective for volunteers who have increased distance by increasing and decreasing the difference between the distance after intake for 4 weeks and before intake. 2-1)).
- Increase / decrease in maximum blood pressure Measure blood pressure after intake for 4 weeks and before intake, and increase / decrease the difference. Volunteers with decreased (negative) values are shown in Table 2-2 as effective persons. -3) and Table 2-3 (Comparative Example 2-1). 5.
- Increase / decrease in blood triglycerides Measure blood triglycerides after intake for 4 weeks and before intake, and increase / decrease the difference between them. The results are shown in 2-2 (when ingesting Example 2-3) and Table 2-3 (when ingesting Comparative Example 2-1).
- Example 2-3 In Table 2-2 (intake result of Example 2-3), the improvement trend was remarkable for each item as compared to Table 2-3 (intake result of Comparative Example 2-1).
- Example 2-3 When Example 2-3 was ingested, it was confirmed that all 8 people showed an improvement effect with 2 or more items, but no significant improvement effect was seen when Comparative Example 2-1 was ingested.
- Comparative Example 2-1 In addition, everyone took 2 tablets daily for 4 weeks as instructed, and none of the volunteers had any abnormal physical condition due to tablet intake.
- Example 2-1 Soft Drink (Composition) (Parts by Mass) PPAR activator of Example 2-1 0.5 Fructose glucose liquid sugar 11.0 Citric acid 0.2 Trisodium citrate 0.06 L-ascorbic acid 0.01 Fragrance 0.2 Dye 0.1 Water 87.93 Total amount 100.00
- Example 2-2 Candy (composition) (parts by mass) PPAR Activator of Example 2-1 1.0 Sugar 47.0 Minamata 49.0 Fragrance 1.0 Water 2.0 Total amount 100.0
- Example 2-3 Gummy candy (composition) (parts by mass) PPAR Activator of Example 2-1 10.0 Sugar 30.0 Minamata 32.0 Reduced water tank 12.0 Gum arabic 5.0 Ion exchange water 5.0 Gelatin 5.0 Coloring agent 0.2 Fragrance 0.8 Total amount 100.0
- Example 2-4 Chocolate (composition) (parts by mass) PPAR Activator of Example 2-1 1.0 Sugar 44.5 Whole milk powder 20.0 Cocoa Butter 20.0 Cacaomas 13.5 Emulsifier 0.5 Fragrance 0.5 Total amount 100.0
- the dextrin mixed filtration solution was concentrated under reduced pressure using a rotary evaporator, and the resulting concentrated solution was dried with a freeze dryer to obtain 20 g of the benzylglucosinolate-containing composition of Example 3-1.
- 20 g of the benzylglucosinolate-containing composition of Example 3-2 was obtained in the same manner as in Example 3-1, except that cyclodextrin was used instead of maltodextrin.
- 20 g of the benzylglucosinolate-containing composition of Example 3-3 was obtained in the same manner as in Example 3-1, except that galactomannan was used instead of maltodextrin.
- the benzylglucosinolate-containing composition of Example 3-4 was prepared in the same manner as in Example 3-1, except that the amount of maltodextrin added was 75 parts by mass with respect to 100 parts by mass of the total solid content of Moringa extract. 16 g was obtained.
- the benzylglucosinolate-containing composition of Example 3-5 was prepared in the same manner as in Example 3-1, except that the amount of maltodextrin to be added was 1000 parts by mass with respect to 100 parts by mass of the total solid content of Moringa extract. 110 g was obtained.
- reaction solution is diluted in a phosphate buffer solution (pH 8.5), 1,2-benzenedithiol is added, the mixture is treated at 65 ° C. for 2 hours, and the product (1,3-benzenedithiol- 2-thione) was quantitatively analyzed by reverse phase high performance liquid chromatography under the following conditions.
- Example 3-1 Soft Drink (Composition) (parts by mass) Composition of Example 3-1 1.0 Fructose glucose liquid sugar 10.5 Citric acid 0.2 Trisodium citrate 0.06 L-ascorbic acid 0.01 Fragrance 0.2 Dye 0.1 Water 87.93 Total amount 100.00
- Example 3-2 Carbonated beverage (composition) (parts by mass) Composition of Example 3-1 1.0 Granulated sugar 7.5 Concentrated lemon juice 1.0 L-ascorbic acid 0.01 Citric acid 0.10 Sodium citrate 0.04 Coloring agent 0.05 Fragrance 0.15 Carbonated water 90.15 Total amount 100.00
- Example 3-3 Candy (composition) (parts by mass) Composition of Example 3-1 2.0 Sugar 46.0 Minamata 49.0 Fragrance 1.0 Water 2.0 Total amount 100.0
- Example 3-4 Lozenges (composition) (parts by weight) Composition of Example 3-1 1.0 Gum arabic 6.0 Glucose 71.0 Potassium phosphate 0.2 Potassium phosphate 0.1 Lactose 17.0 Fragrance 0.1 Magnesium stearate 4.1 Total amount 100.0
- Formulation Example 3-5 Tablet (Composition) (Mass) Composition of Example 3-1 80.0 Crystal Cellulose 10.0 Reduced maltose starch syrup powder 6.0 Calcium stearate 2.0 Shellac 2.0 Total amount 100.0
- Example 3-6 Powdered Soft Drink (Composition) (Parts by Mass) Composition of Example 3-1 10.0 Dextrin 60.0 Raspberry juice powder 15.0 Black tea extract powder 5.0 Oligosaccharide 5.0 Cyclic oligosaccharide 2.5 Fragrance 0.9 Citric acid 1.0 Trisodium citrate 0.3 Stevia extract 0.3 Total amount 100.0
- Formulation Example 3-7 Gummy Candy (composition) (parts by weight) Composition of Example 3-1 20.0 Sugar 25.0 Minamata 27.0 Reduced water tank 12.0 Gum arabic 5.0 Ion exchange water 5.0 Gelatin 5.0 Coloring agent 0.2 Fragrance 0.8 Total amount 100.0
- the moringa extract of the present invention that solves the first problem is useful in the field of food and drink.
- the PPAR activator of the present invention that solves the second problem has an excellent PPAR activation action, can be taken for a long time without problems of side effects, and is preferably used for foods and drinks, etc. Can do. Therefore, the PPAR activator of the present invention that solves the second problem is only for the prevention of diseases such as insulin resistance, hyperinsulinemia, type 2 diabetes, hypertension, hyperlipidemia, arteriosclerosis, obesity, etc. Rather, it can be expected to be used as a food, supplement, and medicine for recovery from fatigue and improvement in endurance by improving basal metabolism.
- the benzyl glucosinolate containing composition of this invention which solves a 3rd subject is useful in field
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Food Science & Technology (AREA)
- Polymers & Plastics (AREA)
- Pharmacology & Pharmacy (AREA)
- Botany (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Inorganic Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Nutrition Science (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Diabetes (AREA)
- Natural Medicines & Medicinal Plants (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Epidemiology (AREA)
- Mycology (AREA)
- Molecular Biology (AREA)
- Microbiology (AREA)
- Medical Informatics (AREA)
- Biotechnology (AREA)
- Alternative & Traditional Medicine (AREA)
- Cardiology (AREA)
- Heart & Thoracic Surgery (AREA)
- Child & Adolescent Psychology (AREA)
- Endocrinology (AREA)
- Emergency Medicine (AREA)
- Urology & Nephrology (AREA)
- Vascular Medicine (AREA)
- Ophthalmology & Optometry (AREA)
- Medicines Containing Plant Substances (AREA)
Abstract
Description
[1]ベンジルグルコシノレートの含有量が、エキス乾燥固形分換算で6質量%以上であり、アルカロイドを実質的に含まない、モリンガエキス、
[2]80℃以上にて前処理したモリンガを10~50℃の溶媒中で抽出する工程を含む、モリンガエキスの製造方法、
および、
[3][1]記載のモリンガエキスを含む、飲食品に関する。
[1]モリンガエキスを含む、ペルオキシソーム増殖剤応答性受容体(PPAR)活性化剤、
[2][1]記載のPPAR活性化剤を含む、飲食品、および
[3] 80℃以上にて前処理したモリンガを10~50℃の溶媒中で抽出する工程を含む、モリンガエキスを含むPPAR活性化剤の製造方法に関する。
[1]モリンガエキスと、賦形剤とを含むベンジルグルコシノレート含有組成物であって、前記賦形剤の含有量が、前記モリンガエキスの乾燥固形分100質量部に対して65質量部以上である、組成物、
[2][1]記載の組成物を含む、飲食品、
および、
[3]モリンガエキスと賦形剤とを混合する工程を含む、ベンジルグルコシノレート含有組成物の製造方法であって、前記賦形剤の配合量が、前記モリンガエキスの乾燥固形分100質量部に対して65~1000質量部である、製造方法に関する。
薄層プレートにはシリカゲル60F(メルク社製)を、展開溶媒にはクロロホルム/メタノール/25%アンモニア水(75/25/2、v/v/v)を、発色試薬にはドラーゲンドルフ試薬をそれぞれ用いて測定する。
また、このモリンガエキスを、アイスクリーム、シャーベット、かき氷などの冷菓や、そば、うどん、はるさめ、ぎょうざの皮、しゅうまいの皮、中華麺、即席麺などの麺類に添加することもできる。さらに、このモリンガエキスを、飴、キャンディ、ガム、チョコレート、錠菓、グミキャンディー、スナック菓子、ビスケット、ゼリー、プリン、ジャム、クリーム、焼き菓子などの菓子類に添加することもできる。
また、モリンガエキスを、かまぼこ、ハム、ソーセージなどの水産・畜産加工食品や、加工乳、発酵乳などの乳製品に添加したり、サラダ油、てんぷら油、マーガリン、マヨネーズ、ショートニング、ホイップクリームおよびドレッシングなどの油脂および油脂加工食品、ソース、たれなどの調味料や、スープ、シチュー、サラダ、惣菜、漬物などに添加することもできる。加えて、錠剤、カプセル剤、ドリンク剤など種々の形態の健康・栄養補助食品、その他口中清涼剤、口臭防止剤などの口腔内で使用する口腔清涼剤、歯磨剤、洗口液などの医薬部外品、エモリエントクリーム、エモリエントローションなどに添加して用いることができる。
また、このベンジルグルコシノレート含有組成物を、アイスクリーム、シャーベット、かき氷などの冷菓や、そば、うどん、はるさめ、ぎょうざの皮、しゅうまいの皮、中華麺、即席麺などの麺類に添加することもできる。さらに、このベンジルグルコシノレート含有組成物を、飴、キャンディ、ガム、チョコレート、錠菓、グミキャンディー、スナック菓子、ビスケット、ゼリー、プリン、ジャム、クリーム、焼き菓子などの菓子類に添加することもできる。
また、ベンジルグルコシノレート含有組成物を、かまぼこ、ハム、ソーセージなどの水産・畜産加工食品や、加工乳、発酵乳などの乳製品に添加したり、サラダ油、てんぷら油、マーガリン、マヨネーズ、ショートニング、ホイップクリームおよびドレッシングなどの油脂および油脂加工食品、ソース、たれなどの調味料や、スープ、シチュー、サラダ、惣菜、漬物などに添加することもできる。加えて、錠剤、カプセル剤、ドリンク剤など種々の形態の健康・栄養補助食品、その他口中清涼剤、口臭防止剤などの口腔内で使用する口腔清涼剤、歯磨剤、洗口液などの医薬部外品、エモリエントクリーム、エモリエントローションなどに添加して用いることができる。
モリンガ種子をミルにて粉砕し、種子粉砕物を得た。種子粉砕物100gに対して、500gの脱イオン水(90℃)を加え、5分間攪拌した後に、脱イオン水(10℃)を1500g加えて35℃とし、2時間攪拌した。その後ろ紙にてろ過し、ろ液をロータリーエバポレーターにより減圧濃縮した。得られた濃縮液を凍結乾燥機にて乾燥し、実施例1-1のモリンガエキスを10g得た。
乾燥モリンガ葉をミルにて粉砕し、得られた葉粉砕物を用いた以外は実施例1-1と同様にして、実施例1-2のモリンガエキスを15g得た。
モリンガ茎をハンマーミルにて粉砕し、得られた茎粉砕物を用いた以外は実施例1-1と同様にして、実施例1-3のモリンガエキスを10g得た。
モリンガ鞘を1センチ程度に切り分け、凍結乾燥させ、ミルにて粉砕し、得られた鞘乾燥粉砕物を用いた以外は実施例1-1と同様にして、実施例1-4のモリンガエキスを10g得た。
ミルで粉砕したモリンガ種子粉砕物100gを、140℃に熱した食用油で5分間揚げたものに、脱イオン水(35℃)を1500g加えて2時間攪拌した。その後の工程を実施例1-1と同様にして、実施例1-5のモリンガエキスを8g得た。
ミルで粉砕したモリンガ種子粉砕物100gを、170℃に熱した鉄板で5分間炒めたものに、脱イオン水(35℃)を1500g加えて2時間攪拌した。その後の工程を実施例1-1と同様にして、実施例1-6のモリンガエキスを8g得た。
ミルで粉砕したモリンガ種子粉砕物100gを、蒸し器にて100℃で5分間蒸したものに、脱イオン水(35℃)を1500g加えて2時間攪拌した。その後の工程を実施例1-1と同様にして、実施例1-7のモリンガエキスを7g得た。
モリンガ種子をミルにて粉砕し、種子粉砕物を得た。種子粉砕物100gに対して、2000gの脱イオン水(90℃)を加え、2時間攪拌した。その後ろ紙にてろ過し、ろ液をロータリーエバポレーターにより減圧濃縮した。得られた濃縮液を凍結乾燥機にて乾燥し、比較例1-1のモリンガエキスを15g得た。
乾燥モリンガ葉をミルにて粉砕し、得られた葉粉砕物を用いた以外は比較例1-1と同様にして、比較例1-3のモリンガエキスを20g得た。
モリンガ茎をハンマーミルにて粉砕し、得られた茎粉砕物を用いた以外は比較例1-1と同様にして、比較例1-5のモリンガエキスを20g得た。
モリンガ鞘を1センチ程度に切り分け、凍結乾燥させ、ミルにて粉砕し、得られた鞘乾燥粉砕物を用いた以外は比較例1-1と同様にして、比較例1-7のモリンガエキスを18g得た。
モリンガ種子をミルにて粉砕し、種子粉砕物を得た。種子粉砕物100gに対して、2000gの50%(v/v)エタノール水溶液(55℃)を加え、2時間攪拌した。その後ろ紙にてろ過し、ろ液をロータリーエバポレーターにより減圧濃縮した。得られた濃縮液を凍結乾燥機にて乾燥し、比較例1-2のモリンガエキスを12g得た。
乾燥モリンガ葉をミルにて粉砕し、得られた葉粉砕物を用いた以外は比較例1-2と同様にして、比較例1-4のモリンガエキスを25g得た。
モリンガ茎をハンマーミルにて粉砕し、得られた茎粉砕物を用いた以外は比較例1-2と同様にして、比較例1-6のモリンガエキスを25g得た。
モリンガ鞘を1センチ程度に切り分け、凍結乾燥させ、ミルにて粉砕し、得られた鞘乾燥粉砕物を用いた以外は比較例1-2と同様にして、比較例1-8のモリンガエキスを20g得た。
モリンガ種子をミルにて粉砕し、種子粉砕物を得た。種子粉砕物100gに対して、2000gの脱イオン水(35℃)を加え、2時間攪拌した。その後ろ紙にてろ過し、ろ液をロータリーエバポレーターにより減圧濃縮した。得られた濃縮液を凍結乾燥機にて乾燥し、比較例1-9のモリンガエキスを8g得た。
各実施例および比較例のモリンガエキスのベンジルグルコシノレート含有量(BGLs含有量、エキス乾燥固形分換算)について、以下の条件に基づき分析した。結果を表1に示す。
各実施例および比較例のモリンガエキス水溶液(濃度:0.2%(w/v))を調製した。これらのサンプル溶液にミロシナーゼ(シグマ製)を加え、30℃にて16時間反応させた。反応液をリン酸緩衝溶液(pH8.5)中に希釈し、1,2-ベンゼンジチオールを加え、65℃で2時間処理し、処理物中に含まれる生成物(1,3-ベンゼンジチオール-2-チオン)を以下の条件で逆相高速液体クロマトグラフィーにて定量分析した。
HPLC:LC-20AD(島津製作所)
ガラム:L-カラムODS(メーカー:一般財団法人 化学物質評価研究機構、内径:4.6mm、長さ:250mm、粒径:5ミクロン)
サンプル注入量:10μL
検出:UV365nm
溶媒:水/メタノール(20/80,v/v)
流速:0.5mL/分
各実施例および比較例のモリンガエキスからアルカロイドが検出されるか否かについて、薄層クロマトグラフィーにて分析した。結果を表1に示す。
試料:実施例および比較例のメタノール溶液(濃度:1mg/mL)
プレートへの滴下量:100μL(サンプルの固形滴下量:約100マイクログラム)
薄層プレート:シリカゲル60F(メルク社製)
展開溶媒:クロロホルム/メタノール/25%アンモニア水(75/25/2、v/v/v)
発色試薬:ドラーゲンドルフ試薬
各実施例および比較例のモリンガエキスの有用性を確認するために、DPPHラジカルの消去活性をRaoらの方法を参考にして分析した(参考文献:Austin J Nutr Metab - Volume 1 Issue 1 - 2014)。
すなわち、各サンプル(乾燥粉末)100mgを100mlの脱イオン水に溶解させ、段階的に希釈し、0.5,0.25,0.125,0.6125mg/mlの濃度となるように調整した。各濃度のサンプル溶液0.05mlに対して、1mlの2,2-ジフェニル-1-ピクリルヒドラジル(DPPH)のエタノール溶液(濃度:0.1mM)、0.45mlの50mMトリス―塩酸緩衝液(pH7.4)を加え、1時間常温暗所にて保持後に、吸光度517nmで測定し、50%阻害濃度(IC50)を算出した。
一方、90℃で2時間の熱水抽出、あるいは55℃で2時間のエタノール抽出した比較例1-1~1-9のモリンガエキスは、ベンジルグルコシノレートの含有量が実施例1-1~1-7よりも少なく、また、DPPHラジカル消去活性が認められなかった。
また、実施例1-1~1-7のモリンガエキスからはいずれもアルカロイドが検出されず、以下の安全性評価1、2において示すように、安全性に問題のないものであった。
得られた実施例1-1のモリンガエキスの安全性を簡易的に評価するために、単回投与試験(ラット)を行ったところ、体重1kgあたり5gおよび2.5g摂取させても死亡にいたるラットは見られなかった。よって、実施例1-1のモリンガエキスの安全性は問題ないものと考えられる。
得られた実施例1-1および比較例1-3のエキス粉末を用いて、安全性について、堕胎活性をラットを用いた動物実験で確認した。すなわち、8~10週齢のメスアルビノラット(1匹あたりの体重幅160~200g)を用い、所定期間飼育環境に慣れさせた後に、雄ラットとペアリングさせた。ペアリング後に膣内観察にて精子が確認されたラットを別ゲージに写し、実施例1-1および比較例1-3を、1日あたり体重1kgあたり200mgを、10日間強制投与し、投与後の子宮内胎児の状況を確認した。コントロールとして、ペアリング後に自由飲食させた試験群を設定した。各試験区には7匹のラットを用いた。その結果、比較例1-3を摂取させたラットは、ペアリングを確認したにもかかわらず、子宮内胎児は全て死亡していたが、実施例1-1摂取群およびコントロール群は、子宮内胎児は同様の発育を示していた。よって、実施例1-1のモリンガエキスの安全性は問題ないものと考えられる。
(組成) (質量部)
実施例1-1のモリンガエキス 0.5
果糖ブドウ糖液糖 11.0
クエン酸 0.2
クエン酸3ナトリウム 0.06
L-アスコルビン酸 0.01
香料 0.2
色素 0.1
水 87.93
全量 100.00
(組成) (質量部)
実施例1-1のモリンガエキス 0.5
グラニュー糖 8.0
濃縮レモン果汁 1.0
L-アスコルビン酸 0.01
クエン酸 0.10
クエン酸ナトリウム 0.04
着色料 0.05
香料 0.15
炭酸水 90.15
全量 100.00
(組成) (質量部)
実施例1-1のモリンガエキス 1.0
砂糖 47.0
水飴 49.0
香料 1.0
水 2.0
全量 100.0
(組成) (質量部)
実施例1-1のモリンガエキス 0.5
アラビアガム 6.0
ブドウ糖 72.0
リン酸第二カリウム 0.2
リン酸第一カリウム 0.1
乳糖 17.0
香料 0.1
ステアリン酸マグネシウム 4.1
全量 100.0
(組成) (質量部)
実施例1-1のモリンガエキス 80.0
結晶セルロール 10.0
還元麦芽糖水飴粉末 6.0
ステアリン酸カルシウム 2.0
セラック 2.0
全量 100.0
(組成) (質量部)
実施例1-1のモリンガエキス 5.0
デキストリン 65.0
ラズベリー果汁粉末 15.0
紅茶エキス粉末 5.0
オリゴ糖 5.0
環状オリゴ糖 2.5
香料 0.9
クエン酸 1.0
クエン酸3ナトリウム 0.3
ステビア抽出物 0.3
全量 100.0
(組成) (質量部)
実施例1-1のモリンガエキス 10.0
砂糖 30.0
水飴 32.0
還元水飴 12.0
アラビアガム 5.0
イオン交換水 5.0
ゼラチン 5.0
着色料 0.2
香料 0.8
全量 100.0
(組成) (質量部)
実施例1-1のモリンガエキス 0.05
ガムベース 20.0
炭酸カルシウム 2.0
ステビア抽出物 0.1
乳糖 76.85
香料 1.0
全量 100.00
(組成) (質量部)
実施例1-1のモリンガエキス 5.0
グラニュー糖 27.0
水飴 20.0
粉乳 40.0
硬化油 4.0
食塩 0.6
香料 0.2
水 3.2
全量 100.0
(組成) (質量部)
実施例1-1のモリンガエキス 1.0
グラニュー糖 15.0
ゼラチン 1.0
コーヒーエキス 5.0
水 78.0
全量 100.0
(組成) (質量部)
実施例1-1のモリンガエキス 1.0
生クリーム(45%脂肪) 33.8
脱脂粉乳 11.0
グラニュー糖 14.8
加糖卵黄 0.3
バニラエッセンス 0.1
水 39.0
全量 100.0
(組成) (質量部)
実施例1-1のモリンガエキス 0.1
牛乳 47.5
全卵 31.9
上白糖 17.1
水 3.4
全量 100.0
(組成) (質量部)
実施例1-1のモリンガエキス 0.05
第二リン酸カルシウム 42.0
グリセリン 18.0
カラギーナン 0.9
ラウリル硫酸ナトリウム 1.2
サッカリンナトリウム 0.09
パラオキシ安息香酸ブチル 0.005
香料 1.0
水 36.755
全量 100.00
(組成) (質量部)
実施例1-1のモリンガエキス 0.05
ラウリル硫酸ナトリウム 0.8
グリセリン 7.0
ソルビトール 5.0
エチルアルコール 15.0
l-メントール 0.05
香料 0.04
サッカリンナトリウム 0.1
水 71.96
全量 100.00
(組成) (質量部)
実施例1-1のモリンガエキス 0.1
ミツロウ 2.0
ステアリルアルコール 5.0
ステアリン酸 8.0
スクアラン 10.0
自己乳化型プロピレングリコールモノステアレート 3.0
ポリオキシエチレンセチルエーテル(20EO) 1.0
香料 0.5
酸化防止剤 微量
防腐剤 微量
プロピレングリコール 4.8
グリセリン 3.0
ヒアルロン酸ナトリウム 0.1
トリエタノールアミン 1.0
精製水 61.5
全量 100.0
(組成) (質量部)
実施例1-1のモリンガエキス 0.1
ステアリン酸 2.0
セタノール 1.5
ワセリン 3.0
ラノリンアルコール 2.0
流動パラフィン 10.0
ポリオキシエチレンモノオレイン酸エステル(10EO) 2.0
香料 0.5
酸化防止剤 微量
防腐剤 微量
プロピレングリコール 4.8
グリセリン 3.0
ヒアルロン酸ナトリウム 0.1
トリエタノールアミン 1.0
精製水 70.0
全量 100.0
モリンガ種子をミルにて粉砕し、種子粉砕物を得た。種子粉砕物100gに対して、500gの脱イオン水(90℃)を加え、5分間攪拌した後に、脱イオン水(10℃)を1500g加えて35℃とし、2時間攪拌しながらエキスを抽出した。その後ろ紙にてろ過し、ろ液をロータリーエバポレーターにより減圧濃縮した。得られた濃縮液を凍結乾燥機にて乾燥し、種子エキスを10g得た。得られた種子エキスを、PPAR活性化剤とした。このPPAR活性化剤からアルカロイドが検出されるか否かについて、薄層クロマトグラフィー(以下条件)にて分析したところ、発色スポットが見られず、アルカロイドが検出されなかった。
試料:PPAR活性化剤のメタノール溶液(濃度:1mg/mL)
プレートへの滴下量:100μL(サンプルの固形滴下量:約100マイクログラム)
薄層プレート:シリカゲル60F(メルク社製)
展開溶媒:クロロホルム/メタノール/25%アンモニア水(75/25/2、v/v/v)
発色試薬:ドラーゲンドルフ試薬
実施例2-1、2-2
培養プレートに、BAEC(ウシ血管内皮細胞)を植え込み、37℃、5%CO2条件下で一晩(約15時間)培養した。培地にはFetal Bovine Serum(SIGMA)を10%添加したDMEM(High Glucose)(ナカライテスク)を用いた。次いで、TransIT-LT1(Mirus)を用いて、PPAR応答性エレメント(PPRE)をもつレポーターベクター(tK-PPREx3-LUC)、ヒトPPARα、β/δ、γの発現ベクター(それぞれGS-hPPARα、pCMX-hNUCI、pCMX-hPPARg1)、およびβーガラクトシダーゼ発現ベクター(pSV-β-Galactosidase)を共導入し、37℃、5%CO2条件下で4時間培養し、培地交換した後にさらに24時間培養した。培養後、前記のとおり調製したPPAR活性化剤の水溶液(実施例2-1:0.004質量%、実施例2-2:0.006質量%)を各ウェルに25μl加え、24時間培養した。なお、蒸留水のみを25μl加えて培養したものを対照群とした。培養終了後、細胞を回収し、Luciferase Assay Reagent(Progma)を用いて、細胞抽出液のルシフェラーゼ活性を測定した。なお、βーガラクトシダーゼの酵素活性で遺伝子導入効率を補正した。各実施例について、実施例及び対照群の発光強度の平均値(n=3)の比(実施例/対照群)を算出し、対照群に対する相対ルシフェラーゼ活性を実施例のPPAR活性化能とした。この結果を図1のグラフに示す。
前記のとおり調製したPPAR活性化剤を用いた錠剤を、表2-1に記載の組成で調製し、実施例2-3の錠剤とした。また、PPAR活性化剤を含まない錠剤をあわせて調製し、比較例2-1の錠剤とした。これらを用いたボランティア試験を以下のように行った。
・ボランティア対象者
35才~55才のデスクワーク中心であり、かつ日常的な運動をしていない男性会社員8名(他の薬やサプリメントを摂取していない、病院にかかっていない、および規則的な食生活を送っている)とした。
二重盲検比較試験として行った。すなわち、実施例2-3および比較例2-1の錠剤を、ボランティアはどちらを摂取しているかわからない状態で、1日2錠を4週間連続で摂取した。摂取後、1ヶ月間のウォッシュアウト期間を経て、先に摂取したものとは異なる錠剤を、1日2錠を4週間連続で摂取した。
以下のとおり行った。
1.全身疲労感:摂取後のボランティア自身の主観的な評価として、摂取前と4週間摂取後の変化について、効果があったものを○、効果を感じられなかった、あるいは悪化したものを×とし、○をつけた人数を表2-2(実施例2-3摂取時)および表2-3(比較例2-1摂取時)に記した。
2.眼の疲労感:全身疲労感と同じ方法で行った。
3.3分間歩行距離:全身持久力の指標として評価した。すなわち、陸上競技場の400メートルトラックを使用し、ボランティアが3分間のうちに何メートル歩いたかを計測した。4週間摂取後の距離と摂取前との差を増減とし、距離が伸びたボランティアには効果があった人として表2-2(実施例2-3摂取時)および表2-3(比較例2-1摂取時)に記した。
4.最高血圧の増減:4週間摂取後および摂取前の血圧を測定し、その差を増減とし、数値が下がった(マイナスとなった)ボランティアは効果があった人として表2-2(実施例2-3摂取時)および表2-3(比較例2-1摂取時)に記した。
5.血中中性脂肪の増減:4週間摂取後および摂取前の血中中性脂肪を測定し、その差を増減とし、数値が下がった(マイナスとなった)ボランティアは効果があった人として表2-2(実施例2-3摂取時)および表2-3(比較例2-1摂取時)に記した。
(組成) (質量部)
実施例2-1のPPAR活性化剤 0.5
果糖ブドウ糖液糖 11.0
クエン酸 0.2
クエン酸3ナトリウム 0.06
L-アスコルビン酸 0.01
香料 0.2
色素 0.1
水 87.93
全量 100.00
(組成) (質量部)
実施例2-1のPPAR活性化剤 1.0
砂糖 47.0
水飴 49.0
香料 1.0
水 2.0
全量 100.0
(組成) (質量部)
実施例2-1のPPAR活性化剤 10.0
砂糖 30.0
水飴 32.0
還元水飴 12.0
アラビアガム 5.0
イオン交換水 5.0
ゼラチン 5.0
着色料 0.2
香料 0.8
全量 100.0
(組成) (質量部)
実施例2-1のPPAR活性化剤 1.0
砂糖 44.5
全粉乳 20.0
ココアバター 20.0
カカオマス 13.5
乳化剤 0.5
香料 0.5
全量 100.0
モリンガ種子をミルにて粉砕し、種子粉砕物を得た。種子粉砕物100gに対して、500gの脱イオン水(90℃)を加え、5分間攪拌した後に、脱イオン水(10℃)を1500g加えて35℃とし、2時間攪拌した。その後ろ紙にてろ過し、固形分含量を加熱残分として測定し、総固形分含量と同量のマルトデキストリンを加えた。デキストリン混合ろ過溶液を、ロータリーエバポレーターにより減圧濃縮し、得られた濃縮液を凍結乾燥機にて乾燥し、実施例3-1のベンジルグルコシノレート含有組成物を20g得た。
マルトデキストリンに代えて、サイクロデキストリンを用いた以外は実施例3-1と同様にして、実施例3-2のベンジルグルコシノレート含有組成物を20g得た。
マルトデキストリンに代えて、ガラクトマンナンを用いた以外は実施例3-1と同様にして、実施例3-3のベンジルグルコシノレート含有組成物を20g得た。
添加するマルトデキストリン量を、モリンガエキス総固形分含量100質量部に対して75質量部とした以外は実施例3-1と同様にして、実施例3-4のベンジルグルコシノレート含有組成物を16g得た。
添加するマルトデキストリン量を、モリンガエキス総固形分含量100質量部に対して1000質量部とした以外は実施例3-1と同様にして、実施例3-5のベンジルグルコシノレート含有組成物を110g得た。
マルトデキストリンを加えず、ろ過後に、減圧濃縮した以外は実施例3-1と同様にして、比較例3-1のベンジルグルコシノレート含有組成物を10g得た。
添加するマルトデキストリン量を、モリンガエキス総固形分含量100質量部に対して50質量部とした以外は実施例3-1と同様にして、比較例3-2のベンジルグルコシノレート含有組成物を13g得た。
各実施例および比較例のベンジルグルコシノレート含有組成物をポリエチレン袋(内袋)とアルミ袋(外袋)に入れ、55℃にて保管するとともに、所定期間ごとにサンプルを袋から取り出し、ベンジルグルコシノレート含有量(BGLs含有量)について、以下の条件に基づき分析した。分析結果を表3に示す。
各実施例および比較例のモリンガエキス水溶液(濃度:0.2%(w/v))を調製した。これらのサンプル溶液にミロシナーゼ(シグマ製)を加え、30℃にて16時間反応させた。反応液をリン酸緩衝溶液(pH8.5)中に希釈し、1,2-ベンゼンジチオールを加え、65℃で2時間処理し、処理物中に含まれる生成物(1,3-ベンゼンジチオール-2-チオン)を以下の条件で逆相高速液体クロマトグラフィーにて定量分析した。
HPLC:LC-20AD(島津製作所)
ガラム:L-カラムODS(メーカー:一般財団法人 化学物質評価研究機構、内径:4.6mm、長さ:250mm、粒径:5ミクロン)
サンプル注入量:10μL
検出:UV365nm
溶媒:水/メタノール(20/80,v/v)
流速:0.5mL/分
(組成) (質量部)
実施例3-1の組成物 1.0
果糖ブドウ糖液糖 10.5
クエン酸 0.2
クエン酸3ナトリウム 0.06
L-アスコルビン酸 0.01
香料 0.2
色素 0.1
水 87.93
全量 100.00
(組成) (質量部)
実施例3-1の組成物 1.0
グラニュー糖 7.5
濃縮レモン果汁 1.0
L-アスコルビン酸 0.01
クエン酸 0.10
クエン酸ナトリウム 0.04
着色料 0.05
香料 0.15
炭酸水 90.15
全量 100.00
(組成) (質量部)
実施例3-1の組成物 2.0
砂糖 46.0
水飴 49.0
香料 1.0
水 2.0
全量 100.0
(組成) (質量部)
実施例3-1の組成物 1.0
アラビアガム 6.0
ブドウ糖 71.0
リン酸第二カリウム 0.2
リン酸第一カリウム 0.1
乳糖 17.0
香料 0.1
ステアリン酸マグネシウム 4.1
全量 100.0
(組成) (質量部)
実施例3-1の組成物 80.0
結晶セルロール 10.0
還元麦芽糖水飴粉末 6.0
ステアリン酸カルシウム 2.0
セラック 2.0
全量 100.0
(組成) (質量部)
実施例3-1の組成物 10.0
デキストリン 60.0
ラズベリー果汁粉末 15.0
紅茶エキス粉末 5.0
オリゴ糖 5.0
環状オリゴ糖 2.5
香料 0.9
クエン酸 1.0
クエン酸3ナトリウム 0.3
ステビア抽出物 0.3
全量 100.0
(組成) (質量部)
実施例3-1の組成物 20.0
砂糖 25.0
水飴 27.0
還元水飴 12.0
アラビアガム 5.0
イオン交換水 5.0
ゼラチン 5.0
着色料 0.2
香料 0.8
全量 100.0
Claims (18)
- ベンジルグルコシノレートの含有量が、エキス乾燥固形分換算で6質量%以上であり、アルカロイドを実質的に含まない、モリンガエキス。
- エキスの抽出溶媒が水である、請求項1に記載のモリンガエキス。
- モリンガエキスを含む、ペルオキシソーム増殖剤応答性受容体(PPAR)活性化剤。
- 前記モリンガエキスが種子エキスである、請求項3記載のPPAR活性化剤。
- PPARα、PPARβ/δ、およびPPARγの少なくともいずれかを活性化させる、請求項3または4記載のPPAR活性化剤。
- 前記モリンガエキスが、モリンガ・オレイフェラ(Moringa oleifera)を原料とするものである、請求項3~5いずれか記載のPPAR活性化剤。
- 前記モリンガエキスの抽出溶媒が水である、請求項3~6いずれか記載のPPAR活性化剤。
- 前記モリンガエキスが、請求項1または2記載のモリンガエキスである、請求項3~7いずれか記載のPPAR活性化剤。
- モリンガエキスと、賦形剤とを含むベンジルグルコシノレート含有組成物であって、前記賦形剤の含有量が、前記モリンガエキスの乾燥固形分100質量部に対して65質量部以上である、組成物。
- 前記賦形剤の含有量が、前記モリンガエキスの乾燥固形分100質量部に対して65~1000質量部である、請求項9記載の組成物。
- 前記賦形剤が、マルトデキストリン、ガラクトマンナン、サイクロデキストリン、デキストリン、でんぷん、および乳糖からなる群より選ばれる1種以上を含む、請求項9または10記載の組成物。
- 前記モリンガエキスが、請求項1または2記載のモリンガエキスである、請求項9~11いずれか記載の組成物。
- モリンガエキスと賦形剤とを混合する工程を含む、ベンジルグルコシノレート含有組成物の製造方法であって、前記賦形剤の配合量が、前記モリンガエキスの乾燥固形分100質量部に対して65~1000質量部である、製造方法。
- 請求項1もしくは2記載のモリンガエキス、請求項3~8いずれか記載のPPAR活性化剤、または請求項9~12いずれか記載の組成物を含む、飲食品。
- 清涼飲料、炭酸飲料、栄養飲料、果実飲料、これらの飲料の調整用粉末、キャンディ、ガム、錠菓、グミキャンディー、および健康・栄養補助食品(錠剤、カプセル剤、ドリンク剤)からなる群より選択される、請求項14記載の飲食品。
- 肉体および眼精疲労の回復、全身および筋持久力の向上、高血圧の予防および改善、高脂血症の予防および改善、肥満の予防および改善、糖尿病の予防および改善、ならびにインスリン抵抗性および高インスリン血症の予防および改善からなる群より選択される少なくとも1以上の目的のための、請求項14または15記載の飲食品。
- 80℃以上で前処理したモリンガを10~50℃の溶媒中で抽出する工程を含む、モリンガエキスの製造方法。
- モリンガエキスにおけるベンジルグルコシノレートの含有量が、エキス乾燥固形分換算で6質量%以上であり、アルカロイドを実質的に含まないものである、請求項17記載のモリンガエキスの製造方法。
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2017512397A JP6200122B1 (ja) | 2015-10-28 | 2016-10-21 | モリンガエキス |
| US15/770,620 US10441618B2 (en) | 2015-10-28 | 2016-10-21 | Moringa extract |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015-211780 | 2015-10-28 | ||
| JP2015211780 | 2015-10-28 | ||
| JP2015-211781 | 2015-10-28 | ||
| JP2015211781 | 2015-10-28 | ||
| JP2015-247740 | 2015-12-18 | ||
| JP2015247740 | 2015-12-18 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2017073473A1 true WO2017073473A1 (ja) | 2017-05-04 |
Family
ID=58630352
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2016/081237 Ceased WO2017073473A1 (ja) | 2015-10-28 | 2016-10-21 | モリンガエキス |
Country Status (3)
| Country | Link |
|---|---|
| US (1) | US10441618B2 (ja) |
| JP (2) | JP6200122B1 (ja) |
| WO (1) | WO2017073473A1 (ja) |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107467446A (zh) * | 2017-08-07 | 2017-12-15 | 云南肠和健康科技股份有限公司 | 一种火龙果酵素饮料及其制备方法 |
| CN107616394A (zh) * | 2017-08-07 | 2018-01-23 | 云南肠和健康科技股份有限公司 | 一种酵素饮料及其制备方法 |
| JP2018062477A (ja) * | 2016-10-12 | 2018-04-19 | 日清食品ホールディングス株式会社 | 脂肪蓄積抑制剤及びこれを含有する飲食品 |
| CN108713621A (zh) * | 2018-05-30 | 2018-10-30 | 海口海味榔生物科技专业合作社 | 一种辣木咖啡奶及其制备方法 |
| CN108771074A (zh) * | 2018-05-17 | 2018-11-09 | 广西柳州市桑辰科技有限公司 | 一种辣木籽固体饮料的制作方法 |
| JP6693007B1 (ja) * | 2018-12-07 | 2020-05-13 | 太陽化学株式会社 | モリンガ抽出物及び/又は粉砕物を含む組成物 |
| US10869829B1 (en) | 2018-12-07 | 2020-12-22 | Taiyo Kagaku Co., Ltd. | Composition containing moringa extract and/or pulverized product |
| JP2021029182A (ja) * | 2019-08-26 | 2021-03-01 | 太陽化学株式会社 | 腰痛改善用組成物 |
| JP2022116861A (ja) * | 2021-01-29 | 2022-08-10 | 株式会社ファーマフーズ | イソチオシアネート富化モリンガおよびそれを製造する方法 |
| JP2023098105A (ja) * | 2021-12-28 | 2023-07-10 | サッポロビール株式会社 | 飲料、飲料の製造方法、及び、飲料の香味向上方法 |
| JP2025076522A (ja) * | 2021-03-31 | 2025-05-15 | 株式会社ナリス化粧品 | 乳頭突起形成促進剤のスクリーニング方法および、乳頭突起形成促進剤 |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10441618B2 (en) * | 2015-10-28 | 2019-10-15 | Taiyo Kagaku Co., Ltd. | Moringa extract |
| JP7116462B2 (ja) * | 2016-12-22 | 2022-08-10 | トスレック株式会社 | 飲料製造システムおよび飲料製造方法 |
| KR102084859B1 (ko) * | 2018-10-16 | 2020-03-04 | 사단법인 한국불교 바로알기 | 울금꽃 모링가 발효효소 및 이의 제조방법 |
| JP7126759B2 (ja) * | 2018-11-05 | 2022-08-29 | カゴメ株式会社 | グルコシノレート含有飲食品、グルコシノレート含有飲食品の製造方法、グルコシノレート含有飲食品の経時変化によるグルコシノレート量の減少を抑制する方法 |
| JP7048475B2 (ja) * | 2018-11-05 | 2022-04-05 | カゴメ株式会社 | グルコシノレート含有飲食品、グルコシノレート含有飲食品の製造方法、グルコシノレート含有飲食品の経時変化によるグルコシノレート量の減少を抑制する方法 |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2008237117A (ja) * | 2007-03-27 | 2008-10-09 | Kinos:Kk | 抗疲労食品素材及び抗疲労食品 |
| US20100227828A1 (en) * | 2007-11-22 | 2010-09-09 | Laila Nutraceuticals | New synergistic phytochemical composition for the treatment of obesity |
| JP2012530769A (ja) * | 2009-06-22 | 2012-12-06 | ピエール ファーブル デルモ−コスメティック | モリンガ種の完全な種子の抽出物並びに化粧品組成物及び/又は皮膚科学的組成物におけるその使用 |
| WO2013183065A2 (en) * | 2012-06-06 | 2013-12-12 | Ramaswamy Rajendran | Extraction of phytochemicals from plant matter |
| WO2014053944A1 (en) * | 2012-10-03 | 2014-04-10 | University Of The Witwatersrand, Johannesburg | Extract from morigaceae and a method to prepare the extract |
| JP2014208608A (ja) * | 2013-03-22 | 2014-11-06 | ライオン株式会社 | 糖代謝改善剤 |
| US20150209395A1 (en) * | 2013-11-01 | 2015-07-30 | Rutgers, The State University Of New Jersey | Extracts From Plants of the Moringaceae Family and Methods of Making |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH07304685A (ja) | 1994-03-18 | 1995-11-21 | Takeda Shokuhin Kogyo Kk | 発癌プロモーション抑制組成物 |
| JP4719372B2 (ja) | 2000-06-21 | 2011-07-06 | 花王株式会社 | Ppar依存的遺伝子転写活性化剤 |
| JP4032393B2 (ja) | 2004-07-09 | 2008-01-16 | 有限会社イッシンランバー | 栄養機能補助食品、モリンガ茶葉及びモリンガ茶飲料 |
| US10441618B2 (en) * | 2015-10-28 | 2019-10-15 | Taiyo Kagaku Co., Ltd. | Moringa extract |
-
2016
- 2016-10-21 US US15/770,620 patent/US10441618B2/en active Active
- 2016-10-21 WO PCT/JP2016/081237 patent/WO2017073473A1/ja not_active Ceased
- 2016-10-21 JP JP2017512397A patent/JP6200122B1/ja active Active
-
2017
- 2017-08-24 JP JP2017161489A patent/JP6745250B2/ja active Active
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2008237117A (ja) * | 2007-03-27 | 2008-10-09 | Kinos:Kk | 抗疲労食品素材及び抗疲労食品 |
| US20100227828A1 (en) * | 2007-11-22 | 2010-09-09 | Laila Nutraceuticals | New synergistic phytochemical composition for the treatment of obesity |
| JP2012530769A (ja) * | 2009-06-22 | 2012-12-06 | ピエール ファーブル デルモ−コスメティック | モリンガ種の完全な種子の抽出物並びに化粧品組成物及び/又は皮膚科学的組成物におけるその使用 |
| WO2013183065A2 (en) * | 2012-06-06 | 2013-12-12 | Ramaswamy Rajendran | Extraction of phytochemicals from plant matter |
| WO2014053944A1 (en) * | 2012-10-03 | 2014-04-10 | University Of The Witwatersrand, Johannesburg | Extract from morigaceae and a method to prepare the extract |
| JP2014208608A (ja) * | 2013-03-22 | 2014-11-06 | ライオン株式会社 | 糖代謝改善剤 |
| US20150209395A1 (en) * | 2013-11-01 | 2015-07-30 | Rutgers, The State University Of New Jersey | Extracts From Plants of the Moringaceae Family and Methods of Making |
Non-Patent Citations (4)
| Title |
|---|
| D.I.GERNAH: "Effect of boiling time on the quality of 'Zogale':A snack food produced from peanut(Arachis hypogea)cake and boiled Moringa oleifera Leaves", AFRICAN JOURNAL OF FOOD SCIENCE, vol. 6, no. 10, 30 May 2012 (2012-05-30), pages 287 - 293, XP055380038 * |
| EMMANUEL ORMAN: "Investigating the In-vivo Antiplasmodial Properties of Aqueous Extract of Moringa oleifera Lam(Moringaceae)Leaves", BRITISH JOURNAL OF PHARMACEUTICAL RESEARCH, vol. 5, no. 6, 2 February 2015 (2015-02-02), pages 419 - 430, XP055380026 * |
| MARIATERESA MALDINI: "Moringa oleifera:study of phenolics and glucosinolates by mass spectrometry", J.MASS SPECTROM, vol. 49, 2014, pages 900 - 910, XP055380031 * |
| W.SANGKITIKOMOL: "Effect of Moringa oleifera on advanced glycation end-product formation and lipid metabolism gene expression in HepG2 cells", GENETICS AND MOLECULAR RESEARCH, vol. 13, no. 1, 2014, pages 723 - 735, XP055380025 * |
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2018062477A (ja) * | 2016-10-12 | 2018-04-19 | 日清食品ホールディングス株式会社 | 脂肪蓄積抑制剤及びこれを含有する飲食品 |
| WO2018070312A1 (ja) * | 2016-10-12 | 2018-04-19 | 日清食品ホールディングス株式会社 | 脂肪蓄積抑制剤及びこれを含有する飲食品 |
| CN107467446A (zh) * | 2017-08-07 | 2017-12-15 | 云南肠和健康科技股份有限公司 | 一种火龙果酵素饮料及其制备方法 |
| CN107616394A (zh) * | 2017-08-07 | 2018-01-23 | 云南肠和健康科技股份有限公司 | 一种酵素饮料及其制备方法 |
| CN108771074A (zh) * | 2018-05-17 | 2018-11-09 | 广西柳州市桑辰科技有限公司 | 一种辣木籽固体饮料的制作方法 |
| CN108713621A (zh) * | 2018-05-30 | 2018-10-30 | 海口海味榔生物科技专业合作社 | 一种辣木咖啡奶及其制备方法 |
| JP6693007B1 (ja) * | 2018-12-07 | 2020-05-13 | 太陽化学株式会社 | モリンガ抽出物及び/又は粉砕物を含む組成物 |
| US10869829B1 (en) | 2018-12-07 | 2020-12-22 | Taiyo Kagaku Co., Ltd. | Composition containing moringa extract and/or pulverized product |
| JP2021029182A (ja) * | 2019-08-26 | 2021-03-01 | 太陽化学株式会社 | 腰痛改善用組成物 |
| JP2022116861A (ja) * | 2021-01-29 | 2022-08-10 | 株式会社ファーマフーズ | イソチオシアネート富化モリンガおよびそれを製造する方法 |
| JP7362134B2 (ja) | 2021-01-29 | 2023-10-17 | 株式会社ファーマフーズ | イソチオシアネート富化モリンガおよびそれを製造する方法 |
| JP2025076522A (ja) * | 2021-03-31 | 2025-05-15 | 株式会社ナリス化粧品 | 乳頭突起形成促進剤のスクリーニング方法および、乳頭突起形成促進剤 |
| JP7826534B2 (ja) | 2021-03-31 | 2026-03-09 | 株式会社ナリス化粧品 | 乳頭突起形成促進剤のスクリーニング方法および、乳頭突起形成促進剤 |
| JP2023098105A (ja) * | 2021-12-28 | 2023-07-10 | サッポロビール株式会社 | 飲料、飲料の製造方法、及び、飲料の香味向上方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP6745250B2 (ja) | 2020-08-26 |
| US20180318367A1 (en) | 2018-11-08 |
| JPWO2017073473A1 (ja) | 2017-10-26 |
| JP2017217006A (ja) | 2017-12-14 |
| JP6200122B1 (ja) | 2017-09-20 |
| US10441618B2 (en) | 2019-10-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6200122B1 (ja) | モリンガエキス | |
| CN110023487B (zh) | 新型清酒乳杆菌和包含其的组合物 | |
| KR102798356B1 (ko) | 락토바실러스 플란타룸 mg989를 포함하는 항산화용 조성물 | |
| JP5027361B2 (ja) | ヒアルロン酸産生促進剤 | |
| JP2025157605A (ja) | 肝機能改善剤 | |
| JP2016008180A (ja) | 筋肉持久力向上剤 | |
| JP7296611B2 (ja) | 一酸化窒素産生促進剤 | |
| JP2010037221A (ja) | アディポネクチン産生増強剤 | |
| JP2024060053A (ja) | 糖化に起因した保湿機能低下の抑制剤、皮膚化粧料および飲食品 | |
| JP7481023B2 (ja) | エンテロコッカス・フェカーリスを有効成分として含有する肥満または肥満から誘導された代謝症候群の予防または治療用の組成物 | |
| JP6456032B2 (ja) | Sirt1活性化剤および当該Sirt1活性化剤の利用 | |
| JP5175442B2 (ja) | ヤーコン由来の抗ガン剤 | |
| JP7605576B2 (ja) | 皮膚老化改善剤のスクリーニング方法 | |
| JP2010013416A (ja) | 育毛効果を有する藻類エキス | |
| JP6518744B2 (ja) | 経口用組成物 | |
| JP7834457B2 (ja) | IL-1α発現抑制剤 | |
| JP6010482B2 (ja) | 経口用組成物 | |
| JP2012206964A (ja) | PPAR−α活性調節剤 | |
| JP6629036B2 (ja) | 皮膚化粧料および飲食品 | |
| JP2018123077A (ja) | Adl改善剤 | |
| JP6241631B2 (ja) | 経口用組成物 | |
| JP2025155784A (ja) | 睡眠の質向上用組成物 | |
| JP2023059099A (ja) | マイオネクチン産生促進剤 | |
| JP2026041600A (ja) | Trpa1活性化剤およびtrpv1活性化剤 | |
| WO2023243210A1 (ja) | 筋芽細胞増殖促進剤及び筋芽細胞増殖促進用組成物、並びに筋萎縮抑制剤及び筋萎縮抑制用組成物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2017512397 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 16859703 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15770620 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 16859703 Country of ref document: EP Kind code of ref document: A1 |