WO2024257822A1 - Film polarisant, plaque de polarisation, et procédé de fabrication de plaque de polarisation - Google Patents
Film polarisant, plaque de polarisation, et procédé de fabrication de plaque de polarisation Download PDFInfo
- Publication number
- WO2024257822A1 WO2024257822A1 PCT/JP2024/021440 JP2024021440W WO2024257822A1 WO 2024257822 A1 WO2024257822 A1 WO 2024257822A1 JP 2024021440 W JP2024021440 W JP 2024021440W WO 2024257822 A1 WO2024257822 A1 WO 2024257822A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- film
- polarizing
- pva
- polarizing film
- polarizing plate
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Classifications
-
- G—PHYSICS
- G02—OPTICS
- G02B—OPTICAL ELEMENTS, SYSTEMS OR APPARATUS
- G02B5/00—Optical elements other than lenses
- G02B5/30—Polarising elements
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/80—Constructional details
- H10K50/86—Arrangements for improving contrast, e.g. preventing reflection of ambient light
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K59/00—Integrated devices, or assemblies of multiple devices, comprising at least one organic light-emitting element covered by group H10K50/00
- H10K59/10—OLED displays
Definitions
- the present invention relates to a polarizing film, a polarizing plate, and a method for manufacturing a polarizing plate.
- Polarizing plates which have the function of transmitting and blocking light, are a basic component of liquid crystal displays (LCDs) along with liquid crystals, which change the polarization state of light.
- LCDs liquid crystal displays
- Many polarizing plates have a structure in which a protective film such as a triacetyl cellulose (TAC) film is laminated to the surface of a polarizing film.
- TAC triacetyl cellulose
- the mainstream polarizing film that constitutes a polarizing plate is a stretched film in which a dichroic dye such as an iodine-based dye or a dichroic organic dye is adsorbed onto a stretched film that is uniaxially stretched and oriented.
- Such polarizing films are manufactured by uniaxially stretching a PVA film that already contains a dichroic dye, by uniaxially stretching the PVA film and adsorbing the dichroic dye at the same time, or by uniaxially stretching the PVA film and then adsorbing the dichroic dye.
- LCDs are used in a wide range of applications, including small devices such as calculators and watches, laptops, LCD TVs, mobile phones, and tablet devices. In recent years, LCDs have also been used as in-vehicle image display devices such as car navigation systems and rearview monitors. As a result, LCDs are required to be more durable in harsh environments than ever before.
- Patent Document 1 describes that by using a protective film with low water absorption, a polarizing plate with excellent durability and little decrease in transmittance in high temperature environments can be obtained.
- Patent Document 2 describes that polyenation can be suppressed by adjusting the thickness of an adhesive layer provided between the polarizing plate and a transparent plate, etc.
- Patent Documents 1 and 2 tend to suppress the decrease in the light transmittance of the polarizing plate, they were unable to confirm a sufficient effect of suppressing polyenation in durability tests in high-temperature environments of 105°C or higher, which have become necessary in recent years.
- the present invention was made based on the above circumstances, and aims to provide a polarizing film, a polarizing plate, and a manufacturing method for a polarizing plate that can sufficiently suppress the decrease in the light transmittance of the polarizing plate during high-temperature durability tests.
- the present invention provides [1] A polarizing film containing polyvinyl alcohol and an iodine-based dye, wherein a peak top temperature of an endothermic peak appearing in a temperature range of 50°C to 150°C in differential scanning calorimetry of the polarizing film is 102°C or higher; [2] A polarizing plate comprising the polarizing film of [1] and a protective film laminated on at least one surface of the polarizing film, the protective film containing a cellulose ester resin; [3] A method for producing a polarizing plate, comprising the steps of: (1) laminating a protective film on at least one surface of a polarizing film containing polyvinyl alcohol and an iodine-based dye to obtain a laminate; (2) adjusting the moisture content of the laminate to 4.0% by mass or less; and (3) annealing the laminate by heating at 60° C. or more for 4 hours or more; [4] The method for producing a polarizing plate according to [3], wherein
- the present invention provides a polarizing film, a polarizing plate, and a method for manufacturing a polarizing plate that can sufficiently suppress the decrease in light transmittance of the polarizing plate during high-temperature durability tests.
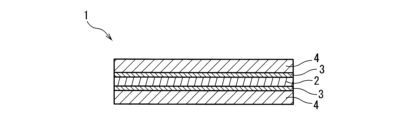
- FIG. 1 is a schematic cross-sectional view of a polarizing plate according to one embodiment of the present invention.
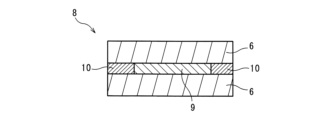
- FIG. 2 is a schematic cross-sectional view of a laminate (a sample for heat resistance test) prepared in a heat resistance test in the examples.
- FIG. 3 is a schematic cross-sectional view of a laminate subjected to an annealing treatment in the examples.
- FIG. 4 is a DSC curve obtained by differential scanning calorimetry for the polarizing film of Example 1.
- a polarizing film according to one embodiment of the present invention is a polarizing film containing PVA and an iodine-based dye, and in differential scanning calorimetry of the polarizing film, the peak top temperature of an endothermic peak appearing in the temperature range of 50°C to 150°C is 102°C or higher.
- This polarizing film can sufficiently suppress the decrease in the light transmittance of the polarizing plate in a high-temperature durability test.
- the reason for this is unclear, but the following is speculated.
- the polyenization of the PVA contained in the polarizing film is thought to occur due to a dehydration reaction with an iodine-based dye as one of the catalysts.
- the protective film of the polarizing plate contains a cellulose ester resin
- the cellulose ester resin is hydrolyzed when the polarizing plate is placed in a high-temperature environment for a long period of time, generating an acid.
- This acid also serves as a catalyst for the dehydration reaction of the PVA, so the polyenization of the PVA occurs particularly significantly.
- the endothermic peak that appears in the temperature range of 50°C to 150°C in the differential scanning calorimetry of the polarizing film is thought to originate from a structure that is presumed to occur due to the crosslinking of the PVA contained in the polarizing film with boric acid (hereinafter, the "structure presumed to occur due to the crosslinking of PVA with boric acid” may be referred to as the "BCC (Boric acid Cross-linked chain Crystallite) structure").
- BCC Boric acid Cross-linked chain Crystallite
- the peak top temperature of the endothermic peak is 102°C or higher, and the thermal stability of the BCC structure is high. For this reason, it is presumed that the polarizing film can sufficiently suppress the decrease in the light transmittance of the polarizing plate in high-temperature heat resistance tests.
- the lower limit of the peak top temperature is 102°C, preferably 103°C, more preferably 104°C, and even more preferably 105°C.
- the upper limit of the peak top temperature may be, for example, 120°C, 110°C, 107°C, or 105°C.
- the peak top temperature can be adjusted by the manufacturing conditions of the polarizing film (stretching temperature, drying temperature, moisture content before annealing treatment, annealing treatment conditions, etc.) as described later.
- the peak top temperature is a value measured by the method described in the examples described later.
- the endothermic peak at the lowest temperature side is taken as the peak top temperature.
- the polarizing film according to one embodiment of the present invention is typically a film in which an iodine-based dye is adsorbed onto a uniaxially stretched PVA film.
- an unstretched film containing PVA may be referred to as a "PVA film.”
- a polarizing film can be manufactured by stretching a PVA film that already contains an iodine-based dye, by adsorbing an iodine-based dye simultaneously with stretching the PVA film, or by adsorbing an iodine-based dye after stretching the PVA film to form a matrix.
- Each component of the polarizing film is described in detail below.
- PVA polyvinyl alcohol
- PVA polyvinyl alcohol
- the PVA may be one obtained by saponifying a polyvinyl ester obtained by polymerizing one or more vinyl esters.
- vinyl esters include vinyl acetate, vinyl formate, vinyl propionate, vinyl butyrate, vinyl pivalate, vinyl versatate, vinyl laurate, vinyl stearate, vinyl benzoate, and isopropenyl acetate.
- vinyl esters compounds having a vinyloxycarbonyl group (H 2 C ⁇ CH—O—CO—) in the molecule are preferred, and vinyl acetate is more preferred, in terms of ease of production, availability, cost, and the like.
- the polyvinyl ester is preferably one obtained by using only one or more vinyl esters as a monomer, and more preferably one obtained by using only one vinyl ester as a monomer.
- the polyvinyl ester may be a copolymer resin of one or more vinyl esters and other monomers copolymerizable therewith.
- the upper limit of the proportion of structural units derived from other copolymerizable monomers is preferably 15 mol%, more preferably 10 mol%, even more preferably 5 mol%, and even more preferably 1 mol%, based on the total structural units constituting the PVA.
- ⁇ -olefins having 2 to 30 carbon atoms such as ethylene, propylene, 1-butene, and isobutene
- (meth)acrylic acid or its salts such as methyl (meth)acrylate, ethyl (meth)acrylate, n-propyl (meth)acrylate, i-propyl (meth)acrylate, n-butyl (meth)acrylate, i-butyl (meth)acrylate, t-butyl (meth)acrylate, 2-ethylhexyl (meth)acrylate, dodecyl (meth)acrylate, and octadecyl (meth)acrylate; (meth)acrylamide; N-methyl (meth)acrylamide, N-ethyl (meth)acrylamide, N,N-dimethyl (meth)acrylamide, di
- the polyvinyl ester may have structural units derived from one or more of the other monomers.
- the PVA one that has not been graft-copolymerized can be preferably used.
- the PVA may be modified with one or more types of graft-copolymerizable monomers.
- Graft copolymerization can be performed on at least one of polyvinyl ester and PVA obtained by saponifying it.
- examples of graft-copolymerizable monomers include unsaturated carboxylic acids or derivatives thereof; unsaturated sulfonic acids or derivatives thereof; and ⁇ -olefins having 2 to 30 carbon atoms.
- the proportion of structural units derived from graft-copolymerizable monomers in polyvinyl ester or PVA is preferably 5 mol % or less of the total structural units constituting polyvinyl ester or PVA.
- the lower limit of the polymerization degree of PVA is preferably 1,000, more preferably 1,500, even more preferably 1,700, and even more preferably 2,000. By having the polymerization degree of PVA be equal to or greater than the lower limit, it is possible to improve the flexibility of the polarizing film.
- the upper limit of this polymerization degree is preferably 10,000, more preferably 8,000, even more preferably 5,000, and even more preferably 3,000. By having the polymerization degree of PVA be equal to or less than the upper limit, it is possible to suppress an increase in the manufacturing cost of PVA and the occurrence of defects during film formation.
- the polymerization degree of PVA means the average polymerization degree measured in accordance with the description of JIS K6726-1994.
- the lower limit of the saponification degree of PVA is preferably 90 mol%, more preferably 95 mol%, even more preferably 99 mol%, and even more preferably 99.5 mol%, because this improves the wet heat resistance of the polarizing film.
- the upper limit of this saponification degree is not particularly limited, and may be 100% or 99.99 mol%.
- the saponification degree of PVA refers to the ratio (mol %) of vinyl alcohol units to the total number of moles of vinyl ester units and vinyl alcohol units.
- the saponification degree can be measured in accordance with the description of JIS K6726-1994.
- the lower limit of the PVA content in the polarizing film is preferably 50% by mass, and may be 60% by mass or 70% by mass.
- the upper limit of the PVA content may be 95% by mass, and may be 90% by mass or 80% by mass.
- the iodine-based dye may be a dichroic dye containing an iodine element.
- the iodine-based dye include I 3 - , I 5 - , and salts containing these anions.
- Examples of counter cations for I 3 - , I 5 - , and the like include alkali metal ions such as potassium ions.
- the iodine-based dye can be obtained, for example, by contacting iodine (I 2 ) with potassium iodide.
- One or more types of iodine-based dyes can be used.
- the content of the iodine-based dye in the polarizing film is not particularly limited, and it is sufficient that an appropriate amount is contained so that the desired polarizing performance is exhibited.
- the polarizing film usually further contains a boron compound.
- the boron compound may be a component that functions as a crosslinking agent. A part or all of the boron compound may form a crosslinked structure with the PVA. Examples of the boron compound include boric acid and borate salts (such as borax), and boric acid is preferred. One or more types of boron compounds may be used.
- the lower limit of the content of the boron compound in the polarizing film is preferably 1 mass%, more preferably 5 mass%, even more preferably 10 mass%, even more preferably 15 mass%, and particularly preferably 20 mass%.
- the upper limit of the content of the boron compound is preferably 45 mass%, more preferably 40 mass%, even more preferably 35 mass%, and even more preferably 30 mass%.
- the polarizing film and the PVA film for producing the polarizing film may contain a plasticizer.
- a plasticizer By containing a plasticizer in the PVA film, the handling property and stretchability of the PVA film can be improved.
- the plasticizer may be released from the PVA film, for example, during swelling treatment, dyeing treatment, etc. Therefore, the plasticizer may not remain in the polarizing film.
- a polyhydric alcohol is preferably used.
- the polyhydric alcohol ethylene glycol, glycerin, propylene glycol, diethylene glycol, diglycerin, triethylene glycol, tetraethylene glycol, trimethylolpropane, etc. can be mentioned, and glycerin is preferable.
- One or more kinds of plasticizers can be used.
- the lower limit of the plasticizer content in the PVA film is preferably 2 parts by mass, more preferably 3 parts by mass, and even more preferably 4 parts by mass, per 100 parts by mass of PVA.
- the upper limit of this content is preferably 20 parts by mass, more preferably 17 parts by mass, and even more preferably 14 parts by mass, per 100 parts by mass of PVA.
- the polarizing film and the PVA film for producing the polarizing film may contain a surfactant.
- a surfactant When a PVA film is produced using a film-forming stock solution as described below, the film-forming property is improved and the occurrence of unevenness in the film thickness is suppressed by adding a surfactant to the film-forming stock solution.
- the PVA film is easily peeled off from the metal roll or belt.
- the PVA film and the polarizing film obtained from the PVA film may contain a surfactant.
- the type of surfactant to be added to the film-forming stock solution for producing a PVA film, and thus the surfactant to be contained in the PVA film and the polarizing film, is not particularly limited, but from the viewpoint of peelability from the metal roll or belt, anionic surfactants and nonionic surfactants are preferred, and nonionic surfactants are particularly preferred.
- anionic surfactants and nonionic surfactants are preferred, and nonionic surfactants are particularly preferred.
- One or more types of surfactants can be used.
- anionic surfactants include carboxylic acid types such as potassium laurate; sulfate ester types such as sodium polyoxyethylene lauryl ether sulfate and octyl sulfate; and sulfonic acid types such as dodecylbenzenesulfonate.
- Nonionic surfactants include, for example, alkyl ether types such as polyoxyethylene oleyl ether; alkyl phenyl ether types such as polyoxyethylene octylphenyl ether; alkyl ester types such as polyoxyethylene laurate; alkyl amine types such as polyoxyethylene lauryl amino ether; alkyl amide types such as polyoxyethylene lauric acid amide; polypropylene glycol ether types such as polyoxyethylene polyoxypropylene ether; alkanolamide types such as lauric acid diethanolamide and oleic acid diethanolamide; and allyl phenyl ether types such as polyoxyalkylene allyl phenyl ether.
- alkyl ether types such as polyoxyethylene oleyl ether
- alkyl phenyl ether types such as polyoxyethylene octylphenyl ether
- alkyl ester types such as polyoxyethylene laurate
- alkyl amine types such
- the lower limit of the surfactant content in the film-forming solution, and therefore the surfactant content in the PVA film or polarized film is preferably 0.01 parts by mass, more preferably 0.02 parts by mass, per 100 parts by mass of PVA contained in the film-forming solution, PVA film, or polarized film.
- the upper limit of the surfactant content is preferably 0.5 parts by mass, more preferably 0.1 parts by mass, per 100 parts by mass of PVA contained in the film-forming solution, PVA film, or polarized film.
- the polarizing film may contain other components other than the PVA, iodine-based pigment, boron compound, plasticizer, and surfactant, such as antioxidants, antifreeze agents, pH adjusters, masking agents, coloring inhibitors, and oils, as necessary.
- the content of other components other than the PVA, iodine-based pigment, boron compound, plasticizer, and surfactant in the polarizing film may be preferably 1% by mass or less, more preferably 0.1% by mass or less.
- the content of inorganic compounds other than the iodine-based pigment in the polarizing film may also be preferably 1% by mass or less, more preferably 0.1% by mass or less.
- the other components and inorganic compounds other than the iodine-based pigment may cause defects such as voids in the polarizing film. For this reason, by reducing the content of the other components and inorganic compounds other than the iodine-based pigment, the polarization performance of the polarizing film tends to be improved.
- the upper limit of the thickness of the polarizing film is, for example, 100 ⁇ m, preferably 50 ⁇ m, more preferably 30 ⁇ m, and even more preferably 20 ⁇ m.
- the lower limit of the thickness may be 1 ⁇ m, preferably 5 ⁇ m, and more preferably 8 ⁇ m.
- the polarizing film may be a single-layer film or a multi-layer film, but from the standpoint of the complexity of lamination work (coating, etc.), cost, etc., a single-layer film is preferable.
- the polarizing film is not particularly limited in its applications, and can be used in the same applications as conventional polarizing films.
- the polarizing film is suitable as a component of a polarizing plate used in image display devices and the like.
- the polarizing film is suitable for applications requiring high-temperature durability, particularly as a polarizing plate for in-vehicle image display devices.
- a polarizing plate according to one embodiment of the present invention includes a polarizing film according to one embodiment of the present invention and a protective film laminated directly or via another layer (e.g., an adhesive layer) on at least one surface of the polarizing film.
- a polarizing plate 1 shown in Fig. 1 includes a polarizing film 2 and a protective film 4 laminated on both surfaces of the polarizing film 2 via an adhesive layer 3. That is, the polarizing plate 1 shown in Fig. 1 has a structure in which a first protective film 4, a first adhesive layer 3, the polarizing film 2, a second adhesive layer 3, and a second protective film 4 are laminated in this order.
- the protective film laminated on at least one side of the polarizing film contains a cellulose ester resin.
- the protective film may be a cellulose ester film containing a cellulose ester resin as a main component.
- the content of the cellulose ester resin in the protective film is preferably 70% by mass or more, and more preferably 90% by mass or more.
- cellulose ester resins examples include cellulose triacetate (triacetyl cellulose: TAC), cellulose diacetate, cellulose acetate propionate, cellulose acetate butyrate, cellulose acetate benzoate, cellulose acetate propionate benzoate, cellulose propionate, cellulose butyrate, cellulose acetate biphenylate, cellulose acetate propionate biphenylate, etc., with cellulose triacetate being preferred.
- both of the two protective films may contain a cellulose ester resin.
- one of the two protective films may be a protective film that does not contain a cellulose ester resin.
- protective films include acrylic films, polyester films, and cycloolefin polymer films.
- the polarizing film and the protective film are usually attached together with an adhesive.
- this adhesive include PVA-based adhesives and UV-curable adhesives, with PVA-based adhesives being preferred.
- PVA-based adhesive an aqueous solution of PVA can be used.
- the solid or cured product of such an adhesive becomes the adhesive layer.
- the specific and preferred forms of the PVA used as the adhesive are the same as those described above as the specific and preferred forms of the PVA contained in the polarizing film.
- the polarizing plate may have a surface treatment such as an anti-reflection treatment, an anti-sticking treatment, an anti-glare treatment, or other coating treatment.
- the polarizing plate may also be used as an optical film with other optical layers further laminated thereon. Examples of other optical layers include a retardation plate and a viewing angle compensation film.
- the polarizing plate can be used in various image display devices such as liquid crystal display devices and organic EL display devices.
- a polarizing film provided in the method for producing a polarizing plate (a polarizing film not subjected to an annealing treatment) may be referred to as a polarizing film (X), and a polarizing film provided in a polarizing plate obtained through the method for producing a polarizing plate (a polarizing film subjected to an annealing treatment) may be referred to as a polarizing film (Y).
- the method for producing the PVA film is not particularly limited, and a method for producing the film with uniform thickness and width after film production can be preferably adopted.
- the PVA film can be produced using a film-forming stock solution in which the PVA constituting the PVA film, and one or more of a plasticizer, a surfactant, and other components as necessary are dissolved in a liquid medium, a film-forming stock solution containing PVA, and one or more of a plasticizer, a surfactant, other components, and a liquid medium as necessary, and in which the PVA is melted, etc.
- the film-forming stock solution contains at least one of a plasticizer, a surfactant, and other components, it is preferable that these components are uniformly mixed.
- Liquid media used to prepare the film-forming solution include, for example, water, dimethyl sulfoxide, dimethylformamide, dimethylacetamide, N-methylpyrrolidone, ethylene glycol, glycerin, propylene glycol, diethylene glycol, triethylene glycol, tetraethylene glycol, trimethylolpropane, ethylenediamine, diethylenetriamine, etc., with water being preferred.
- the volatile content of the film-forming solution (the content in the film-forming solution of volatile components such as liquid medium that are removed by volatilization during film formation) varies depending on the film-forming method and film-forming conditions, but the lower limit is preferably 50 mass%, more preferably 60 mass%, and even more preferably 70 mass%. On the other hand, the upper limit is preferably 96 mass%, and more preferably 92 mass%.
- the volatile content of the film-forming solution is equal to or higher than the lower limit, the viscosity of the film-forming solution does not become too high, and filtration and degassing during preparation of the film-forming solution are carried out smoothly, facilitating the production of PVA films with fewer foreign matter and defects.
- the volatile content of the film-forming solution is equal to or lower than the upper limit, the concentration of the film-forming solution does not become too low, and industrial production of PVA films is facilitated.
- the film-forming method for producing a PVA film using the film-forming solution includes, for example, the cast film-forming method, the extrusion film-forming method, the wet film-forming method, and the gel film-forming method, with the cast film-forming method and the extrusion film-forming method being preferred.
- the extrusion film-forming method is more preferred because it produces a PVA film that is uniform in thickness and width and has good physical properties.
- the PVA film can be dried or heat-treated as necessary.
- the heat treatment temperature there are no particular limitations on the heat treatment temperature, and it may be adjusted as appropriate depending on the degree of swelling of the PVA film, etc.
- the upper limit of the heat treatment temperature is preferably 200°C, more preferably 180°C, and even more preferably 150°C, from the viewpoint of suppressing discoloration and deterioration of the PVA film.
- the lower limit of the heat treatment temperature may be, for example, 80°C.
- heat treatment time there is no particular limit to the heat treatment time, and it may be adjusted appropriately depending on the swelling degree of the PVA film, etc., but from the viewpoint of efficiently producing the PVA film, 1 to 60 minutes is preferable, 2 to 40 minutes is more preferable, and 3 to 30 minutes is even more preferable.
- the method for producing the polarizing film (X) is not particularly limited, and any method that has been conventionally used may be used.
- the polarizing film (X) can be produced by subjecting a PVA film to a swelling treatment, a dyeing treatment, a uniaxial stretching treatment, and, if necessary, a crosslinking treatment, a fixing treatment, a drying treatment, a heat treatment, or the like.
- the order of each treatment such as the swelling treatment, the dyeing treatment, the uniaxial stretching treatment, and the fixing treatment is not particularly limited, and one or more treatments can be performed simultaneously. Also, one or more treatments can be performed twice or more.
- the swelling treatment can be carried out by immersing the PVA film in water.
- the lower limit of the water temperature when immersing in water is preferably 20°C, more preferably 22°C, and even more preferably 25°C. Meanwhile, the upper limit is preferably 40°C, more preferably 38°C, and even more preferably 35°C.
- the time for immersion in water is preferably, for example, 0.1 to 5 minutes.
- the water used when immersing in water is not limited to pure water, and may be an aqueous solution in which various components are dissolved, or a mixture of water and an aqueous medium.
- the dyeing process is carried out using an iodine-based dye.
- Dyeing may be carried out at any stage before, during, or after uniaxial stretching.
- Dyeing is generally carried out by immersing the PVA film in a solution (particularly an aqueous solution) containing iodine and potassium iodide as a dye bath.
- the iodine concentration in the dye bath is preferably 0.01 to 0.5% by mass, and the potassium iodide concentration is preferably 0.01 to 10% by mass.
- the temperature of the dye bath is preferably 20 to 60°C.
- the crosslinking treatment can be carried out by immersing the PVA film in an aqueous solution containing a crosslinking agent.
- a crosslinking agent that can be used include the boron compounds mentioned above.
- the concentration of the crosslinking agent in the aqueous solution containing the crosslinking agent is preferably 1 to 15% by mass, more preferably 2 to 7% by mass.
- the temperature of the aqueous solution containing the crosslinking agent is preferably 20 to 60°C.
- the uniaxial stretching process may be performed by either a wet stretching method or a dry stretching method.
- the wet stretching method it may be performed in an aqueous solution containing a crosslinking agent, or in the dye bath described above or in a fixing treatment bath described below.
- the dry stretching method it may be performed in air.
- the wet stretching method is preferred, and it is more preferred to perform uniaxial stretching in an aqueous solution containing boric acid.
- the concentration of boric acid in the aqueous boric acid solution is preferably 0.5 to 6.0% by mass, more preferably 1.0 to 5.0% by mass, and even more preferably 1.5 to 4.0% by mass.
- the aqueous boric acid solution may also contain potassium iodide, and the concentration is preferably 0.01 to 10% by mass.
- the upper limit of the stretching temperature is preferably 60°C, more preferably 55°C, and may be 50°C.
- the thermal stability of the BCC structure is higher, and therefore the BCC structure is likely to remain during a high-temperature heat resistance test, and polyenization of the PVA is more suppressed.
- the lower limit of the stretching temperature is, for example, preferably 20°C, more preferably 30°C.
- the lower limit of the stretching ratio in the uniaxial stretching process is preferably 5 times, more preferably 5.5 times, from the viewpoint of the polarizing performance of the resulting polarizing film. There is no particular upper limit to the stretching ratio, but 8 times is preferred.
- the fixation bath used for the fixation treatment can be an aqueous solution containing one or more boron compounds such as boric acid and borax. If necessary, iodine compounds, metal compounds, etc. may also be added to the fixation bath.
- the concentration of the boron compound in the fixation bath is generally 0.5 to 15 mass%, and preferably 1 to 10 mass%.
- the temperature of the fixation bath is preferably 15 to 60°C, and more preferably 20 to 40°C.
- the drying temperature for the polarizing film (X) is preferably 40°C or higher, and more preferably 80°C or higher. Drying at this temperature tends to increase the peak top temperature of the endothermic peak in the resulting polarizing film (Y) when the annealing process described below is carried out.
- the upper limit of the drying temperature is, for example, 100°C, or may be 90°C.
- the drying time may be, for example, 1 to 20 minutes, or 2 to 10 minutes.
- a method for producing a polarizing plate according to one embodiment of the present invention includes the steps of (1) obtaining a laminate by laminating a protective film on at least one surface of a polarizing film containing PVA and an iodine-based dye, (2) adjusting the moisture content of the laminate to 4.0 mass% or less, and (3) annealing the laminate by heating at 60°C or higher for 4 hours or more, in that order.
- a polarizing plate is obtained by annealing the laminate.
- the laminate is the polarizing plate before the annealing treatment
- the polarizing plate is the laminate after the annealing treatment.
- a laminate adjusted to a moisture content of 4.0 mass% or less is annealed by heating at 60°C or higher for 4 hours or more, thereby obtaining a polarizing plate in which the decrease in light transmittance is sufficiently suppressed in a high-temperature durability test. This is presumably because the thermal stability of the BBC structure in the polarizing film is increased by performing a predetermined annealing treatment.
- the polarizing film (Y) provided in the polarizing plate obtained by this manufacturing method is a polarizing film containing PVA and an iodine-based dye, and the peak top temperature of the endothermic peak appearing in the temperature range of 50°C to 150°C in differential scanning calorimetry of the polarizing film may be 102°C or higher.
- this manufacturing method includes a method for manufacturing a polarizing film according to one embodiment of the present invention.
- the polarizing film used in step (1) may be the polarizing film (X) obtained by the manufacturing method of the polarizing film (X) described above.
- the laminate can be obtained, for example, by bonding a protective film to at least one surface (preferably both surfaces) of the polarizing film (X) using an adhesive.
- the protective film used in step (1) may be a protective film containing a cellulose ester resin. When using a protective film containing a cellulose ester resin, it is preferable to use a film whose surface (the surface to be bonded to the polarizing film (X)) has been saponified in order to increase adhesion.
- the specific forms of the protective film and adhesive used in this manufacturing method are the same as those described in the description of the polarizing plate according to one embodiment of the present invention.
- step (2) the moisture content of the laminate obtained in step (1) is adjusted to 4.0% by mass or less.
- This step (2) can be performed, for example, by leaving the laminate in an environment adjusted to a predetermined temperature and humidity.
- the temperature at this time is preferably, for example, 5 to 50°C, more preferably 10 to 35°C.
- the humidity at this time may be, for example, 5 to 90% RH or 5 to 50% RH.
- the leaving time may be, for example, 1 to 30 days or 3 to 14 days.
- the moisture content of the polarizing film (X) and the protective film used in the laminate may be the same or different. Regardless of the moisture content values of the polarizing film (X) and the protective film used in the laminate, the moisture content of the laminate as a whole reaches equilibrium by the treatment in step (2), so that the moisture content of the laminate is adjusted to 4.0% by mass or less.
- step (3) the laminate with the moisture content adjusted to 4.0% by mass or less is annealed.
- the upper limit of the moisture content of the laminate to be annealed is preferably 3.8% by mass, more preferably 3.5% by mass, and even more preferably 3.3% by mass.
- the lower limit of the moisture content is preferably 2.0% by mass, and more preferably 2.5% by mass.
- the lower limit of the heating temperature in the annealing treatment is 60°C, preferably 65°C, and more preferably 70°C.
- the upper limit of the heating temperature is preferably 90°C, more preferably 85°C, even more preferably 80°C, and even more preferably 75°C.
- the lower limit of the heating time in the annealing treatment is 4 hours, preferably 6 hours, more preferably 10 hours, and even more preferably 16 hours.
- the heating time is preferably 48 hours, and more preferably 24 hours.
- the method for manufacturing a polarizing plate may further include other steps such as a surface treatment step in addition to the steps (1) to (3) described above.
- the polarizing film, polarizing plate, and polarizing plate manufacturing method of the present invention are not limited to the above-mentioned embodiment.
- the polarizing film of the present invention may be used for a polarizing plate having a protective film that does not contain a cellulose ester resin, for example.
- the polarizing film and polarizing plate of the present invention may be manufactured by a method other than the above-mentioned manufacturing method.
- the adhesive film, the test piece (polarizing plate), and the adhesive film were laminated one by one on the glass plate in this order using a laminator ("Lamyman IKO-360EII” manufactured by Yubon Co., Ltd.).
- a laminator (“Lamyman IKO-360EII” manufactured by Yubon Co., Ltd.).
- another glass plate was placed on the adhesive film to obtain a laminate 5 having a laminated structure of "glass plate 6/adhesive film 7/polarizing plate 1/adhesive film 7/glass plate 6" as shown in Fig. 2.
- the obtained laminate was evacuated at 50°C for 5 minutes, and then pressurized at 10 kPa for 5 minutes to obtain a sample for heat resistance testing.
- the heat resistance test sample was taken out of the metal cage, and the absorbance (Abs) of the heat resistance test sample at a wavelength of 450 nm was measured using an ultraviolet-visible spectrophotometer ("UV-2450" manufactured by Shimadzu Corporation).
- UV-2450 ultraviolet-visible spectrophotometer
- Example 1 Production of PVA film 100 parts by mass of PVA (polymerization degree 2,400, saponification degree 99.95 mol%), 10 parts by mass of glycerin as a plasticizer, 0.03 parts by mass of sodium polyoxyethylene lauryl ether sulfate as a surfactant, and water as a liquid medium were mixed, and then heated at 90 ° C for 4 hours to obtain a PVA aqueous solution with a volatile content of 90% by mass. The obtained aqueous solution was then kept at 85 ° C for 16 hours to degas, and used as a film-forming stock solution for PVA film. This film-forming stock solution was dried on a metal roll at 80 ° C, and then heat-treated for 10 minutes in a dryer at 110 ° C to obtain a PVA film (swelling degree 200%, thickness 30 ⁇ m).
- test piece after the swelling treatment was uniaxially stretched in the length direction to 3.3 times its original length at a stretching speed of 12 cm/min (dyeing treatment).
- test piece after the dyeing treatment was immersed in an aqueous solution (temperature 30° C.) containing 3% by mass of boric acid and 3% by mass of potassium iodide for about 20 seconds, while being uniaxially stretched in the length direction to 3.6 times the original length at a stretching speed of 12 cm/min (crosslinking treatment).
- test piece after the crosslinking treatment was immersed in an aqueous solution (temperature 53° C.) containing 4% by mass of boric acid and about 5% by mass of potassium iodide, while being uniaxially stretched in the length direction to 5.5 times the original length at a stretching speed of 12 cm/min (uniaxial stretching treatment).
- test piece after the uniaxial stretching treatment was immersed in an aqueous solution (temperature 22° C.) containing 1.5% by mass of boric acid and 3% by mass of potassium iodide for 10 seconds (fixing treatment). Finally, it was dried in a dryer at 80° C. for 4 minutes (drying treatment) to obtain a polarizing film (X) (thickness 13 ⁇ m) before annealing treatment.
- TAC film was laminated on each side of the above test piece (polarizing film (X)) via a PVA-based adhesive, and a laminated plate having a configuration of "TAC film / PVA-based adhesive / polarizing film / PVA-based adhesive / TAC film" was produced.
- the resulting laminate was then fixed in a metal frame and dried in a hot air dryer at 60° C. for 10 minutes.
- the boric acid content in the polarizing film (X) before annealing obtained in "(2) Production of polarizing film (X)" was measured by the method described above.
- the peak top temperature was measured by the method described above for the polarizing film (Y) after annealing obtained in "(3) Production of polarizing plate” above.
- a heat resistance test was performed by the method described above for the polarizing plate obtained in "(3) Production of polarizing plate” above. The results are shown in Table 1.
- the obtained DSC curve is shown in Figure 4.
- Examples 2 to 3, Comparative Example 2 A PVA film, a polarizing film, and a polarizing plate were produced and evaluated in the same manner as in Example 1, except that the temperature of the aqueous solution in the uniaxial stretching treatment and the temperature in the drying treatment in the "(2) Production of polarizing film (X)" above, and the moisture content of the laminate (polarizing plate before annealing treatment) in "(ii) Moisture content adjustment” in "(3) Production of polarizing plate” above were changed as shown in Table 1. The results are shown in Table 1.
- the polarizing plates equipped with the polarizing films (Y) of Examples 1 to 3 had low absorbance at a wavelength of 450 nm in the heat resistance test.
- the absorbance of the polarizing plates (heat resistance test samples) of Examples 1 to 3 and Comparative Examples 1 to 3 before the heat resistance test was all 0.3, and there was no difference in the absorbance before the heat resistance test.
- the polarizing films (Y) of Examples 1 to 3 were able to sufficiently suppress the decrease in light transmittance of the polarizing plate.
- the polarizing film and polarizing plate of the present invention can be used in image display devices, etc.
- Polarizing plate 1 Polarizing plate 2 Polarizing film 3 Adhesive layer 4 Protective film 5 Laminate (heat resistance test sample) 6 Glass plate 7 Adhesive film 8 Laminate to be subjected to annealing treatment 9 Laminate plate 10 Silicon-based sealant
Landscapes
- Physics & Mathematics (AREA)
- General Physics & Mathematics (AREA)
- Optics & Photonics (AREA)
- Polarising Elements (AREA)
- Processing And Handling Of Plastics And Other Materials For Molding In General (AREA)
Abstract
L'invention concerne une plaque de polarisation, et un procédé de fabrication de la plaque de polarisation, et un film polarisant capable de supprimer de manière satisfaisante une diminution de la transmittance de lumière de la plaque de polarisation pendant des tests de durabilité à haute température. Le film polarisant contient de l'alcool polyvinylique et un colorant à base d'iode, la température supérieure de pic d'un pic endothermique apparaissant dans une plage de température de 50°C à 150°C étant de 102°C ou plus dans une mesure de calorimétrie différentielle à balayage du film polarisant.
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202480039208.6A CN121359058A (zh) | 2023-06-15 | 2024-06-13 | 偏振膜、偏振板和偏振板的制造方法 |
| JP2025527982A JPWO2024257822A1 (fr) | 2023-06-15 | 2024-06-13 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2023-098464 | 2023-06-15 | ||
| JP2023098464 | 2023-06-15 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024257822A1 true WO2024257822A1 (fr) | 2024-12-19 |
Family
ID=93852198
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2024/021440 Pending WO2024257822A1 (fr) | 2023-06-15 | 2024-06-13 | Film polarisant, plaque de polarisation, et procédé de fabrication de plaque de polarisation |
Country Status (4)
| Country | Link |
|---|---|
| JP (1) | JPWO2024257822A1 (fr) |
| CN (1) | CN121359058A (fr) |
| TW (1) | TW202505230A (fr) |
| WO (1) | WO2024257822A1 (fr) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2017097048A (ja) * | 2015-11-19 | 2017-06-01 | 日本合成化学工業株式会社 | 偏光板用積層体および偏光板 |
| WO2019044741A1 (fr) * | 2017-08-31 | 2019-03-07 | 日本合成化学工業株式会社 | Plaque de polarisation, dispositif d'affichage à cristaux liquides, procédé de production de dispositif d'affichage à cristaux liquides et film de polarisation à base d'alcool polyvinylique |
| JP2022140205A (ja) * | 2021-03-10 | 2022-09-26 | 長春石油化學股▲分▼有限公司 | ポリビニルアルコールフィルム、その製造方法及びそれを使用した光学フィルム |
-
2024
- 2024-06-13 WO PCT/JP2024/021440 patent/WO2024257822A1/fr active Pending
- 2024-06-13 JP JP2025527982A patent/JPWO2024257822A1/ja active Pending
- 2024-06-13 TW TW113121812A patent/TW202505230A/zh unknown
- 2024-06-13 CN CN202480039208.6A patent/CN121359058A/zh active Pending
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2017097048A (ja) * | 2015-11-19 | 2017-06-01 | 日本合成化学工業株式会社 | 偏光板用積層体および偏光板 |
| WO2019044741A1 (fr) * | 2017-08-31 | 2019-03-07 | 日本合成化学工業株式会社 | Plaque de polarisation, dispositif d'affichage à cristaux liquides, procédé de production de dispositif d'affichage à cristaux liquides et film de polarisation à base d'alcool polyvinylique |
| JP2022140205A (ja) * | 2021-03-10 | 2022-09-26 | 長春石油化學股▲分▼有限公司 | ポリビニルアルコールフィルム、その製造方法及びそれを使用した光学フィルム |
Non-Patent Citations (1)
| Title |
|---|
| TA INSTRUMENTS JAPAN: "TA Instruments DSC Basics 1 Glass Transition and Enthalpy Relaxation", XP093274123, Retrieved from the Internet <URL:https://www.youtube.com/watch?v=DbGpordpweg> [retrieved on 20240815] * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN121359058A (zh) | 2026-01-16 |
| JPWO2024257822A1 (fr) | 2024-12-19 |
| TW202505230A (zh) | 2025-02-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7345541B2 (ja) | ポリビニルアルコールフィルム、偏光フィルム、及び偏光板 | |
| TWI762598B (zh) | 偏光薄膜、偏光板、及其製造方法 | |
| TWI710806B (zh) | 偏光膜及其製造方法 | |
| WO2010061706A1 (fr) | Film polarisant contenant de l'iode et procédé de fabrication associé | |
| CN105874364B (zh) | 光学膜制造用初始膜 | |
| JP2010276815A (ja) | 偏光子の製造方法、それによって製造される偏光子、偏光板、及び画像表示装置 | |
| WO2020184657A1 (fr) | Adhésif, et plaque de polarisation | |
| JP7199343B2 (ja) | 偏光フィルム、偏光板、及びそれらの製造方法 | |
| WO2019146678A1 (fr) | Film polarisant et son procédé de fabrication | |
| JP2023053968A (ja) | 偏光板、液晶表示装置およびその製造方法、ならびにポリビニルアルコール系偏光フィルム | |
| WO2018016542A1 (fr) | Procédé de production de film de polarisation | |
| CN105518497A (zh) | 偏光膜 | |
| WO2024257822A1 (fr) | Film polarisant, plaque de polarisation, et procédé de fabrication de plaque de polarisation | |
| JP7583745B2 (ja) | ポリビニルアルコールフィルム及び偏光フィルム | |
| JP7610509B2 (ja) | 偏光フィルム及びその製造方法 | |
| WO2022113958A1 (fr) | Procédé de production de film polarisant et film polarisant | |
| JP7561262B2 (ja) | 偏光フィルム及びその製造方法 | |
| JP7627120B2 (ja) | 偏光子およびその製造方法 | |
| JP7839743B2 (ja) | 偏光フィルムの製造方法及び偏光フィルム | |
| CN109917506B (zh) | 偏振膜 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24823437 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2025527982 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2025527982 Country of ref document: JP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |