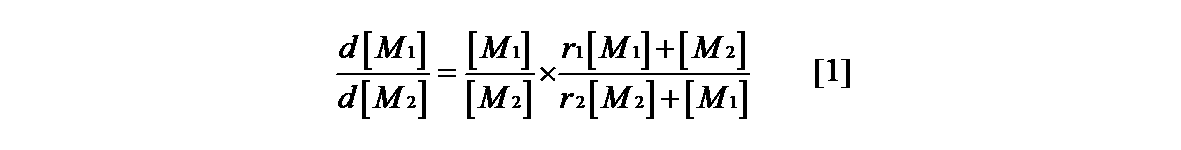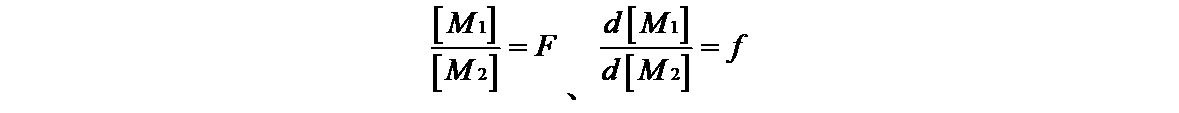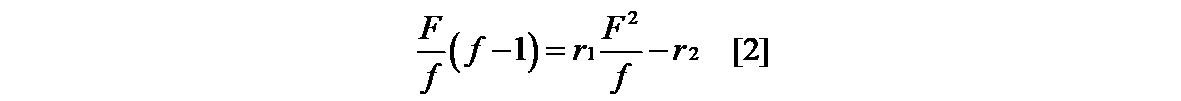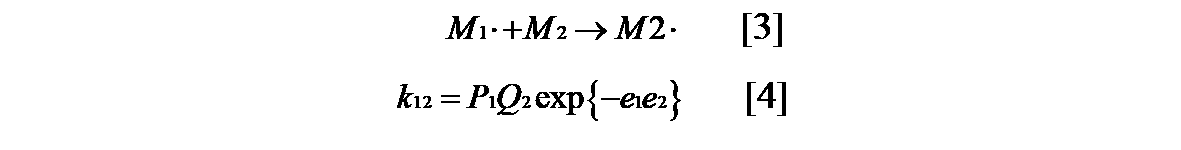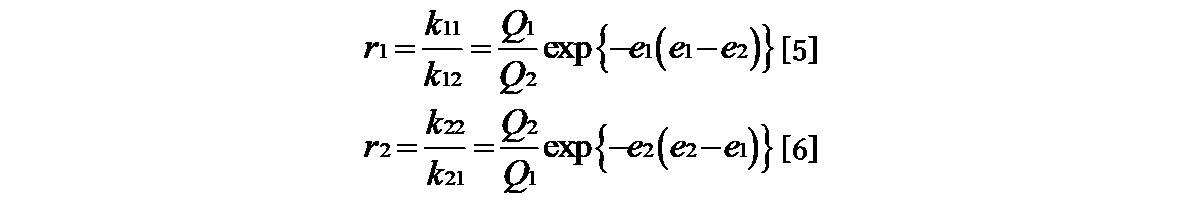WO2021177459A1 - 分散液 - Google Patents
分散液 Download PDFInfo
- Publication number
- WO2021177459A1 WO2021177459A1 PCT/JP2021/008794 JP2021008794W WO2021177459A1 WO 2021177459 A1 WO2021177459 A1 WO 2021177459A1 JP 2021008794 W JP2021008794 W JP 2021008794W WO 2021177459 A1 WO2021177459 A1 WO 2021177459A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- monomer
- fluorine
- weight
- group
- repeating unit
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D185/00—Coating compositions based on macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing atoms other than silicon, sulfur, nitrogen, oxygen, and carbon; Coating compositions based on derivatives of such polymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L27/00—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Compositions of derivatives of such polymers
- C08L27/02—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L27/12—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Compositions of derivatives of such polymers not modified by chemical after-treatment containing fluorine atoms
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/21—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/244—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of halogenated hydrocarbons
- D06M15/248—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of halogenated hydrocarbons containing chlorine
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F214/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen
- C08F214/02—Monomers containing chlorine
- C08F214/04—Monomers containing two carbon atoms
- C08F214/06—Vinyl chloride
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F214/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen
- C08F214/02—Monomers containing chlorine
- C08F214/04—Monomers containing two carbon atoms
- C08F214/08—Vinylidene chloride
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F214/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen
- C08F214/02—Monomers containing chlorine
- C08F214/14—Monomers containing three or more carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/10—Esters
- C08F220/22—Esters containing halogen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/10—Esters
- C08F220/22—Esters containing halogen
- C08F220/24—Esters containing halogen containing perhaloalkyl radicals
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L27/00—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Compositions of derivatives of such polymers
- C08L27/02—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L27/04—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Compositions of derivatives of such polymers not modified by chemical after-treatment containing chlorine atoms
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L33/00—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides or nitriles thereof; Compositions of derivatives of such polymers
- C08L33/04—Homopolymers or copolymers of esters
- C08L33/14—Homopolymers or copolymers of esters of esters containing halogen, nitrogen, sulfur, or oxygen atoms in addition to the carboxy oxygen
- C08L33/16—Homopolymers or copolymers of esters containing halogen atoms
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/21—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/263—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of unsaturated carboxylic acids; Salts or esters thereof
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/21—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/263—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of unsaturated carboxylic acids; Salts or esters thereof
- D06M15/277—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of unsaturated carboxylic acids; Salts or esters thereof containing fluorine
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/21—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/285—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of unsaturated carboxylic acid amides or imides
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M2200/00—Functionality of the treatment composition and/or properties imparted to the textile material
- D06M2200/10—Repellency against liquids
- D06M2200/11—Oleophobic properties
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M2200/00—Functionality of the treatment composition and/or properties imparted to the textile material
- D06M2200/10—Repellency against liquids
- D06M2200/12—Hydrophobic properties
Definitions
- This disclosure relates to dispersions.
- An unreacted vinyl chloride monomer or vinylidene chloride simple containing a copolymer obtained by copolymerizing a polyfluoroalkyl group-containing monomer, vinyl chloride or vinylidene chloride, and other copolymerizable monomers By using a water-dispersible water-repellent oil-repellent having a monomer concentration of 10 ppm or less, a water-repellent oil-repellent with excellent storage stability is provided in which problems such as morphological change and performance deterioration of the water-repellent oil-repellent are reduced. It is known that it can be used (Patent Document 1).
- An object of the present disclosure is to provide a novel dispersion liquid capable of achieving both product stability and water repellency and / or oil repellency.
- One embodiment in the present disclosure is: [Item 1] A dispersion containing a fluorine-containing polymer and a liquid medium. Fluorine-containing polymer (a) having a Q value of 2.0 or more and containing a fluoroalkyl group. A repeating unit derived from, and a chloride monomer (b) which is at least one selected from vinyl chloride and vinylidene chloride. A dispersion containing a repeating unit derived from, and having a concentration of unreacted chloride monomer (b) of 2.0 ppm or less.
- Item 2 The dispersion liquid according to Item 1, which is a compound represented by.
- Item 3 Item 2.
- Item 4 Item 2.
- Item 5 Item 2.
- the fluorine-containing polymer A 22 is a long-chain hydrocarbon group-containing monomer (c1) which is an acyclic aliphatic hydrocarbon group having 12 to 30 carbon atoms.
- Item 6 The dispersion according to Item 6, which comprises a repeating unit derived from.
- Item 8 The total of the repeating unit derived from the fluorine-containing monomer (a), the repeating unit derived from the chloride monomer (b), and the repeating unit derived from the hydrocarbon group-containing monomer (c) is Item 6.
- Item 2 The dispersion liquid according to any one of Items 1 to 8, further comprising a repeating unit derived from.
- the repeating unit derived from the fluorine-containing monomer (a) is 25% by weight or more based on the fluorine-containing polymer, and the repeating unit derived from the crosslinkable monomer (d) is based on the fluorine-containing polymer.
- the dispersion liquid according to Item 9 which is 10% by weight or less.
- Item 11 Item 2.
- Item 12 Item 2.
- Item 13 A fluorine-containing monomer (a) containing a fluoroalkyl group and having a Q value of 2.0 or more in the Q-e scheme in a liquid medium, and chloride which is at least one selected from vinyl chloride and vinylidene chloride. 2.
- Step of reducing to 0 ppm or less (ii) A method for producing a dispersion liquid, including. [Item 14] A method for producing an object to be treated, which comprises a step of applying the dispersion liquid according to any one of Items 1 to 12 to a substrate.
- the dispersion liquid in the present disclosure is excellent in both product stability and water repellency and oil repellency.
- Dispersions (especially aqueous dispersions) It contains a fluorine-containing polymer and a liquid medium.
- the dispersion may further contain a surfactant and / or a curing agent.
- the dispersion may contain other components.
- Fluorine-containing polymer Fluorine-containing polymer Fluorine-containing monomer (a) Repeating unit derived from, and chloride monomer (b) Includes repeating units derived from.
- the fluorine-containing polymer further includes a hydrocarbon group-containing monomer (c) and / or a crosslinkable monomer (d). May include.
- the fluorine-containing polymer may contain another monomer (e).
- Fluorine-containing monomer (a) Fluorine-containing polymer Fluorine-containing monomer (a) having a Q value of 2.0 or more and containing a fluoroalkyl group. Includes repeating units derived from.
- the Q value of the fluorine-containing monomer (a) may be 2.0 or more, 2.2 or more, 2.4 or more, 2.6 or more or 2.8 or more, preferably 2.6 or more or 2 It is 0.8 or more.
- the Q value of the fluorine-containing monomer (a) may be 10.0 or less, 8.0 or less, 6.0 or more, 4.0 or less, 3.5 or less, or 3.0 or less, preferably 3.0 or less. It is 3.5 or less or 3.0 or less.
- the e value of the fluorine-containing monomer (a) may be 0.6 or more, 0.8 or more, 0.9 or more, or 1.0 or more, preferably 0.9 or more or 1.0 or more. be.
- the e value of the fluorine-containing monomer (a) may be 1.5 or less, 1.4 or less, 1.3 or less, 1.2 or less, or 1.1 or less, preferably 1.3 or less. It is 1.2 or less, or 1.1 or less.
- the Q value is an index showing the degree of resonance stabilizing effect between the double bond of the radically polymerizable monomer and its substituent.
- the e value of the monomer is an index indicating the degree of polarity of the double bond of the radically polymerizable monomer.
- the monomer reactivity ratio (r 1 , r 2 ) for estimating the composition of the copolymer is the formula [1] of Mayo-Lewis 1) as shown in the following formula. ], And then it is generally obtained from the equation [2] linearized by the Fineman-Ross method 2).
- each monomer M1, M2, ⁇ M1 ⁇ , ⁇ M2 ⁇ shows a radical, and the reaction rate constants k 11 , k 12 , k 22 of the four growth reactions, When k 21 is set, it is shown by the following formula.
- Equation [1] the equation [2] is derived by transforming it into the equation of Fineman-Ross method.
- R 1 and r 2 can be determined from the graph obtained by plotting F ⁇ (f-1) / f ⁇ against ⁇ F (2 / f) ⁇ and linearly approximating it. It is also possible to apply the non-linear least squares method 3) and calculate the combination of r 1 and r 2 by applying the solver function of the software so that the sum of ⁇ f 2 becomes the minimum value.
- the reactivity ratio (r 1 , r 2 ) of the monomer in the radical copolymerization is determined by the structure of the monomer and is not affected by temperature, pressure, solvent and the like. In general, the structural effect of the monomer on the relative velocity of the growth reaction can be expressed by the resonance effect and the polar effect when the steric effect is negligible. Alfrrey and Price hypothesized that the growth reaction rate constant k 12 of the radical copolymer represented by the formula [3] is represented by the formula [4] 5) .
- P 1 shows the general reactivity of M 1 ⁇
- Q 2 shows the resonance stabilizing effect of the monomer of M 2
- e 1 and e 2 show the polar effect of M 1 and M 2, respectively.
- Monomer reactivity ratio calculated from the copolymerization of styrene and various fluorinated monomers (r 1, r 2) of the formula [5] and the fluorine-containing monomer by substituting in Equation [6] (a ) Q value and e value can be calculated.
- the fluorine-containing monomer (a) is a fluorine-containing monomer containing a fluoroalkyl group.
- the fluorine-containing monomer (a) is generally a polymerizable compound having a perfluoroalkyl group or a perfluoroalkenyl group and a (meth) acryloyl group or an ⁇ -substituted acryloyl group.
- acrylic when not explicitly indicated, when simply referred to as "acrylic", not only the compound in which the ⁇ -position is a hydrogen atom but also the monovalent group in which the ⁇ -position is another group (for example, a methyl group) Also includes compounds substituted with (organic groups or halogen atoms) of.
- (meth) acryloyl means acryloyl or methacryloyl
- (meth) acrylate means acrylate or methacrylate
- (meth) acrylamide means acrylamide or methacrylamide.
- X is preferably a fluorine atom, a chlorine atom, a bromine atom or an iodine atom, and more preferably a chlorine atom.
- Y is preferably —O—.
- Z is a direct bond, an aliphatic group having 1 to 10 carbon atoms, an aromatic group having 6 to 18 carbon atoms, or a cyclic aliphatic group.
- Group represented by the formula -R 2 (R 1 ) N-SO 2- or -R 2 (R 1 ) N-CO- in the formula, R 1 is an alkyl group having 1 to 10 carbon atoms and R 2 Is a linear alkylene group or a branched alkylene group having 1 to 10 carbon atoms
- formula-CH 2 CH (OR 3 ) CH 2- (Ar-O) p- in the formula, R 3 is A group represented by a hydrogen atom or an acyl group having 1 to 10 carbon atoms (for example, formyl or acetyl)
- Ar is an arylene group having a substituent as required, and p represents 0 or 1), the formula- (CH).
- Rf is preferably a perfluoroalkyl group.
- the carbon number of Rf may be 1 to 12, 1 to 8, 1 to 6, 3 to 6, 4 to 6, or 6, preferably 3 to 6, more preferably 4 to 6, and particularly preferably 6. Is.
- Rf groups are -CF 3 , -CF 2 CF 3 , -CF 2 CF 2 CF 3 , -CF (CF 3 ) 2 , -CF 2 CF 2 CF 2 CF 3 , -CF 2 CF (CF 3).
- the monomer (a) include, but are not limited to, the following. These may be used alone or in combination of two or more.
- the fluorine-containing polymer contains a repeating unit derived from a chloride monomer (b), which is at least one selected from vinyl chloride and vinylidene chloride.
- the chloride monomer (b) is preferably vinyl chloride.
- a 21 is a hydrogen atom, a monovalent organic group or a halogen atom
- a 22 is a hydrocarbon group having 2 to 40 carbon atoms.
- Hydrocarbon group-containing monomer (c) represented by It may have repeating units derived from.
- a 21 is preferably a hydrogen atom, a methyl group or a chlorine atom.
- a 22 (hydrocarbon group) may be an acyclic aliphatic hydrocarbon group having 1 to 30 carbon atoms, a cyclic hydrocarbon group having 4 to 30 carbon atoms, or the like.
- the acyclic aliphatic hydrocarbon group preferably has 12 to 30 carbon atoms, more preferably 18 to 25 carbon atoms.
- Specific examples of acyclic aliphatic hydrocarbon groups are lauryl, cetyl, stearyl, and behenyl.
- Specific examples of the cyclic hydrocarbon group are a cyclohexyl group, a t-butylcyclohexyl group, an isobornyl group, a dicyclopentenyl group, a dicyclopentenyl group, and an adamantyl group.
- chain or cyclic hydrocarbon groups having 1 to 30 carbon atoms are linear or branched saturated or unsaturated (eg, ethylenically unsaturated) aliphatic hydrocarbon groups having 1 to 30 carbon atoms. It is a saturated or unsaturated (for example, ethylenically unsaturated) cyclic aliphatic hydrocarbon group of 4 to 30, an aromatic hydrocarbon group having 6 to 30 carbon atoms, and an aromatic aliphatic hydrocarbon group having 7 to 30 carbon atoms.
- the hydrocarbon group-containing monomer (c) does not have a polyfluoroalkyl group.
- the crosslinkable monomer (d) does not have to have a fluorine atom.
- a 22 fluorinated polymer in the formula (c) is a non-cyclic aliphatic hydrocarbon group having 12 to 30 carbon atoms long chain hydrocarbon group-containing monomer (c1) It is preferable to include repeating units derived from.
- the 12 to 30 acyclic aliphatic hydrocarbon groups may be linear or branched chain, preferably linear.
- long-chain hydrocarbon group-containing monomer (c1) examples include acyclic aliphatic carbides such as lauryl (meth) acrylate, cetyl (meth) acrylate, stearyl (meth) acrylate, and behenyl (meth) acrylate.
- examples thereof include an acrylate ester monomer having a hydrogen group. These may be used alone or in combination of two or more.
- Cyclic hydrocarbon group-containing monomer (c2) Cyclic hydrocarbon group-containing monomer (c2) in which A 22 is a cyclic hydrocarbon group in the formula (c). It is preferable to include repeating units derived from.
- Specific examples of the cyclic hydrocarbon group include a cyclohexyl group-containing group, a t-butylcyclohexyl group-containing group, an isobornyl group-containing group, a dicyclopentanyl group-containing group, a dicyclopentenyl group-containing group, and an adamantyl group-containing group. Be done.
- cyclic hydrocarbon group-containing monomer (c2) examples include cyclohexyl (meth) acrylate, t-butylcyclohexyl (meth) acrylate, benzyl (meth) acrylate, isobornyl (meth) acrylate, and dicyclopentanyl (meth).
- the fluorine-containing polymer is a crosslinkable monomer (d) having at least two selected from the group consisting of a reactive group and an olefinic carbon-carbon double bond. It may have repeating units derived from.
- the crosslinkable monomer (d) may be a compound having at least two ethylenically unsaturated double bonds, or a compound having at least one ethylenically unsaturated double bond and at least one reactive group.
- the crosslinkable monomer (d) preferably has a (meth) acrylate group or a (meth) acrylamide group. Examples of reactive groups are hydroxyl groups, epoxy groups, chloromethyl groups, blocked isocyanate groups, amino groups, carboxyl groups, and the like.
- the crosslinkable monomer (d) does not have a polyfluoroalkyl group.
- the crosslinkable monomer (d) does not have to have a fluorine atom.
- E 1 is a hydrogen atom, a methyl group or a halogen atom (for example, a chlorine atom, a bromine atom and an iodine atom).
- E 2 is -O- or -NH- and
- E 3 is an organic group having 1 to 20 carbon atoms, for example, a linear or branched aliphatic group having 1 to 20 carbon atoms (particularly an alkylene group), for example, the formula- (CH 2 ) x- (in the formula).
- X is a group represented by 1 to 10).
- E 4 is a hydroxyl group, an epoxy group, a chloromethyl group, a blocked isocyanate group, an amino group, and a carboxyl group.
- It may be a compound represented by.
- crosslinkable monomer examples include diacetone (meth) acrylamide, N-methylol (meth) acrylamide, hydroxyethyl (meth) acrylamide, glycidyl (meth) crylate, hydroxymethyl (meth) acrylate, and hydroxyethyl (meth).
- the fluorine-containing polymer is a monomer (e) other than the monomers (a) to (d). May contain repeating units derived from.
- the other monomer (b3) include, for example, ethylene, vinyl acetate, acrylonitrile, styrene, polyethylene glycol (meth) acrylate, polypropylene glycol (meth) acrylate, methoxypolyethylene glycol (meth) acrylate, and methoxypolypropylene glycol.
- (Meta) acrylate, vinyl alkyl ether and the like are included.
- Other non-fluorine monomers are not limited to these examples. These may be used alone or in combination of two or more.
- the amount of the repeating unit derived from the fluorine-containing monomer (a) may be 15% by weight or more, 25% by weight or more, 35% by weight or more, or 45% by weight or more with respect to the fluorine-containing polymer. ..
- the amount of the repeating unit derived from the fluorine-containing monomer (a) may be 90% by weight or less, for example, 80% by weight or less, 70% by weight or less, or 60% by weight or less with respect to the fluorine-containing polymer. ..
- the amount of the repeating unit derived from the chloride monomer (b) is 5% by weight or more, 10% by weight or more, 15% by weight or more, or 20% by weight or more, or 25% by weight, based on the fluorine-containing polymer. It may be% or more.
- the amount of the repeating unit derived from the chloride monomer (b) is 45% by weight or less, 40% by weight or less, 30% by weight or less, 25% by weight or less, or 22% by weight, based on the fluorine-containing polymer. It may be less than or equal to 25% by weight or less.
- the amount of the repeating unit derived from the hydrocarbon group-containing monomer (c) is 5% by weight or more, 10% by weight or more, 15% by weight or more, or 20% by weight or more with respect to the fluorine-containing polymer. It's okay.
- the amount of the repeating unit derived from the hydrocarbon group-containing monomer (c) is 45% by weight or less, 40% by weight or less, 30% by weight or less, 25% by weight or less, or 22 with respect to the fluorine-containing polymer. It may be less than or equal to% by weight.
- the amount of the repeating unit derived from the long-chain hydrocarbon group-containing monomer (c1) is 5% by weight or more, 10% by weight or more, 15% by weight or more, or 20% by weight or more with respect to the fluorine-containing polymer. May be.
- the amount of the repeating unit derived from the long-chain hydrocarbon group-containing monomer (c1) was 45% by weight or less, 40% by weight or less, 30% by weight or less, 25% by weight or less, based on the fluorine-containing polymer. Alternatively, it may be 22% by weight or less.
- the amount of the repeating unit derived from the crosslinkable monomer (d) is 0.5% by weight or more, 1% by weight or more, 3% by weight or more, or 4% by weight or more with respect to the fluorine-containing polymer. It's okay.
- the amount of the repeating unit derived from the crosslinkable monomer (d) is 15% by weight or less, 10% by weight or less, 7.5% by weight or less, or 5% by weight or less with respect to the fluorine-containing polymer. It's okay.
- the amount of the repeating unit derived from the other monomer (e) is 0.5% by weight or more, 1% by weight or more, 3% by weight or more, or 4% by weight or more with respect to the fluorine-containing polymer. It's okay.
- the amount of the repeating unit derived from the other monomer (e) may be 15% by weight or less, 10% by weight or less, or 5% by weight or less with respect to the fluorine-containing polymer.
- the amount of the repeating unit derived from the chloride monomer (b) is 5 to 500 parts by weight and 10 to 200 parts by weight with respect to 100 parts by weight of the repeating unit derived from the fluorine-containing monomer (a). It may be 10 to 150 parts by weight, 15 to 200 parts by weight, 20 to 150 parts by weight, or 10 to 50 parts by weight.
- the amount of the repeating unit derived from the hydrocarbon group-containing monomer (c) is 5 to 500 parts by weight and 10 to 200 parts by weight with respect to 100 parts by weight of the repeating unit derived from the fluorine-containing monomer (a). It may be 10 to 150 parts by weight, 15 to 200 parts by weight, or 20 to 150 parts by weight.
- the amount of the repeating unit derived from the long-chain hydrocarbon group-containing monomer (c1) is 5 to 500 parts by weight, 10 parts by weight, based on 100 parts by weight of the repeating unit derived from the fluorine-containing monomer (a). It may be up to 200 parts by weight, 10 to 150 parts by weight, 15 to 200 parts by weight, or 20 to 150 parts by weight.
- the amount of the repeating unit derived from the cyclic hydrocarbon group-containing monomer (c2) is 5 to 500 parts by weight and 10 to 500 parts by weight with respect to 100 parts by weight of the repeating unit derived from the fluorine-containing monomer (a). It may be 200 parts by weight, 10 to 150 parts by weight, 15 to 200 parts by weight, or 20 to 150 parts by weight.
- the amount of the repeating unit derived from the crosslinkable monomer (d) is 1 to 50 parts by weight and 3 to 40 parts by weight with respect to 100 parts by weight of the repeating unit derived from the fluorine-containing monomer (a). It may be 3 to 30 parts by weight, 5 to 25 parts by weight, or 5 to 20 parts by weight.
- the amount of the repeating unit derived from the other monomer (e) is 1 to 50 parts by weight and 3 to 40 parts by weight with respect to 100 parts by weight of the repeating unit derived from the fluorine-containing monomer (a). It may be 3 to 30 parts by weight, 5 to 25 parts by weight, or 5 to 20 parts by weight.
- the total of the repeating unit derived from the fluorine-containing monomer (a), the repeating unit derived from the chloride monomer (b), and the repeating unit derived from the hydrocarbon group-containing monomer (c) is , 85% by weight or more, 90% by weight or more, or 95% by weight or more with respect to the fluorine-containing polymer.
- a repeating unit derived from the fluorine-containing monomer (a), a repeating unit derived from the chloride monomer (b), a hydrocarbon group-containing monomer (c), and a hydrocarbon group-containing monomer ( The total of the repeating units derived from d) may be 85% by weight or more, 90% by weight or more, or 95% by weight or more with respect to the fluoropolymer.
- the unreacted chloride monomer (b) means at least one selected from vinyl chloride and vinylidene chloride.
- the dispersion contains a liquid medium.
- the liquid medium may be an aqueous medium.
- the liquid medium may be water alone, an organic solvent alone, or a mixture of water and a (miscible) organic solvent.
- the amount of water may be 30% by weight or more, 50% by weight or more, or 80% by weight or more with respect to the liquid medium.
- the amount of the organic solvent may be 30% by weight or less, for example, 10% by weight or less (preferably 0.1% or more) with respect to the liquid medium.
- the liquid medium may be added after the fluorine-containing polymer is produced by polymerization.
- the monomer is polymerized in the presence of an organic solvent to produce a fluorine-containing polymer, water is added, and then the organic solvent is distilled off.
- the organic solvent does not have to be distilled off.
- the dispersion liquid may contain a surfactant when it is an aqueous dispersion liquid.
- Surfactants include at least one of nonionic surfactants, cationic surfactants and anionic surfactants.
- the surfactant may include an amphoteric surfactant. In addition, it does not have to contain a surfactant.
- the dispersion liquid generally contains a surfactant when it is an aqueous dispersion liquid.
- the surfactant may or may not be added before or after the polymerization. Even when no surfactant is added, an aqueous dispersion in which the fluorine-containing polymer is dispersed in an aqueous medium can be obtained.
- the nonionic surfactant is a nonionic surfactant having an oxyalkylene group.
- the alkylene group in the oxyalkylene group preferably has 2 to 10 carbon atoms.
- the number of oxyalkylene groups in the molecule of the nonionic surfactant is generally preferably 2 to 100.
- Nonionic surfactants are linear and / or branched aliphatic (saturated and / or unsaturated) group alkylene oxide adducts, linear and / or branched fatty acids (saturated and / or unsaturated).
- Polyalkylene glycol ester polyoxyethylene (POE) / polyoxypropylene (POP) copolymer (random copolymer or block copolymer), alkylene oxide adduct of acetylene glycol and the like may be used.
- the structure of the alkylene oxide addition moiety and the polyalkylene glycol moiety is polyoxyethylene (POE) or polyoxypropylene (POP) or POE / POP copolymer (block copolymer even if it is a random copolymer). It may be).
- the nonionic surfactant preferably has a structure that does not contain an aromatic group due to environmental problems (biodegradability, environmental hormones, etc.).
- the cationic surfactant may be an amine salt, a quaternary ammonium salt, or an oxyethylene-added ammonium salt.
- Specific examples of the cationic surfactant are not particularly limited, but are alkylamine salts, aminoalcohol fatty acid derivatives, polyamine fatty acid derivatives, amine salt-type surfactants such as imidazoline, alkyltrimethylammonium salts, dialkyldimethylammonium salts, and the like. Examples thereof include quaternary ammonium salt-type surfactants such as alkyldimethylbenzylammonium salt, pyridinium salt, alkylisoquinolinium salt and benzethonium chloride.
- cationic surfactants include dodecyltrimethylammonium acetate, trimethyltetradecylammonium chloride, hexadecyltrimethylammonium bromide, trimethyloctadecylammonium chloride, (dodecylmethylbenzyl) trimethylammonium chloride, benzyldodecyldimethylammonium chloride, and methyldodecyl.
- Examples thereof include di (hydropolyoxyethylene) ammonium chloride, benzyldodecyldi (hydropolyoxyethylene) ammonium chloride, and N- [2- (diethylamino) ethyl] oleamide hydrochloride.
- anionic surfactants include fatty acid salts (fatty acid has, for example, 8 to 30 carbon atoms), sulfonates (for example, alkyl sulfonic acids, alkylbenzene sulfonates (alkyl groups have, for example, 8 to 30 carbon atoms). 30.)), Sulfate ester salt (for example, alkyl sulfate ester salt (the number of carbon atoms of the alkyl group is, for example, 8 to 30)).
- fatty acid salts fatty acid has, for example, 8 to 30 carbon atoms
- sulfonates for example, alkyl sulfonic acids, alkylbenzene sulfonates (alkyl groups have, for example, 8 to 30 carbon atoms). 30.
- Sulfate ester salt for example, alkyl sulfate ester salt (the number of carbon atoms of the alkyl group is, for example, 8 to 30)
- anionic surfactants are sodium lauryl sulfate, triethanolamine lauryl sulfate, sodium polyoxyethylene lauryl ether sulfate, sodium polyoxyethylene nonylphenyl ether sulfate, triethanolamine polyoxyethylene lauryl ether sulfate, sodium cocoyl sarcosin, Sodium N-cocoyl methyl taurine, sodium polyoxyethylene palm alkyl ether sulfate, sodium dietherhexyl sulfosuccinate, sodium ⁇ -olefin sulfonate, sodium lauryl phosphate, sodium polyoxyethylene lauryl ether phosphate and the like are included.
- amphoteric surfactant examples include alanines, imidazolinium betaines, amide betaines, betaine acetate and the like, and specific examples thereof include lauryl betaine, stearyl betaine, laurylcarboxymethyl hydroxyethyl imidazolinium betaine and lauryl dimethyl. Examples thereof include betaine aminoacetate and betaine fatty acid amide propyldimethylaminoacetate.
- Each of the nonionic surfactant, the cationic surfactant, the anionic surfactant, and the amphoteric surfactant may be one kind or a combination of two or more.
- the surfactant is preferably a nonionic surfactant and / or a cationic surfactant. It may be a combination of a nonionic surfactant and a cationic surfactant.
- the dispersion liquid may contain a curing agent (active hydrogen reactive compound or active hydrogen-containing compound). After polymerizing the fluorine-containing polymer, a curing agent may be added to the dispersion liquid.
- a curing agent active hydrogen reactive compound or active hydrogen-containing compound.
- the dispersion liquid may contain a curing agent (crosslinking agent) so as to cure the fluorine-containing polymer satisfactorily.
- a curing agent crosslinking agent
- the non-fluorine crosslinkable (meth) acrylate or (meth) acrylamide monomer is an active hydrogen-containing monomer or an active hydrogen-reactive group-containing monomer
- the fluorine-containing polymer is active hydrogen or active hydrogen-reactive.
- the curing agent is an active hydrogen-reactive compound or an active hydrogen-containing compound so as to react with the active hydrogen or the active hydrogen-reactive group of the fluorine-containing polymer.
- active hydrogen reactive compounds are polyisocyanate compounds, epoxy compounds, chloromethyl group-containing compounds, carboxyl group-containing compounds and hydrazide compounds.
- active hydrogen-containing compounds are hydroxyl group-containing compounds, amino group-containing compounds and carboxyl group-containing compounds, ketone group-containing compounds, hydrazide compounds and melamine compounds.
- the curing agent is preferably a polyisocyanate compound.
- a polyisocyanate compound is a compound having two or more isocyanate groups in one molecule.
- the polyisocyanate compound acts as a cross-linking agent.
- Examples of the polyisocyanate compound include aliphatic polyisocyanates, alicyclic polyisocyanates, aromatic aliphatic polyisocyanates, aromatic polyisocyanates, and derivatives of these polyisocyanates.
- aliphatic polyisocyanates examples include trimethylene diisocyanate, tetramethylene diisocyanate, hexamethylene diisocyanate, pentamethylene diisocyanate, 1,2-propylene diisocyanate, 1,2-butylene diisocyanate, 2,3-butylene diisocyanate, and 1,3-butylene.
- alicyclic polyisocyanates are alicyclic diisocyanates and alicyclic triisocyanates. Specific examples of the alicyclic polyisocyanate are 1,3-cyclopentene diisocyanate, 3-isocyanatomethyl-3,5,5-trimethylcyclohexylisocyanate (isophorone diisocyanate), and 1,3,5-triisocyanatocyclohexane.
- aromatic aliphatic polyisocyanates are aromatic aliphatic diisocyanates and aromatic aliphatic triisocyanates.
- aromatic aliphatic polyisocyanates include 1,3- or 1,4-xylylene diisocyanate or a mixture thereof, 1,3- or 1,4-bis (1-isocyanato-1-methylethyl) benzene (tetramethyl).
- aromatic polyisocyanates are aromatic diisocyanates, aromatic triisocyanates, and aromatic tetraisocyanates.
- aromatic polyisocyanates include m-phenylene diisocyanate, p-phenylene diisocyanate, 4,4'-diphenyl diisocyanate, 1,5-naphthalene diisocyanate, 2,4'-or 4,4'-diphenylmethane diisocyanate or a mixture thereof, 2,4- or 2,6 -Toluene diisocyanate or a mixture thereof, triphenylmethane-4,4', 4 "-triisocyanate, and 4,4'-diphenylmethane-2,2', 5,5'-tetraisocyanate and the like.
- polyisocyanate derivative examples include various derivatives such as dimer, trimmer, biuret, allophanate, carbodiimide, uretdione, uretoimine, isocyanurate, and iminooxadiazinedione of the above-mentioned polyisocyanate compound.
- polyisocyanates can be used alone or in combination of two or more.
- a blocked polyisocyanate compound blocked isocyanate
- a blocked polyisocyanate compound which is a compound in which the isocyanate group of the polyisocyanate compound is blocked with a blocking agent. It is preferable to use a blocked polyisocyanate compound because it is relatively stable even in an aqueous solution and can be used in the same aqueous solution as the dispersion.
- the blocking agent blocks free isocyanate groups.
- the blocked polyisocyanate compound is heated to, for example, 100 ° C. or higher, for example, 130 ° C. or higher, the isocyanate group is regenerated and can easily react with the hydroxyl group.
- blocking agents are phenolic compounds, lactam compounds, fatty alcohol compounds, oxime compounds and the like.
- the polyisocyanate compound can be used alone or in combination of two or more.
- the epoxy compound is a compound having an epoxy group.
- epoxy compounds are epoxy compounds having a polyoxyalkylene group, such as polyglycerol polyglycidyl ether and polypropylene glycol diglycidyl ether; and sorbitol polyglycidyl ether.
- a chloromethyl group-containing compound is a compound having a chloromethyl group. Examples of chloromethyl group-containing compounds are chloromethyl polystyrene and the like.
- the carboxyl group-containing compound is a compound having a carboxyl group. Examples of carboxyl group-containing compounds are (poly) acrylic acid, (poly) methacrylic acid and the like.
- ketone group-containing compound examples include (poly) diacetone acrylamide and diacetone alcohol.
- hydrazide compound examples include hydrazine, carbhydrazide, adipic acid hydrazide and the like.
- melamine compound examples include a melamine resin and a methyl etherified melamine resin.
- the dispersion liquid may contain components other than the above components. Generally, other components are added after the fluorine-containing polymer is produced. Examples of other components are non-fluorine water repellent and / or oil repellent compounds.
- the dispersion may contain a water-repellent and / or oil-repellent compound (non-fluorine water-repellent and / or oil-repellent compound) that does not contain a fluorine atom.
- the non-fluorine water-repellent and / or oil-repellent compound may be a non-fluorine acrylate polymer, a saturated or unsaturated hydrocarbon compound, or a silicone-based compound.
- the non-fluorinated acrylate polymer is a homopolymer composed of one kind of non-fluorinated acrylate monomer, a copolymer composed of at least two kinds of non-fluorinated acrylate monomers, or at least one kind of non-fluorinated polymer. It is a copolymer composed of an acrylate monomer and at least one other non-fluorinated monomer (ethylene unsaturated compound, for example, ethylene or vinyl-based monomer).
- A is a halogen atom other than a hydrogen atom, a methyl group or a fluorine atom (for example, a chlorine atom, a bromine atom and an iodine atom).
- T is a hydrogen atom, a chain or cyclic hydrocarbon group having 1 to 30 carbon atoms, or a chain or cyclic organic group having 1 to 31 carbon atoms having an ester bond.
- chain or cyclic hydrocarbon groups having 1 to 30 carbon atoms are linear or branched aliphatic hydrocarbon groups having 1 to 30 carbon atoms, cyclic aliphatic group groups having 4 to 30 carbon atoms, and 6 to 30 carbon atoms. It is an aromatic hydrocarbon group having 30 carbon atoms and an aromatic aliphatic hydrocarbon group having 7 to 30 carbon atoms.
- non-fluorinated acrylate monomers include, for example, alkyl (meth) acrylate, polyethylene glycol (meth) acrylate, polypropylene glycol (meth) acrylate, methoxypolyethylene glycol (meth) acrylate, methoxypolypropylene glycol (meth) acrylate. Is done.
- the non-fluorine acrylate monomer is preferably an alkyl (meth) acrylate.
- the number of carbon atoms of the alkyl group may be 1 to 30, for example, 6 to 30 (for example, 10 to 30).
- Specific examples of the non-fluorine acrylate monomer are lauryl (meth) acrylate, stearyl (meth) acrylate and behenyl (meth) acrylate.
- the non-fluorine acrylate polymer can be produced by the same polymerization method as the fluorine-containing polymer.
- the saturated or unsaturated hydrocarbon compound is preferably a saturated hydrocarbon.
- the number of carbon atoms may be 15 or more, preferably 20 to 300, for example 25 to 100.
- Specific examples of saturated or unsaturated hydrocarbon compounds are paraffin and the like.
- Silicone compounds are generally used as surface treatment agents (for example, water repellents).
- the silicone-based compound is preferably a compound exhibiting water repellency and / or oil repellency.
- the amount of the fluorine-containing polymer (solid content) is about 0.01 to 60% by weight, preferably about 0.1 to 40% by weight, more preferably about 5 to 35% by weight, based on the dispersion liquid. It may be there. For example, at the time of storage, it may be stored at a high concentration, and when it is used, a liquid medium may be added as needed to dilute it to an arbitrary concentration before use. By improving product stability, it may be possible to supply high-concentration products that have had problems in supply in terms of conventional stability.
- the concentration of unreacted chloride monomer in the dispersion is 2.0 ppm or less, 1.5 ppm or less, 1.0 ppm or less, 0.8 ppm or less, 0.5 ppm or less, 0.3 ppm or less, or 0.1 ppm. It may be:
- the amount of the liquid medium may be 30 to 99.9% by weight, particularly 50 to 99% by weight, based on the dispersion liquid.
- Amount of surfactant In the present disclosure, a dispersion liquid of a fluorine-containing polymer, particularly an aqueous dispersion liquid, can be formed even when a surfactant is not used.
- the amount of the surfactant may be 0.1 to 50 parts by weight, for example, 1 to 30 parts by weight, based on 100 parts by weight of the fluorine-containing polymer (or the total amount of the monomers).
- the amount of the curing agent may be 100 parts by weight or less, for example, 0.01 to 30 parts by weight, based on 100 parts by weight of the fluorine-containing polymer.
- the amount of the non-fluorine water-repellent and / or oil-repellent compound may be 500 parts by weight or less, for example, 5 to 200 parts by weight, particularly 5 to 100 parts by weight, based on 100 parts by weight of the fluorine-containing polymer.
- the method for producing the dispersion liquid in the present disclosure is as follows.
- the fluorine-containing polymer in the present disclosure can be produced by any of the usual polymerization methods, and the conditions of the polymerization reaction can be arbitrarily selected. Examples of such a polymerization method include solution polymerization, suspension polymerization, and emulsion polymerization.
- solution polymerization a method in which a monomer is dissolved in an organic solvent in the presence of a polymerization initiator, and after nitrogen substitution, is heated and stirred in a range of 30 to 120 ° C. for 30 minutes to 48 hours, for example, 3 to 24 hours.
- the polymerization initiator include azobisisobutyronitrile, benzoyl peroxide, di-t-butyl peroxide, lauryl peroxide, cumene hydroperoxide, t-butyl peroxypivalate, diisopropyl peroxy dicarbonate and the like.
- the polymerization initiator is used in the range of 0.01 to 20 parts by weight, for example, 0.01 to 10 parts by weight with respect to 100 parts by weight of the monomer.
- the organic solvent is inert to the monomer and dissolves them, for example, an ester (for example, an ester having 2 to 30 carbon atoms, specifically, ethyl acetate, butyl acetate), a ketone (for example, carbon). It may be a ketone having the number 2 to 30, specifically, a methyl ethyl ketone or a diisobutyl ketone), or an alcohol (for example, an alcohol having 1 to 30 carbon atoms, specifically, an isopropyl alcohol).
- an ester for example, an ester having 2 to 30 carbon atoms, specifically, ethyl acetate, butyl acetate
- a ketone for example, carbon
- It may be a ketone having the number 2 to 30, specifically, a methyl ethyl ketone or a diisobutyl ketone
- an alcohol for example, an alcohol having 1 to 30 carbon atoms, specifically, an isopropyl alcohol
- organic solvent examples include acetone, chloroform, HCHC225, isopropyl alcohol, pentane, hexane, heptane, octane, cyclohexane, benzene, toluene, xylene, petroleum ether, tetrahydrofuran, 1,4-dioxane, methyl ethyl ketone, methyl isobutyl ketone, and the like.
- Examples thereof include diisobutyl ketone, ethyl acetate, butyl acetate, 1,1,2,2-tetrachloroethane, 1,1,1-trichloroethane, trichloroethylene, perchloroethylene, tetrachlorodifluoroethane, and trichlorotrifluoroethane.
- the organic solvent is used in the range of 10 to 2000 parts by weight, for example, 50 to 1000 parts by weight, based on 100 parts by weight of the monomer in total.
- a monomer is emulsified in water in the presence of a polymerization initiator and an emulsifier, and after nitrogen substitution, the monomer is stirred and polymerized in the range of 50 to 80 ° C. for 30 minutes to 48 hours, for example, 3 to 24 hours.
- the method of making it is adopted.
- the polymerization initiators are benzoyl peroxide, lauroyl peroxide, t-butyl perbenzoate, 1-hydroxycyclohexylhydro peroxide, 3-carboxypropionyl peroxide, acetyl peroxide, azobisisobutylamidin-dihydrochloride, azo.
- Water-soluble substances such as bisisobutyronitrile, sodium peroxide, potassium persulfate, ammonium persulfate, azobisisobutyronitrile, benzoyl peroxide, di-t-butyl peroxide, lauryl peroxide, cumene hydroperoxide , T-Butylperoxypivalate, diisopropylperoxydicarbonate and other oil-soluble materials are used.
- the polymerization initiator is used in the range of 0.01 to 10 parts by weight with respect to 100 parts by weight of the monomer.
- the monomer is atomized and polymerized in water using an emulsifying device such as a high-pressure homogenizer or an ultrasonic homogenizer that can apply strong crushing energy.
- an emulsifying device such as a high-pressure homogenizer or an ultrasonic homogenizer that can apply strong crushing energy.
- the emulsifier various anionic, cationic or nonionic emulsifiers can be used, and they are used in the range of 0.5 to 20 parts by weight with respect to 100 parts by weight of the monomer. It is preferred to use anionic and / or nonionic and / or cationic emulsifiers.
- compatibilizer that is sufficiently compatible with these monomers, for example, a water-soluble organic solvent or a low molecular weight monomer. It is possible to improve emulsifying property and copolymerizability by adding a compatibilizer.
- water-soluble organic solvent examples include acetone, methyl ethyl ketone, ethyl acetate, propylene glycol, dipropylene glycol monomethyl ether, dipropylene glycol, tripropylene glycol, ethanol and the like, and 1 to 50 parts by weight with respect to 100 parts by weight of water.
- the water-soluble organic solvent examples include acetone, methyl ethyl ketone, ethyl acetate, propylene glycol, dipropylene glycol monomethyl ether, dipropylene glycol, tripropylene glycol, ethanol and the like, and 1 to 50 parts by weight with respect to 100 parts by weight of water.
- the low molecular weight monomer examples include methyl methacrylate, glycidyl methacrylate, 2,2,2-trifluoroethyl methacrylate and the like, and 1 to 50 parts by weight, based on 100 parts by weight of the total amount of the monomer.
- it may be used in the
- a chain transfer agent may be used.
- the molecular weight of the polymer can be changed according to the amount of the chain transfer agent used.
- chain transfer agents are mercaptan group-containing compounds such as lauryl mercaptan, thioglycol, thioglycerol (particularly alkyl mercaptans (eg, 1-30 carbon atoms)), inorganic salts such as sodium hypophosphite, sodium bisulfite. And so on.
- the amount of the chain transfer agent used may be in the range of 0.01 to 10 parts by weight, for example, 0.1 to 5 parts by weight, based on 100 parts by weight of the total amount of the monomers.
- the fluorine-containing polymer by an emulsion polymerization method or a solution polymerization method.
- water or an aqueous medium
- water may be added after the fluorine-containing polymer is produced by polymerization.
- water is added to the polymer mixture to distill off the organic solvent, and the fluorine-containing polymer is made into water. It may be dispersed.
- the organic solvent does not have to be distilled off.
- the surfactant may or may not be added before or after the polymerization.
- An aqueous dispersion can be obtained even when no surfactant is added.
- the curing agent may be added before or after the polymerization, but it is preferably added after the polymerization. Other components may or may not be added before or after polymerization.
- chloride monomer removal step (ii) Examples of methods for removing unreacted vinyl chloride or vinylidene chloride from the polymerization solution after the polymerization reaction include distillation under reduced pressure, distillation by stirring under normal pressure heating, and air under heating or non-heating. , Nitrogen, steam bubbling (eg, 100 cc / min or more, 200 cc / min or more, 300 cc / min or more, or 400 cc / min or more, 3 hours or more, 6 hours or more, 12 hours or more, or 18 hours or more) Examples include a method using a filling tower, a spin coater, and a cylindrical volatilizer. The temperature, time, airflow intensity, etc. during the removal process can be changed as appropriate.
- the chloride monomer (b) may be removed while leaving a part of the liquid medium in the polymerization solution.
- the dispersion liquid of the present disclosure can be applied to an object to be treated as a surface treatment agent by a conventionally known method.
- a method is adopted in which the dispersion liquid is dispersed in an organic solvent or water, diluted, adhered to the surface of the object to be treated by a known method such as dip coating, spray coating, foam coating, etc., and dried. .. If necessary, it may be applied together with a suitable cross-linking agent for curing.
- an insect repellent, a softener, an antibacterial agent, a flame retardant, an antistatic agent, a paint fixing agent, an anti-wrinkle agent and the like may be 0.01 to 10% by weight (particularly in the case of dip coating), for example, 0.05 to 10% by weight.
- Examples of the object to be treated with the surface treatment agent (for example, water repellent, oil repellent, water repellent and oil repellent) of the present disclosure include textile products, stone materials, filters (for example, electrostatic filters), dustproof masks, and fuel cells. Parts (eg, gas diffusion electrodes and gas diffusion supports), glass, paper, wood, leather, fur, asbestos, bricks, cement, metals and oxides, ceramic products, plastics, coated surfaces, plasters, etc. can. Various examples can be given as textile products.
- animal and vegetable natural fibers such as cotton, hemp, wool and silk
- synthetic fibers such as polyamide, polyester, polyvinyl alcohol, polyacrylonitrile, polyvinyl chloride and polypropylene
- semi-synthetic fibers such as rayon and acetate, glass fibers and carbon fibers.
- Inorganic fibers such as asbestos fibers, or mixed fibers thereof.
- the textile product may be in any form such as fiber or cloth.
- the dispersion liquid of the present disclosure can also be used as an internal release agent or an external release agent.
- the polymer can be applied to textiles by any of the methods known for treating textiles (eg, cloth) with liquids.
- the textile may be immersed in the solution, or the solution may be attached or sprayed onto the textile.
- the treated textile product is preferably dried and cured by heating in order to exhibit water repellency and oil repellency.
- the heating temperature may be, for example, 100 ° C. to 200 ° C., 100 ° C. to 170 ° C., or 100 ° C. to 120 ° C. In the present disclosure, good performance can be obtained even with low temperature heating (for example, 100 ° C. to 140 ° C.).
- the heating time may be 5 seconds to 60 minutes, for example, 30 seconds to 3 minutes.
- the polymer may be applied to textile products by a cleaning method, and may be applied to textile products by, for example, a washing application or a dry cleaning method.
- the textile products to be treated are typically fabrics, including textiles (woven fabrics), knitted fabrics (knitted fabrics) and non-woven fabrics, textiles and carpets in the form of clothing, but fibers or threads or It may be an intermediate textile product (eg, sliver or blister).
- the dispersions of the present disclosure are particularly effective in making textile products (eg, synthetic fibers) water and / or oil repellent.
- the fibers constituting the textile product may be natural fibers, synthetic fibers, semi-synthetic fibers, regenerated fibers or inorganic fibers.
- the fiber may be used alone or in combination of two or more.
- wood pulp examples include mechanical pulp such as ground wood pulp (GP), pressure-rise ground wood pulp (PGW), thermomechanical pulp (TMP), coniferous tree high yield unbleached kraft pulp (HNKP; N material), and coniferous tree.
- GP ground wood pulp
- PGW pressure-rise ground wood pulp
- TMP thermomechanical pulp
- HNKP coniferous tree high yield unbleached kraft pulp
- NKP Bleached kraft pulp
- LKP broad-leaved unbleached kraft pulp
- LKP broad-leaved bleached kraft pulp
- other chemical pulp Dinking pulp (DIP), waste pulp (DIP), waste pulp (L material) Waste paper pulp such as WP) and semi-chemical pulp (CP).
- polyesters such as polyethylene terephthalate, polybutylene terephthalate, polytrimethylene terephthalate, and copolymerized polyester; polyolefins such as linear low-density polyethylene, low-density polyethylene, high-density polyethylene, and polypropylene; nylon 6, nylon 66. , Polyamides such as nylon 610 and nylon 46; acrylic fibers such as polyacrylonitrile; polyvinyl alcohol, polyurethane and polyvinyl chloride.
- semi-synthetic fibers are acetate and triacetate. Examples of regenerated fibers are rayon, cupra, polynosic rayon, lyocell and tencel.
- inorganic fibers are glass fibers and carbon fibers.
- the textile product may be leather.
- the production polymer is aqueous or aqueous emulsified at various stages of leather processing, for example during the wet processing of the leather, or during the finishing of the leather, in order to make the leather hydrophobic and oleophobic. It may be applied from things to leather.
- the textile product may be paper. The production polymer may be applied to preformed paper or at various stages of papermaking, for example during the drying period of the paper.
- Treatment means applying the dispersion to the object to be treated by dipping, spraying, coating, or the like.
- the polymer which is the active ingredient of the dispersion liquid permeates into the inside of the object to be treated and / or adheres to the surface of the object to be treated.
- the reactivity ratio (r 1 , r 2 ) of the fluorine-containing monomer with styrene was determined as follows. Copolymerization is carried out by adding ethyl acetate (25 g), C8FA and styrene to a 100 ml four-necked flask so that the monomer charging ratio is 1/9 to 9/1 (total number of moles is 0.04 mol). I prepared it. The mixture was heated to 60 ° C. under a nitrogen atmosphere with stirring. Next, 2,2'-bis 2,4-hydroxybutyrate (0.5 mol% of the charged monomer) was added as a polymerization initiator.
- the copolymerization was adjusted so that the monomer conversion rate was 10% or less.
- 10 times the amount of methanol was added to the polymerization solution, and the produced precipitate was centrifuged. It was rewashed with the same volume of methanol and separated by the same method. Filter the product. It was dried at 130 ° C. for 1 hour.
- the emulsified dispersion was transferred to a 500 ml autoclave, and after nitrogen substitution for 15 minutes, 0.2 g of lauryl mercaptan and 32 g of vinyl chloride were charged. Further, 1 g of an azo group-containing water-soluble initiator was added, the temperature was raised at 60 ° C., and the mixture was reacted for 4 hours to obtain an aqueous dispersion of the polymer. The conversion rate of the monomer by gas chromatography was 99.0 to 99.8%. This dispersion was further diluted with pure water to prepare an aqueous dispersion having a solid content concentration of 30% by weight.
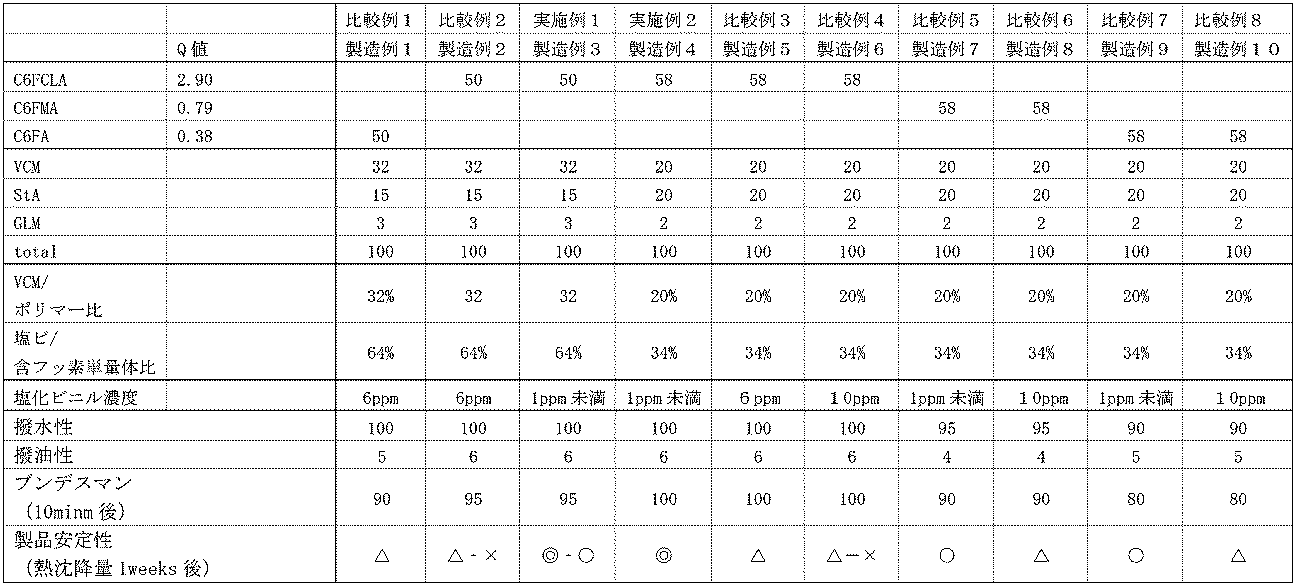
- Example 1 33.3 g of the aqueous dispersion containing the fluorine-containing polymer having a solid content concentration of 30% by weight prepared in Production Example 3 was diluted with tap water to prepare 1000 g of a test solution having a solid content concentration of 1.0%. After immersing the polyester cloth and the nylon cloth in this test solution, the cloth was squeezed with a mangle. Each dough was passed through a pin tenter at 160 ° C. for 1 minute, dried and cured. The fabric treated in this manner was subjected to a water repellency test, an oil repellency test, a Congress test, and a stability evaluation test by a spray method of JIS L-1092. The results are shown in Table 6.
- Example 2 The aqueous dispersion prepared in Production Example 4 containing the fluorine-containing polymer having a solid content concentration of 30% was evaluated in the same manner as in Example 1 with respect to the treatment liquid containing a fixed amount of vinyl chloride monomer. The results are shown in Table 6.
- the dispersion liquid of the present disclosure can be used to impart water repellency and / or oil repellency to various products.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Polymers & Plastics (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Textile Engineering (AREA)
- Materials Engineering (AREA)
- Wood Science & Technology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Addition Polymer Or Copolymer, Post-Treatments, Or Chemical Modifications (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Treatments For Attaching Organic Compounds To Fibrous Goods (AREA)
- Lubricants (AREA)
Abstract
Description
[項1]
含フッ素重合体及び液状媒体を含む、分散液であって、
含フッ素重合体が
Q値が2.0以上であって、フルオロアルキル基を含有する含フッ素単量体(a)
から誘導される繰り返し単位、及び
塩化ビニル及び塩化ビニリデンから選択される少なくとも一種である塩化物単量体(b)
から誘導される繰り返し単位
を含み、未反応の塩化物単量体(b)の濃度が2.0ppm以下である、分散液。
[項2]
含フッ素単量体(a)が、式:
CH2=C(-X)-C(=O)-Y-Z-Rf
[式中、Xはハロゲン原子であり、
Yは-O-又は-NH-であり、
Zは直接結合又は二価の有機基であり、
Rfは炭素数1~20のフルオロアルキル基である。]
で示される化合物である、項1に記載の分散液。
[項3]
Xが塩素原子である、項2に記載の分散液。
[項4]
Rfが炭素数3~6のパーフルオロアルキル基である項2又は3に記載の分散液。
[項5]
塩化物単量体(b)から誘導される繰り返し単位の量が、含フッ素重合体に対して25重量%未満である、項1~4のいずれか一項に記載の分散液。
[項6]
含フッ素重合体が
式:
CH2=CA21-C(=O)-O-A22
[式中、A21は水素原子、一価の有機基又はハロゲン原子であり、A22は炭素数2~40の炭化水素基である。]
で示される炭化水素基含有単量体(c)
から誘導された繰り返し単位をさらに含む、項1~5のいずれか一項に記載の分散液。
[項7]
含フッ素重合体が
A22は炭素数12~30の非環式の脂肪族炭化水素基である長鎖炭化水素基含有単量体(c1)
から誘導された繰り返し単位を含む、項6に記載の分散液。
[項8]
含フッ素単量体(a)から誘導される繰り返し単位、塩化物単量体(b)から誘導される繰り返し単位、及び炭化水素基含有単量体(c)から誘導される繰り返し単位の合計が、含フッ素重合体に対して90重量%以上である、項6又は7に記載の分散液。
[項9]
含フッ素重合体が
反応性基及びオレフィン性炭素-炭素二重結合からなる群から選択される少なくとも2つを有する架橋性単量体(d)
から誘導された繰り返し単位をさらに含む、項1~8のいずれか一項に記載の分散液。
[項10]
含フッ素単量体(a)から誘導される繰り返し単位が含フッ素重合体に対して25重量%以上であり、架橋性単量体(d)から誘導される繰り返し単位が含フッ素重合体に対して10重量%以下である、項9に記載の分散液。
[項11]
未反応の塩化物単量体(b)の濃度が1.0ppm以下である、項1~10のいずれか一項に記載の分散液。
[項12]
液状媒体が少なくとも30重量%以上の水を含む、項1~11のいずれか一項に記載の分散液。
[項13]
液状媒体中、フルオロアルキル基を含有し、Q-eスキームにおけるQ値が2.0以上である含フッ素単量体(a)、及び塩化ビニル及び塩化ビニリデンから選択される少なくとも一種である塩化物単量体(b)を共重合して含フッ素重合体を含む重合液を得る工程(i)、及び
得られた重合液中の未反応の塩化物単量体(b)の濃度を2.0ppm以下まで低減させる工程(ii)
を含む、分散液の製造方法。
[項14]
項1~12のいずれかに記載の分散液を基材に適用する工程を含む、被処理物の製造方法。
分散液(特に水分散液)は、
含フッ素重合体、及び
液状媒体
を含んでなる。分散液は、さらに
界面活性剤、及び/又は
硬化剤
を含んでよい。分散液は、他の成分を含んでもよい。
含フッ素重合体は、
含フッ素単量体(a)
から誘導される繰り返し単位、及び
塩化物単量体(b)
から誘導される繰り返し単位を含む。含フッ素重合体は、さらに
炭化水素基含有単量体(c)、及び/又は
架橋性単量体(d)
を含んでよい。含フッ素重合体は、他の単量体(e)を含んでもよい。
含フッ素重合体は、
Q値が2.0以上であって、フルオロアルキル基を含有する含フッ素単量体(a)
から誘導された繰り返し単位を含む。
1)単量体反応性比の算出
一般的に共重合体の組成を見積もる単量体の反応性比(r1、r2)は下式に示すようにMayo-Lewis1)の式[1]より求め、次にFineman-Ross法2)により線形化した式[2]より求めることが一般的である。2種の単量体を共重合させた場合、各単量体M1、M2、~M1・、~M2・はラジカルを示し、4つの成長反応の反応速度定数k11、k12、k22、k21と置くと下式で示される。
単量体の仕込み量を[M1]、[M2]、単量体の減少量をd[M1]、d[M2]と置くと下式となる。
式[1]において
とおき、Fineman-Ross法の式に変形すると[2]式が導かれる。
F{(f-1)/f}を、{F(2/f)}に対してプロットして直線近似することによって得られるグラフより、r1及びr2を決定することができる。非線形最小自乗法3)を適用しΔf2の総和が最小値となるようにr1及びr2の組み合わせを、ソフトウェアのソルバー機能を適用して算出することもできる。
ラジカル共重合における単量体の反応性比(r1、r2)は単量体の構造によって決まり、温度、圧力、溶媒などの影響は受けないとされている。一般的に生長反応の相対速度に及ぼす単量体の構造効果は、立体効果が無視できる場合、共鳴効果と極性効果によって表わすことができる。AlfrreyとPriceは式[3]で示されるラジカル共重合の生長反応速度定数k12が式[4]で表されると仮定した5)。
ここでP1はM1・の一般反応性、Q2はM2の単量体の共鳴安定化効果、e1、e2はそれぞれM1、M2の極性効果を示す。式[4]をラジカル共重合の単量体反応比r1=k11/k12、r2=k22/k21に代入すると式[5]と式[6]が得られる。
スチレンを基準単量体としてQ値を1.0、e値を-0.8とする。単量体とそのラジカルのe値を等しいとする。スチレンと各種含フッ素単量体の共重合より算出される単量体反応性比(r1、r2)を式[5]と式[6]に代入することにより含フッ素単量体(a)のQ値、e値を算出できる。
1)F. R. Mayo, F. M. Lewis, J. Am. Chem. Soc.,1944, 66, 1594.
2)M. Fineman and S. D. Ross, J. Polym. Sci., 1950, 5, 259.
3)P. W. Tidwell, G. A. Mortimer, J. Polymer. Sci., 1965, A3, 369.
4)T. Otsu, in Progress in Polymer Science Japan, ed. by M. Imoto, S. Onogi,
Kodansha Ltd., Tokyo, 1970, Vol.1, PP.4
5)T. Alfrey Jr., C. C. Price, J. Polym. Sci., 1947, 2. 101.
CH2=C(-X)-C(=O)-Y-Z-Rf
[式中、Xはハロゲン原子であり、
Yは-O-又は-NH-であり、
Zは直接結合又は二価の有機基であり、
Rfは炭素数1~20のフルオロアルキル基である。]
で示される化合物であることが好ましい。
式-R2(R1)N-SO2-もしくは-R2(R1)N-CO-で示される基(式中、R1は、炭素数1~10のアルキル基であり、R2は、炭素数1~10の直鎖アルキレン基又は分枝状アルキレン基である。)、式-CH2CH(OR3)CH2-(Ar-O)p-(式中、R3は、水素原子又は炭素数1~10のアシル基(例えば、ホルミル又はアセチルなど)、Arは、置換基を必要により有するアリーレン基、pは0又は1を表す。)で示される基、式-(CH2)r-Ar-(O)q-(式中、Arは、置換基を必要により有するアリーレン基、qは0又は1、rは0~10である。)で示される基、あるいは式-(CH2)m-SO2-(CH2)n-もしくは-(CH2)m-S-(CH2)n-(式中、mは1~10、nは0~10である)で示される基であってよい。脂肪族基は、アルキレン基(特に炭素数は1~4、例えば1又は2である。)であることが好ましい。芳香族基又は環状脂肪族基は、置換又は非置換であってよい。S基又はSO2基はRf基に直接に結合していてよい。
CH2=C(-Cl)-C(=O)-O-(CH2)2-Rf
CH2=C(-Cl)-C(=O)-O-(CH2)2-S-Rf
CH2=C(-Cl)-C(=O)-O-(CH2)2-S-(CH2)2-Rf
CH2=C(-Cl)-C(=O)-O-(CH2)2-SO2-Rf
CH2=C(-Cl)-C(=O)-O-(CH2)2-SO2-(CH2)2-Rf
CH2=C(-Cl)-C(=O)-NH-(CH2)2-Rf
[Rfは1~20のフルオロアルキル基である。]
CH2=C(-Cl)-C(=O)-O-(CH2)2-Rf
が特に好ましい。
含フッ素重合体は、塩化ビニル及び塩化ビニリデンから選択される少なくとも一種である塩化物単量体(b)から誘導された繰り返し単位を含む。塩化物単量体(b)は塩化ビニルであることが好ましい。
含フッ素重合体は
CH2=CA21-C(=O)-O-A22 式(c)
[式(c)中、A21は水素原子、一価の有機基又はハロゲン原子であり、A22は炭素数2~40の炭化水素基である。]
で示される炭化水素基含有単量体(c)
から誘導された繰り返し単位を有してよい。A21は、水素原子、メチル基又は塩素原子であることが好ましい。A22(炭化水素基)は、炭素数1~30の非環式の脂肪族炭化水素基、及び炭素数4~30の環状炭化水素含有基などであってよい。非環式の脂肪族炭化水素基の炭素数は、好ましくは12~30、より好ましくは18~25である。非環式の脂肪族炭化水素基の具体例は、ラウリル、セチル、ステアリル、及びベヘニルである。環状炭化水素基の具体例は、シクロヘキシル基、t-ブチルシクロヘキシル基、イソボルニル基、ジシクロペンタニル基、ジシクロペンテニル基、及びアダマンチル基である。炭素数1~30の鎖状又は環状の炭化水素基の例は、炭素数1~30の直鎖又は分岐の飽和又は不飽和(例えば、エチレン性不飽和)の脂肪族炭化水素基、炭素数4~30の飽和又は不飽和(例えば、エチレン性不飽和)の環状脂肪族基、炭素数6~30の芳香族炭化水素基、炭素数7~30の芳香脂肪族炭化水素基である。
含フッ素重合体が
式(c)においてA22が炭素数12~30の非環式の脂肪族炭化水素基である長鎖炭化水素基含有単量体(c1)
から誘導された繰り返し単位を含むことが好ましい。12~30の非環式の脂肪族炭化水素基は、直鎖状又は分岐鎖状であってよく、好ましくは直鎖状である。
式(c)においてA22が環状炭化水素基である環状炭化水素基含有単量体(c2)
から誘導された繰り返し単位を含むことが好ましい。環状炭化水素基の具体例としては、シクロヘキシル基含有基、t-ブチルシクロヘキシル基含有基、イソボルニル基含有基、ジシクロペンタニル基含有基、ジシクロペンテニル基含有基、アダマンチル基含有基等が挙げられる。
含フッ素重合体は
反応性基及びオレフィン性炭素-炭素二重結合からなる群から選択される少なくとも2つを有する架橋性単量体(d)
から誘導された繰り返し単位を有してよい。架橋性単量体(d)は、少なくとも2つのエチレン性不飽和二重結合を有する化合物、又は少なくとも1つのエチレン性不飽和二重結合及び少なくとも1つの反応性基を有する化合物であってよい。架橋性単量体(d)は、好ましくは、(メタ)アクリレート基又は(メタ)アクリルアミド基を有する。反応性基の例は、ヒドロキシル基、エポキシ基、クロロメチル基、ブロックイソシアネート基、アミノ基、カルボキシル基、などである。
CH2=CE1-C(=O)-E2-E3-E4
[式中、E1は、水素原子、メチル基又はハロゲン原子(例えば、塩素原子、臭素原子及びヨウ素原子)であり、
E2は、-O-又は-NH-であり、
E3は、炭素数1~20の有機基、例えば、炭素数1~20の直鎖状又は分岐状脂肪族基(特に、アルキレン基)、例えば、式-(CH2)x-(式中、xは1~10である。)で示される基であり、
E4はヒドロキシル基、エポキシ基、クロロメチル基、ブロックイソシアネート基、アミノ基、カルボキシル基である。]
で示される化合物であってよい。
含フッ素重合体は
単量体(a)~(d)以外の他の単量体(e)
から誘導された繰り返し単位を含有してもよい。
未反応の塩化物単量体(b)とは、塩化ビニル及び塩化ビニリデンから選択される少なくとも一種を意味する。
分散液は、液状媒体を含む。液状媒体は、水系媒体であってよい。液状媒体は、水の単独、有機溶媒の単独、あるいは水と(水混和性)有機溶媒との混合物であってよい。水の量は、液状媒体に対して、30重量%以上、50重量%以上、又は80重量%以上であってよい。有機溶媒の量は、液状媒体に対して、30重量%以下、例えば10重量%以下(好ましくは0.1%以上)であってよい。
分散液は、水性分散液である場合に、界面活性剤を含んでもよい。界面活性剤は、ノニオン性界面活性剤及びカチオン性界面活性剤及びアニオン界面活性剤の少なくとも1種を含む。さらに、界面活性剤は、両性界面活性剤を含んでもよい。また、界面活性剤を含有しなくてもよい。分散液は、水性分散液である場合に、一般に、界面活性剤を含む。界面活性剤は、重合前又は重合後に添加してよく、あるいは添加しなくてもよい。界面活性剤を添加しない場合にも、含フッ素重合体が水性媒体に分散した水性分散液が得られる。
アニオン性界面活性剤の例は、ラウリル硫酸ナトリウム、ラウリル硫酸トリエタノールアミン、ポリオキシエチレンラウリルエーテル硫酸ナトリウム、ポリオキシエチレンノニルフェニルエーテル硫酸ナトリウム、ポリオキシエチレンラウリルエーテル硫酸トリエタノールアミン、ココイルサルコシンナトリウム、ナトリウムN - ココイルメチルタウリン、ポリオキシエチレンヤシアルキルエーテル硫酸ナトリウム、ジエーテルヘキシルスルホコハク酸ナトリウム、α - オレフィンスルホン酸ナトリウム、ラウリルリン酸ナトリウム、ポリオキシエチレンラウリルエーテルリン酸ナトリウム等が包含される。
分散液は、硬化剤(活性水素反応性化合物又は活性水素含有化合物)を含んでよい。含フッ素重合体を重合した後に、分散液に硬化剤を加えてもよい。
活性水素反応性化合物の例は、ポリイソシアネート化合物、エポキシ化合物、クロロメチル基含有化合物、カルボキシル基含有化合物及びヒドラジド化合物である。
活性水素含有化合物の例は、ヒドロキシル基含有化合物、アミノ基含有化合物及びカルボキシル基含有化合物、ケトン基含有化合物、ヒドラジド化合物及びメラミン化合物である。
ポリイソシアネート化合物は、1分子中にイソシアネート基を2個以上有する化合物である。ポリイソシアネート化合物は、架橋剤として働く。ポリイソシアネート化合物の例は、脂肪族ポリイソシアネート、脂環族ポリイソシアネート、芳香脂肪族ポリイソシアネート、芳香族ポリイソシアネート及びこれらポリイソシアネートの誘導体などを挙げることができる。
m-フェニレンジイソシアネート、p-フェニレンジイソシアネート、4,4’-ジフェニルジイソシアネート、1,5-ナフタレンジイソシアネート、2,4’-又は4,4’-ジフェニルメタンジイソシアネートもしくはその混合物、2,4-又は2,6-トリレンジイソシアネートもしくはその混合物、トリフェニルメタン-4,4’,4’’-トリイソシアネート、及び4,4’-ジフェニルメタン-2,2’,5,5’-テトライソシアネートなどである。
ポリイソシアネート化合物として、ポリイソシアネート化合物のイソシアネート基をブロック剤でブロックした化合物であるブロック化ポリイソシアネート化合物(ブロックイソシアネート)を使用することが好ましい。水溶液中でも比較的安定であり、分散液と同じ水溶液中でも使用可能である等の理由からブロック化ポリイソシアネート化合物を使用することが好ましい。
クロロメチル基含有化合物はクロロメチル基を有する化合物である。クロロメチル基含有化合物の例は、クロロメチルポリスチレンなどである。
カルボキシル基含有化合物はカルボキシル基を有する化合物である。カルボキシル基含有化合物の例は、(ポリ)アクリル酸、(ポリ)メタクリル酸などである。
ヒドラジド化合物の具体例としては、ヒドラジン、カルボヒドラジド、アジピン酸ヒドラジドなどが挙げられる。
メラミン化合物の具体例としては、メラミン樹脂、メチルエーテル化メラミン樹脂などが挙げられる。
分散液は、上記成分以外の他の成分を含んでよい。一般に、含フッ素重合体を製造した後に、他の成分を加える。他の成分の例は、非フッ素撥水性及び/又は撥油性化合物である。
分散液は、フッ素原子を含まない撥水性及び/又は撥油性化合物(非フッ素撥水性及び/又は撥油性化合物)を含有することがある。
非フッ素撥水性及び/又は撥油性化合物は、非フッ素アクリレート重合体、飽和又は不飽和の炭化水素化合物又はシリコーン系化合物であってよい。
非フッ素アクリレート重合体を構成する非フッ素アクリレート単量体は、式:
CH2=CA-T
[式中、Aは、水素原子、メチル基又はフッ素原子以外のハロゲン原子(例えば、塩素原子、臭素原子及びヨウ素原子)であり、
Tは、水素原子、炭素数1~30の鎖状又は環状の炭化水素基又はエステル結合を有する鎖状又は環状の炭素数1~31の有機基である。]
で示される化合物である。
非フッ素アクリレート重合体は、含フッ素重合体と同様の重合方法で製造できる。
シリコーン系化合物は、一般に、表面処理剤(例えば撥水剤)として使用されているものである。シリコーン系化合物は、撥水性及び/又は撥油性を示す化合物であることが好ましい。
(含フッ素重合体の量)
含フッ素重合体(固形分)の量は、分散液に対して、約0.01~60重量%、好ましくは約0.1~40重量%、より好ましくは約5~35重量%の量であってよい。例えば、保管時においては、高濃度で保管しておき、用いる際に必要に応じて液状媒体を添加して任意の濃度に希釈して使用してもよい。製品安定性が向上したことにより、従来安定性の面から供給に問題のあった高濃度品を供給することが可能となり得る。
分散液中における未反応の塩化物単量体の濃度は、2.0ppm以下、1.5ppm以下、1.0ppm以下、0.8ppm以下、0.5ppm以下、0.3ppm以下、又は0.1ppm以下であってよい。
液状媒体の量は、分散液に対して、30~99.9重量%、特に50~99重量%であってよい。
(界面活性剤の量)
本開示において、界面活性剤を使用しない場合にも、含フッ素重合体の分散液、特に水性分散液を形成できる。界面活性剤の量は、含フッ素重合体(又は単量体の合計)100重量部に対して、0.1~50重量部、例えば、1~30重量部であってよい。
(硬化剤の量)
硬化剤の量は、含フッ素重合体100重量部に対して、100重量部以下、例えば、0.01~30重量部でよい。
(他の成分の量)
非フッ素撥水性及び/又は撥油性化合物の量は、含フッ素重合体100重量部に対して、500重量部以下、例えば、5~200重量部、特に、5~100重量部であってよい。
本開示における分散液の製造方法は、
液状媒体中、フルオロアルキル基を含有し、Q-eスキームにおけるQ値が2.0以上である含フッ素単量体(a)、及び塩化ビニル及び塩化ビニリデンから選択される少なくとも一種である塩化物単量体(b)を共重合して含フッ素重合体を含む重合液を得る工程(i)、及び
得られた重合液中の未反応の塩化物単量体(b)の濃度を2.0ppm以下まで低減させる工程(ii)
を含む。
[含フッ素重合体を含む重合液を得る工程(i)]
本開示における含フッ素重合体は通常の重合方法の何れでも製造でき、また重合反応の条件も任意に選択できる。このような重合方法として、溶液重合、懸濁重合、乳化重合が挙げられる。
含フッ素重合体を重合により製造した後に、水(又は水性媒体)を添加して、含フッ素重合体を水に分散させることが好ましい。
水(又は水性媒体)は、含フッ素重合体を重合により製造した後に、添加してもよい。例えば、単量体を有機溶媒の存在下で重合して、含フッ素重合体を製造した後に、重合体混合物に水を添加して、有機溶媒を留去して、含フッ素重合体を水に分散させてよい。有機溶媒は留去しなくてもよい。界面活性剤は、重合前又は重合後に添加してよく、あるいは添加しなくてもよい。界面活性剤を添加しない場合にも、水性分散液が得られる。硬化剤は、重合前又は重合後に添加してよいが、重合後に添加することが好ましい。他の成分は、重合前又は重合後に添加してよく、あるいは添加しなくてもよい。
重合反応の後に、重合液から未反応の塩化ビニル又は塩化ビニリデンを除去する方法の例としては、減圧による留去、常圧加熱下での撹拌による留去、加熱下又は非加熱下での空気、窒素、スチームのバブリング(例えば、100cc/分以上、200cc/分以上、300cc/分以上、又は400cc/分以上で、3時間以上、6時間以上、12時間以上、又は18時間以上)による留去、充填搭やスピンコーター、円筒状の揮発装置などを利用する方法などが挙げられる。除去工程の際の、温度、時間、気流強度等は適宜変更できる。重合液中の液状媒体の一部を残したまま、塩化物単量体(b)は除去されてよい。
本開示の分散液は、表面処理剤として、従来既知の方法により被処理物に適用することができる。通常、該分散液を有機溶媒又は水に分散して希釈して、浸漬塗布、スプレー塗布、泡塗布などのような既知の方法により、被処理物の表面に付着させ、乾燥する方法が採られる。また、必要ならば、適当な架橋剤と共に適用し、キュアリングを行ってもよい。さらに、本開示の分散液に、防虫剤、柔軟剤、抗菌剤、難燃剤、帯電防止剤、塗料定着剤、防シワ剤などを添加して併用することも可能である。繊維製品と接触させる処理液における重合体の濃度は0.01~10重量%(特に、浸漬塗布の場合)、例えば0.05~10重量%であってよい。
本開示の表面処理剤(例えば撥水剤、撥油剤、撥水撥油剤)で処理される被処理物としては、繊維製品、石材、フィルター(例えば、静電フィルター)、防塵マスク、燃料電池の部品(例えば、ガス拡散電極及びガス拡散支持体)、ガラス、紙、木、皮革、毛皮、石綿、レンガ、セメント、金属及び酸化物、窯業製品、プラスチック、塗面、及びプラスターなどを挙げることができる。繊維製品としては種々の例を挙げることができる。例えば、綿、麻、羊毛、絹などの動植物性天然繊維、ポリアミド、ポリエステル、ポリビニルアルコール、ポリアクリロニトリル、ポリ塩化ビニル、ポリプロピレンなどの合成繊維、レーヨン、アセテートなどの半合成繊維、ガラス繊維、炭素繊維、アスベスト繊維などの無機繊維、あるいはこれらの混合繊維が挙げられる。
本開示の分散液は、内部離型剤あるいは外部離型剤としても使用できる。
半合成繊維の例は、アセテート、トリアセテートである。
再生繊維の例は、レーヨン、キュプラ、ポリノジックレーヨン、リヨセル、テンセルである。
無機繊維の例は、ガラス繊維、炭素繊維である。
あるいは、繊維製品は紙であってもよい。製造重合体を、予め形成した紙に適用してよく、又は、製紙の様々な段階で、例えば、紙の乾燥期間中に適用してもよい。
含フッ素単量体のスチレンとの反応性比(r1、r2)は下記により求めた。
共重合は100mlの4つ口フラスコへ酢酸エチル(25g)、C8FAとスチレンを、単量体の仕込み比が1/9から9/1となるように、(総モル数が0.04モル)仕込んだ。攪拌しながら窒素雰囲気下、60℃に加温した。次に重合開始剤として2,2’-bis 2,4-hydroxybutyrateを(仕込み単量体の0.5モル%)を加えた。共重合は単量体転化率10%以下となるようにした。
共重合体の精製は重合溶液に10倍量のメタノールを加え、生成した沈殿物を遠心分離した。同容量のメタノールで再洗浄し、同様の方法で分離した。生成物をろ過し。130℃で1h乾燥した。共重合組成比は生成した共重合体中のF分析により求めた。
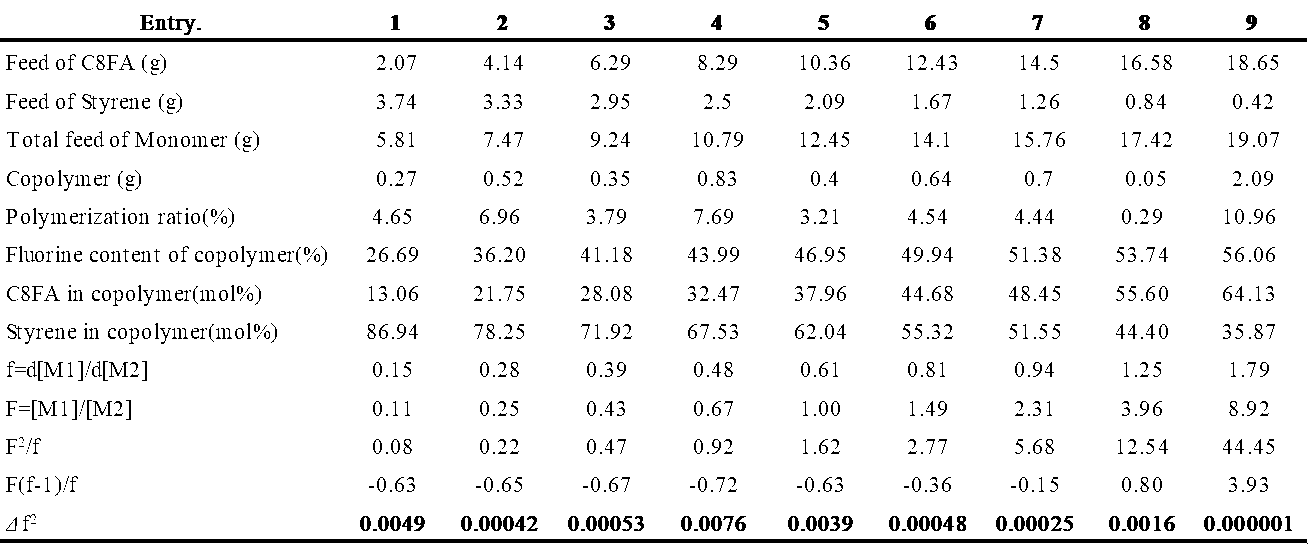
表1に単量体仕込み量、共重合体中のフッ素含有量、共重合体中のFAのモル%とスチレンのモル%を示し、これらより算出されるf=d[M1]/d[M2]、F=[M1]/[M2]、F2/f、F(f-1)/fを表2に示した。
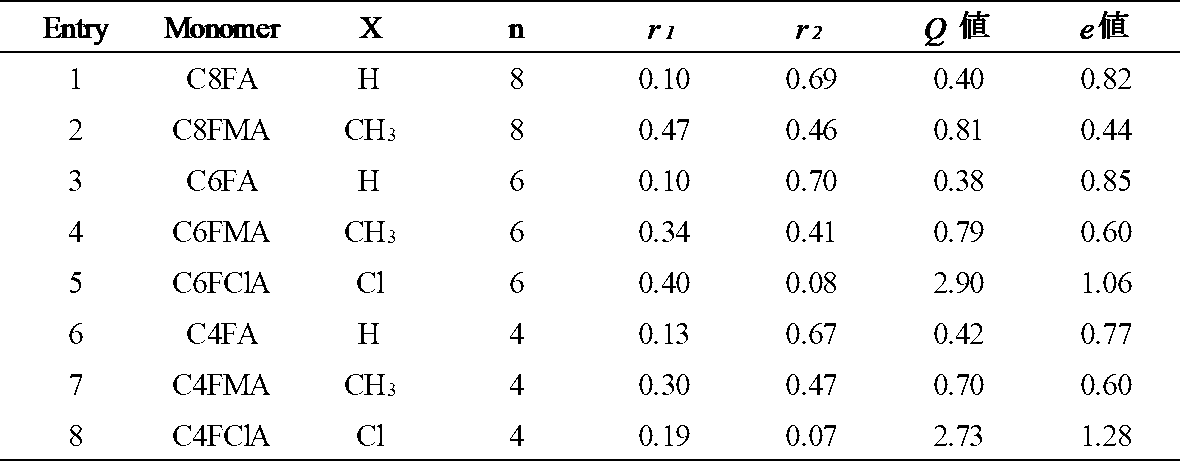
が最小値となるようにr1とr2の組み合わせをもとめた結果、r1=0.10、r2=0.69となった。この値r1=0.10、r2=0.69とスチレンのQ2=1.0、e2=-0.8を式[5]と式[6]へ代入しQ1=0.40、e1=0.82を得た。同様にして各種FA単量体のスチレンとの反応性比(r1、r2)とQ,e値を算出した。結果を表3に示す。
500mlのポリ容器にフルオロアルキルアクリレート(C6FA)50g、ステアリルアクリレート15g、グリシジルメタクリレート3g、水溶性グリコール系溶剤20g、純水294g、ソルビタン脂肪酸エステル2g、カチオン系乳化剤2g、ポリオキシエチレンアルキルエーテル6gを仕込み、60℃に加熱し、ホモミキサーで1分、2000rpmで攪拌した後、超音波で15分間、乳化分散させた。乳化分散物を500mlのオートクレーブに移し、15分間窒素置換後、ラウリルメルカプタン0.2g、塩化ビニルを32g仕込んだ。更にアゾ基含有水溶性開始剤1gを添加し、60℃で昇温し、4時間、反応させて重合体の水性分散液を得た。ガスクロマトクラフィーによる単量体の転化率は99.0~99.8%であった。この分散液を更に純水で希釈して固形分濃度30重量%の水分散体を調製した。続いて、60℃に加熱し、撹拌させながら、窒素ガスによるバブリングを500cc/分で24時間おこない、未反応の塩化ビニル単量体を除いて、含フッ素重合体を含む水分散体を得た。ガスクロマトグラムで分析をおこなった結果、残存する塩化ビニル単量体は検出されなかった(検出限界:1ppm)。次に塩化ビニル単量体を6ppmとなるように添加して、塩化ビニル濃度6ppmである含フッ素重合体を含む水分散体を得た。塩化ビニル濃度はガスクロマトグラム分析により確認した。
製造例1に準じて、表6のとおりに単量体仕込み量と後から添加する塩化ビニル単量体量を設定し、所定の塩化ビニル濃度の含フッ素重合体を含む水分散体を得た。
製造例3で調製した固形分濃度30重量%の含フッ素重合体を含む水分散液33.3gを水道水により希釈して、固形分濃度1.0%の試験液1000gを調製した。この試験液に、ポリエステル生地及びナイロン生地を浸せきした後、マングルで絞った。それぞれの生地を160℃で1分間、ピンテンターに通し、乾燥、キュアリングした。
このようにして処理された生地をJIS L-1092のスプレー法による撥水性試験及び撥油性試験、ブンデスマン試験、安定性評価試験を実施した。その結果を表6に示す。
製造例4で調製した固形分濃度30%の含フッ素重合体を含む水分散液を実施例1と準じて、定量の塩化ビニル単量体を含有する処理液について、評価を行った。その結果を表6に示す。
製造例1、2、及び製造例5~10で調製した固形分濃度30%の含フッ素重合体を含む水分散液を実施例1と同様に処理して、評価を行った。その結果を表6に示す。
処理済試験布を温度21℃、湿度65%の恒温恒湿機に4時間以上保管後、下記評価を行った。上記した各試験用布を、JIS-L-1092(AATCC-22)のスプレー法に準じて処理布の撥水性を評価した。表4に示す基準で撥水性を評価した。なお、点数が大きいほど、撥水性が良好であることを意味する。結果を表6に示す。
処理済試験布を温度21℃、湿度65%の恒温恒湿機に4時間以上保管後、下記評価を行った。
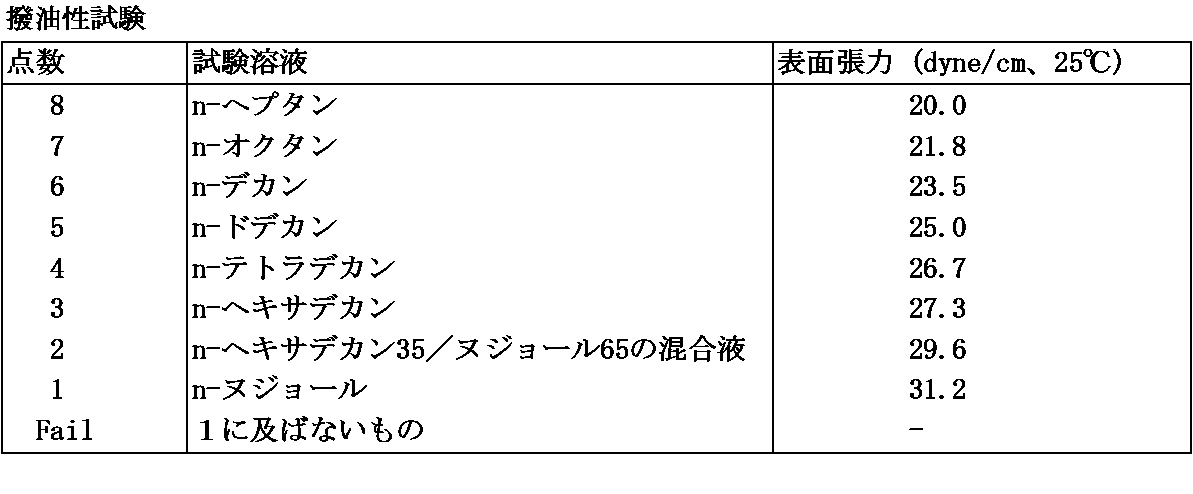
試験布上に試験液(表4)を0.05ml静かに滴下し、30秒間放置後、液滴が試験布上に残っていれば、その試験液をパスしたものとする。撥油性は、パスした試験液の最高点数とし、撥油性不良なものから良好なレベルまでFail、1、2、3、4、5、6、7、及び8の9段階で評価する。結果を表6に示す。
上記した各試験用布について、JIS-L-1092(C)法に記載のブンデスマン試験に従って、降雨量を80cc/分、降雨水温を20℃、降雨時間を1分とする条件で降雨させ、漏水量(mL)を測定した。なお、漏水量とは、ブンデスマン試験中に生地表面を通過した水分量(ml)を示す。その結果を表6に示す。
固形分濃度を30重量%に調整した分散液を50℃にて2週間放置した後、分散液の状態を目視で観察し、下記の基準に従って評価する。
◎:外観に変化無し
○:沈降 は認めらないが、分散液表面に、極少量の析出物が認められる
△:沈降がわずかに認められ、分散液表面に、極少量の析出物が認められる
×:分離又は沈降が認められる
Claims (14)
- 含フッ素重合体及び液状媒体を含む、分散液であって、
含フッ素重合体が
Q値が2.0以上であって、フルオロアルキル基を含有する含フッ素単量体(a)
から誘導される繰り返し単位、及び
塩化ビニル及び塩化ビニリデンから選択される少なくとも一種である塩化物単量体(b)
から誘導される繰り返し単位
を含み、未反応の塩化物単量体(b)の濃度が2.0ppm以下である、分散液。 - 含フッ素単量体(a)が、式:
CH2=C(-X)-C(=O)-Y-Z-Rf
[式中、Xはハロゲン原子であり、
Yは-O-又は-NH-であり、
Zは直接結合又は二価の有機基であり、
Rfは炭素数1~20のフルオロアルキル基である。]
で示される化合物である、請求項1に記載の分散液。 - Xが塩素原子である、請求項2に記載の分散液。
- Rfが炭素数3~6のパーフルオロアルキル基である請求項2又は3に記載の分散液。
- 塩化物単量体(b)から誘導される繰り返し単位の量が、含フッ素重合体に対して25重量%未満である、請求項1~4のいずれか一項に記載の分散液。
- 含フッ素重合体が
式:
CH2=CA21-C(=O)-O-A22 式(c)
[式中、A21は水素原子、一価の有機基又はハロゲン原子であり、A22は炭素数2~40の炭化水素基である。]
で示される炭化水素基含有単量体(c)
から誘導された繰り返し単位をさらに含む、請求項1~5のいずれか一項に記載の分散液。 - 含フッ素重合体が
式(c)においてA22が炭素数12~30の非環式の脂肪族炭化水素基である長鎖炭化水素基含有単量体(c1)
から誘導された繰り返し単位を含む、請求項6に記載の分散液。 - 含フッ素単量体(a)から誘導される繰り返し単位、塩化物単量体(b)から誘導される繰り返し単位、及び炭化水素基含有単量体(c)から誘導される繰り返し単位の合計が、含フッ素重合体に対して90重量%以上である、請求項6又は7に記載の分散液。
- 含フッ素重合体が
反応性基及びオレフィン性炭素-炭素二重結合からなる群から選択される少なくとも2つを有する架橋性単量体(d)
から誘導された繰り返し単位をさらに含む、請求項1~8のいずれか一項に記載の分散液。 - 含フッ素単量体(a)から誘導される繰り返し単位が含フッ素重合体に対して25重量%以上であり、架橋性単量体(d)から誘導される繰り返し単位が含フッ素重合体に対して10重量%以下である、請求項9に記載の分散液。
- 未反応の塩化物単量体(b)の濃度が1.0ppm以下である、請求項1~10のいずれか一項に記載の分散液。
- 液状媒体が少なくとも30重量%以上の水を含む、請求項1~11のいずれか一項に記載の分散液。
- 液状媒体中、フルオロアルキル基を含有し、Q-eスキームにおけるQ値が2.0以上である含フッ素単量体(a)、及び塩化ビニル及び塩化ビニリデンから選択される少なくとも一種である塩化物単量体(b)を共重合して含フッ素重合体を含む重合液を得る工程(i)、及び
得られた重合液中の未反応の塩化物単量体(b)の濃度を2.0ppm以下まで低減させる工程(ii)
を含む、分散液の製造方法。 - 請求項1~12のいずれかに記載の分散液を基材に適用する工程を含む、被処理物の製造方法。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP21764613.2A EP4116342A4 (en) | 2020-03-06 | 2021-03-05 | Dispersion |
| KR1020227019623A KR102722195B1 (ko) | 2020-03-06 | 2021-03-05 | 분산액 |
| CN202180015413.5A CN115135718A (zh) | 2020-03-06 | 2021-03-05 | 分散液 |
| US17/902,394 US20220411663A1 (en) | 2020-03-06 | 2022-09-02 | Dispersion |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2020039222A JP6989800B2 (ja) | 2020-03-06 | 2020-03-06 | 分散液 |
| JP2020-039222 | 2020-03-06 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US17/902,394 Continuation US20220411663A1 (en) | 2020-03-06 | 2022-09-02 | Dispersion |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2021177459A1 true WO2021177459A1 (ja) | 2021-09-10 |
Family
ID=77614334
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2021/008794 Ceased WO2021177459A1 (ja) | 2020-03-06 | 2021-03-05 | 分散液 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20220411663A1 (ja) |
| EP (1) | EP4116342A4 (ja) |
| JP (1) | JP6989800B2 (ja) |
| KR (1) | KR102722195B1 (ja) |
| CN (1) | CN115135718A (ja) |
| TW (1) | TWI815091B (ja) |
| WO (1) | WO2021177459A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022230617A1 (ja) * | 2021-04-27 | 2022-11-03 | ダイキン工業株式会社 | 分散液 |
| WO2023195225A1 (ja) * | 2022-04-06 | 2023-10-12 | ダイキン工業株式会社 | 含フッ素重合体および含フッ素組成物 |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6737417B1 (ja) * | 2019-02-08 | 2020-08-12 | ダイキン工業株式会社 | 撥水剤組成物 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0480218A (ja) | 1990-07-20 | 1992-03-13 | Asahi Glass Co Ltd | 水分散型撥水撥油剤 |
| JP2013511574A (ja) * | 2009-11-20 | 2013-04-04 | ダイキン工業株式会社 | 含フッ素重合体および処理剤 |
| JP2015083619A (ja) * | 2013-10-25 | 2015-04-30 | ダイキン工業株式会社 | 含フッ素剥離剤組成物 |
| WO2017145918A1 (ja) * | 2016-02-23 | 2017-08-31 | ダイキン工業株式会社 | 表面処理剤 |
| WO2018084132A1 (ja) * | 2016-11-01 | 2018-05-11 | ダイキン工業株式会社 | 含フッ素重合体および表面処理剤組成物 |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0756020B2 (ja) * | 1990-07-20 | 1995-06-14 | 旭硝子株式会社 | フッ素系撥水撥油剤 |
| JP3724012B2 (ja) * | 1995-08-04 | 2005-12-07 | チッソ株式会社 | 残留モノマー除去装置およびそれを用いる残留モノマー除去方法 |
| CN104195828B (zh) * | 2008-02-22 | 2018-05-15 | 大金工业株式会社 | 水性聚合物分散组合物和拨水拨油剂 |
| WO2014007343A1 (ja) * | 2012-07-06 | 2014-01-09 | ダイキン工業株式会社 | 透湿防水布帛およびその製法 |
| CN118087269A (zh) * | 2016-08-02 | 2024-05-28 | 大金工业株式会社 | 无纺布 |
-
2020
- 2020-03-06 JP JP2020039222A patent/JP6989800B2/ja active Active
-
2021
- 2021-03-05 KR KR1020227019623A patent/KR102722195B1/ko active Active
- 2021-03-05 TW TW110107950A patent/TWI815091B/zh active
- 2021-03-05 WO PCT/JP2021/008794 patent/WO2021177459A1/ja not_active Ceased
- 2021-03-05 CN CN202180015413.5A patent/CN115135718A/zh active Pending
- 2021-03-05 EP EP21764613.2A patent/EP4116342A4/en active Pending
-
2022
- 2022-09-02 US US17/902,394 patent/US20220411663A1/en not_active Abandoned
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0480218A (ja) | 1990-07-20 | 1992-03-13 | Asahi Glass Co Ltd | 水分散型撥水撥油剤 |
| JP2013511574A (ja) * | 2009-11-20 | 2013-04-04 | ダイキン工業株式会社 | 含フッ素重合体および処理剤 |
| JP2015083619A (ja) * | 2013-10-25 | 2015-04-30 | ダイキン工業株式会社 | 含フッ素剥離剤組成物 |
| WO2017145918A1 (ja) * | 2016-02-23 | 2017-08-31 | ダイキン工業株式会社 | 表面処理剤 |
| WO2018084132A1 (ja) * | 2016-11-01 | 2018-05-11 | ダイキン工業株式会社 | 含フッ素重合体および表面処理剤組成物 |
Non-Patent Citations (7)
| Title |
|---|
| F. R. MAYOF. M. LEWIS, J. AM. CHEM. SOC., vol. 66, 1944, pages 1594 |
| J. BRANDRUPE. H. IMMERGUTE. A. GRULKE: "Polymer Handbook", 1998, JOHN WILEY & SONS INC. |
| M. FINEMANS. D. ROSS, J. POLYM. SCI., vol. 5, 1950, pages 259 |
| P. W. TIDWELLG. A. MORTIMER, J. POLYMER. SCI., vol. A3, 1965, pages 369 |
| See also references of EP4116342A4 |
| T. ALFREY JR.C. C. PRICE, J. POLYM. SCI., vol. 2, 1947, pages 101 |
| T. OTSU: "Progress in Polymer Science Japan", vol. 1, 1970, KODANSHA LTD., pages: 4 |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022230617A1 (ja) * | 2021-04-27 | 2022-11-03 | ダイキン工業株式会社 | 分散液 |
| JP2022169275A (ja) * | 2021-04-27 | 2022-11-09 | ダイキン工業株式会社 | 分散液 |
| WO2023195225A1 (ja) * | 2022-04-06 | 2023-10-12 | ダイキン工業株式会社 | 含フッ素重合体および含フッ素組成物 |
| JP2023154271A (ja) * | 2022-04-06 | 2023-10-19 | ダイキン工業株式会社 | 含フッ素重合体および含フッ素組成物 |
| JP7417141B2 (ja) | 2022-04-06 | 2024-01-18 | ダイキン工業株式会社 | 含フッ素重合体および含フッ素組成物 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2021138878A (ja) | 2021-09-16 |
| US20220411663A1 (en) | 2022-12-29 |
| TW202140586A (zh) | 2021-11-01 |
| JP6989800B2 (ja) | 2022-01-12 |
| EP4116342A4 (en) | 2024-03-13 |
| TWI815091B (zh) | 2023-09-11 |
| KR102722195B1 (ko) | 2024-10-28 |
| CN115135718A (zh) | 2022-09-30 |
| EP4116342A1 (en) | 2023-01-11 |
| KR20220101135A (ko) | 2022-07-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6481748B2 (ja) | 撥水剤 | |
| JP5915641B2 (ja) | 撥水撥油剤組成物 | |
| CN104024292B (zh) | 含氟组合物及含氟聚合物 | |
| JP6819694B2 (ja) | 含フッ素重合体および表面処理剤組成物 | |
| WO2017159754A1 (ja) | 表面処理剤 | |
| CN108884189B (zh) | 共聚物和表面处理剂 | |
| JP6989800B2 (ja) | 分散液 | |
| CN112823170B (zh) | 含氟聚合物和表面处理剂 | |
| WO2013058334A1 (ja) | 含フッ素組成物 | |
| WO2019216117A1 (ja) | 撥水撥油剤および繊維製品 | |
| JP5397520B2 (ja) | 含フッ素組成物および表面処理剤 | |
| WO2013058335A1 (ja) | 含フッ素組成物およびその用途 | |
| JP7417141B2 (ja) | 含フッ素重合体および含フッ素組成物 | |
| WO2022102568A1 (ja) | 共重合体 | |
| JP2013136666A (ja) | 含フッ素重合体の製造方法および含フッ素組成物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 21764613 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 20227019623 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202237049000 Country of ref document: IN |
|
| ENP | Entry into the national phase |
Ref document number: 2021764613 Country of ref document: EP Effective date: 20221006 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |