BRPI0704816B1 - Formulação lipossomal com o fármaco nistatina - Google Patents
Formulação lipossomal com o fármaco nistatina Download PDFInfo
- Publication number
- BRPI0704816B1 BRPI0704816B1 BRPI0704816-5A BRPI0704816A BRPI0704816B1 BR PI0704816 B1 BRPI0704816 B1 BR PI0704816B1 BR PI0704816 A BRPI0704816 A BR PI0704816A BR PI0704816 B1 BRPI0704816 B1 BR PI0704816B1
- Authority
- BR
- Brazil
- Prior art keywords
- drug
- nystatin
- liposomal
- liposomal formulation
- formulation
- Prior art date
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 53
- 239000003814 drug Substances 0.000 title claims abstract description 51
- 238000009472 formulation Methods 0.000 title claims abstract description 50
- 229940079593 drug Drugs 0.000 title claims abstract description 49
- VQOXZBDYSJBXMA-RKEBNKJGSA-N nystatin a1 Chemical compound O[C@H]1[C@@H](N)[C@H](O)[C@@H](C)O[C@@H]1OC1/C=C/C=C/C=C/C=C/CC/C=C/C=C/[C@H](C)[C@@H](O)[C@@H](C)[C@H](C)OC(=O)CC(O)CC(O)CC(O)CCC(O)C(O)C[C@](O)(CC(O)C2C(O)=O)OC2C1 VQOXZBDYSJBXMA-RKEBNKJGSA-N 0.000 title claims description 5
- VQOXZBDYSJBXMA-NQTDYLQESA-N nystatin A1 Chemical compound O[C@H]1[C@@H](N)[C@H](O)[C@@H](C)O[C@H]1O[C@H]1/C=C/C=C/C=C/C=C/CC/C=C/C=C/[C@H](C)[C@@H](O)[C@@H](C)[C@H](C)OC(=O)C[C@H](O)C[C@H](O)C[C@H](O)CC[C@@H](O)[C@H](O)C[C@](O)(C[C@H](O)[C@H]2C(O)=O)O[C@H]2C1 VQOXZBDYSJBXMA-NQTDYLQESA-N 0.000 claims abstract description 41
- 229960000988 nystatin Drugs 0.000 claims abstract description 39
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 claims abstract description 22
- 238000002360 preparation method Methods 0.000 claims abstract description 19
- 239000008350 hydrogenated phosphatidyl choline Substances 0.000 claims abstract description 17
- 235000012000 cholesterol Nutrition 0.000 claims abstract description 11
- 230000009885 systemic effect Effects 0.000 claims abstract description 8
- 208000031888 Mycoses Diseases 0.000 claims abstract description 6
- 150000008105 phosphatidylcholines Chemical class 0.000 claims abstract description 4
- IWDCLRJOBJJRNH-UHFFFAOYSA-N para-hydroxytoluene Natural products CC1=CC=C(O)C=C1 IWDCLRJOBJJRNH-UHFFFAOYSA-N 0.000 claims abstract description 3
- 239000002502 liposome Substances 0.000 claims description 26
- 150000002632 lipids Chemical class 0.000 claims description 14
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 claims description 12
- 150000003904 phospholipids Chemical class 0.000 claims description 11
- 239000000725 suspension Substances 0.000 claims description 11
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 claims description 10
- 238000000034 method Methods 0.000 claims description 10
- 239000011780 sodium chloride Substances 0.000 claims description 10
- 239000008362 succinate buffer Substances 0.000 claims description 8
- 238000005538 encapsulation Methods 0.000 claims description 7
- 239000002245 particle Substances 0.000 claims description 7
- 150000001875 compounds Chemical class 0.000 claims description 6
- 239000003960 organic solvent Substances 0.000 claims description 5
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 claims description 4
- 230000036571 hydration Effects 0.000 claims description 4
- 238000006703 hydration reaction Methods 0.000 claims description 4
- 230000002829 reductive effect Effects 0.000 claims description 4
- 239000003755 preservative agent Substances 0.000 claims description 3
- 230000002335 preservative effect Effects 0.000 claims description 3
- 239000002904 solvent Substances 0.000 claims description 3
- 239000006228 supernatant Substances 0.000 claims description 3
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 claims description 2
- 229910052719 titanium Inorganic materials 0.000 claims description 2
- 239000010936 titanium Substances 0.000 claims description 2
- 230000000593 degrading effect Effects 0.000 claims 1
- 230000008030 elimination Effects 0.000 claims 1
- 238000003379 elimination reaction Methods 0.000 claims 1
- 230000002045 lasting effect Effects 0.000 claims 1
- 239000008188 pellet Substances 0.000 claims 1
- 238000003756 stirring Methods 0.000 claims 1
- 230000000843 anti-fungal effect Effects 0.000 abstract description 11
- 229940121375 antifungal agent Drugs 0.000 abstract description 11
- 240000004808 Saccharomyces cerevisiae Species 0.000 abstract description 7
- 238000013268 sustained release Methods 0.000 abstract description 7
- 239000012730 sustained-release form Substances 0.000 abstract description 7
- 241000222120 Candida <Saccharomycetales> Species 0.000 abstract description 6
- 241001337994 Cryptococcus <scale insect> Species 0.000 abstract description 6
- 230000004071 biological effect Effects 0.000 abstract description 5
- 238000011835 investigation Methods 0.000 abstract description 3
- 230000001737 promoting effect Effects 0.000 abstract description 3
- 238000012360 testing method Methods 0.000 abstract description 3
- 238000011161 development Methods 0.000 abstract description 2
- 231100000419 toxicity Toxicity 0.000 abstract description 2
- 230000001988 toxicity Effects 0.000 abstract description 2
- FVJZSBGHRPJMMA-IOLBBIBUSA-N PG(18:0/18:0) Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC[C@H](COP(O)(=O)OC[C@@H](O)CO)OC(=O)CCCCCCCCCCCCCCCCC FVJZSBGHRPJMMA-IOLBBIBUSA-N 0.000 abstract 1
- 238000013379 physicochemical characterization Methods 0.000 abstract 1
- 239000000243 solution Substances 0.000 description 7
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 5
- 238000004458 analytical method Methods 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- APKFDSVGJQXUKY-INPOYWNPSA-N amphotericin B Chemical compound O[C@H]1[C@@H](N)[C@H](O)[C@@H](C)O[C@H]1O[C@H]1/C=C/C=C/C=C/C=C/C=C/C=C/C=C/[C@H](C)[C@@H](O)[C@@H](C)[C@H](C)OC(=O)C[C@H](O)C[C@H](O)CC[C@@H](O)[C@H](O)C[C@H](O)C[C@](O)(C[C@H](O)[C@H]2C(O)=O)O[C@H]2C1 APKFDSVGJQXUKY-INPOYWNPSA-N 0.000 description 4
- 239000003242 anti bacterial agent Substances 0.000 description 4
- 229940088710 antibiotic agent Drugs 0.000 description 4
- 230000003115 biocidal effect Effects 0.000 description 4
- 210000004027 cell Anatomy 0.000 description 4
- 210000002540 macrophage Anatomy 0.000 description 4
- 244000005700 microbiome Species 0.000 description 4
- SNKAWJBJQDLSFF-NVKMUCNASA-N 1,2-dioleoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCC\C=C/CCCCCCCC SNKAWJBJQDLSFF-NVKMUCNASA-N 0.000 description 3
- 241000222122 Candida albicans Species 0.000 description 3
- AOJJSUZBOXZQNB-TZSSRYMLSA-N Doxorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 AOJJSUZBOXZQNB-TZSSRYMLSA-N 0.000 description 3
- 241000233866 Fungi Species 0.000 description 3
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 3
- 229930182558 Sterol Natural products 0.000 description 3
- 239000000470 constituent Substances 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 238000011156 evaluation Methods 0.000 description 3
- 238000000198 fluorescence anisotropy Methods 0.000 description 3
- 230000000855 fungicidal effect Effects 0.000 description 3
- 230000002209 hydrophobic effect Effects 0.000 description 3
- 239000002609 medium Substances 0.000 description 3
- 239000012528 membrane Substances 0.000 description 3
- 210000004379 membrane Anatomy 0.000 description 3
- 150000003432 sterols Chemical class 0.000 description 3
- 235000003702 sterols Nutrition 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 230000008685 targeting Effects 0.000 description 3
- OILXMJHPFNGGTO-UHFFFAOYSA-N (22E)-(24xi)-24-methylcholesta-5,22-dien-3beta-ol Natural products C1C=C2CC(O)CCC2(C)C2C1C1CCC(C(C)C=CC(C)C(C)C)C1(C)CC2 OILXMJHPFNGGTO-UHFFFAOYSA-N 0.000 description 2
- RQOCXCFLRBRBCS-UHFFFAOYSA-N (22E)-cholesta-5,7,22-trien-3beta-ol Natural products C1C(O)CCC2(C)C(CCC3(C(C(C)C=CCC(C)C)CCC33)C)C3=CC=C21 RQOCXCFLRBRBCS-UHFFFAOYSA-N 0.000 description 2
- OQMZNAMGEHIHNN-UHFFFAOYSA-N 7-Dehydrostigmasterol Natural products C1C(O)CCC2(C)C(CCC3(C(C(C)C=CC(CC)C(C)C)CCC33)C)C3=CC=C21 OQMZNAMGEHIHNN-UHFFFAOYSA-N 0.000 description 2
- 229930183010 Amphotericin Natural products 0.000 description 2
- QGGFZZLFKABGNL-UHFFFAOYSA-N Amphotericin A Natural products OC1C(N)C(O)C(C)OC1OC1C=CC=CC=CC=CCCC=CC=CC(C)C(O)C(C)C(C)OC(=O)CC(O)CC(O)CCC(O)C(O)CC(O)CC(O)(CC(O)C2C(O)=O)OC2C1 QGGFZZLFKABGNL-UHFFFAOYSA-N 0.000 description 2
- 206010007134 Candida infections Diseases 0.000 description 2
- 241000221204 Cryptococcus neoformans Species 0.000 description 2
- DNVPQKQSNYMLRS-NXVQYWJNSA-N Ergosterol Natural products CC(C)[C@@H](C)C=C[C@H](C)[C@H]1CC[C@H]2C3=CC=C4C[C@@H](O)CC[C@]4(C)[C@@H]3CC[C@]12C DNVPQKQSNYMLRS-NXVQYWJNSA-N 0.000 description 2
- 206010018910 Haemolysis Diseases 0.000 description 2
- 241000446313 Lamella Species 0.000 description 2
- 239000000232 Lipid Bilayer Substances 0.000 description 2
- 206010028851 Necrosis Diseases 0.000 description 2
- 229910019142 PO4 Inorganic materials 0.000 description 2
- 229940009444 amphotericin Drugs 0.000 description 2
- 230000000845 anti-microbial effect Effects 0.000 description 2
- 230000000118 anti-neoplastic effect Effects 0.000 description 2
- 239000012736 aqueous medium Substances 0.000 description 2
- 238000011088 calibration curve Methods 0.000 description 2
- 201000003984 candidiasis Diseases 0.000 description 2
- 230000015556 catabolic process Effects 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 230000034994 death Effects 0.000 description 2
- 238000006731 degradation reaction Methods 0.000 description 2
- 238000010790 dilution Methods 0.000 description 2
- 239000012895 dilution Substances 0.000 description 2
- DNVPQKQSNYMLRS-SOWFXMKYSA-N ergosterol Chemical compound C1[C@@H](O)CC[C@]2(C)[C@H](CC[C@]3([C@H]([C@H](C)/C=C/[C@@H](C)C(C)C)CC[C@H]33)C)C3=CC=C21 DNVPQKQSNYMLRS-SOWFXMKYSA-N 0.000 description 2
- 230000001408 fungistatic effect Effects 0.000 description 2
- 230000008588 hemolysis Effects 0.000 description 2
- 238000004128 high performance liquid chromatography Methods 0.000 description 2
- 238000010348 incorporation Methods 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 230000000813 microbial effect Effects 0.000 description 2
- 244000000010 microbial pathogen Species 0.000 description 2
- 230000017074 necrotic cell death Effects 0.000 description 2
- 230000003287 optical effect Effects 0.000 description 2
- 230000000242 pagocytic effect Effects 0.000 description 2
- 239000012071 phase Substances 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 2
- 239000010452 phosphate Substances 0.000 description 2
- 150000004291 polyenes Chemical class 0.000 description 2
- 239000011148 porous material Substances 0.000 description 2
- 239000011550 stock solution Substances 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- 201000008827 tuberculosis Diseases 0.000 description 2
- STQGQHZAVUOBTE-UHFFFAOYSA-N 7-Cyan-hept-2t-en-4,6-diinsaeure Natural products C1=2C(O)=C3C(=O)C=4C(OC)=CC=CC=4C(=O)C3=C(O)C=2CC(O)(C(C)=O)CC1OC1CC(N)C(O)C(C)O1 STQGQHZAVUOBTE-UHFFFAOYSA-N 0.000 description 1
- 229920001817 Agar Polymers 0.000 description 1
- APKFDSVGJQXUKY-KKGHZKTASA-N Amphotericin-B Natural products O[C@H]1[C@@H](N)[C@H](O)[C@@H](C)O[C@H]1O[C@H]1C=CC=CC=CC=CC=CC=CC=C[C@H](C)[C@@H](O)[C@@H](C)[C@H](C)OC(=O)C[C@H](O)C[C@H](O)CC[C@@H](O)[C@H](O)C[C@H](O)C[C@](O)(C[C@H](O)[C@H]2C(O)=O)O[C@H]2C1 APKFDSVGJQXUKY-KKGHZKTASA-N 0.000 description 1
- 241000222173 Candida parapsilosis Species 0.000 description 1
- 238000002965 ELISA Methods 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- 241000187747 Streptomyces Species 0.000 description 1
- 239000008351 acetate buffer Substances 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- NIXOWILDQLNWCW-UHFFFAOYSA-N acrylic acid group Chemical group C(C=C)(=O)O NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000008272 agar Substances 0.000 description 1
- 229940098178 ambisome Drugs 0.000 description 1
- 229960003942 amphotericin b Drugs 0.000 description 1
- 239000003429 antifungal agent Substances 0.000 description 1
- 239000004599 antimicrobial Substances 0.000 description 1
- 239000008346 aqueous phase Substances 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 230000030833 cell death Effects 0.000 description 1
- 210000000170 cell membrane Anatomy 0.000 description 1
- 238000012512 characterization method Methods 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 231100000313 clinical toxicology Toxicity 0.000 description 1
- 230000001332 colony forming effect Effects 0.000 description 1
- 239000006071 cream Substances 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 229960000975 daunorubicin Drugs 0.000 description 1
- STQGQHZAVUOBTE-VGBVRHCVSA-N daunorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(C)=O)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 STQGQHZAVUOBTE-VGBVRHCVSA-N 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 239000008367 deionised water Substances 0.000 description 1
- 229910021641 deionized water Inorganic materials 0.000 description 1
- 239000008121 dextrose Substances 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 238000010494 dissociation reaction Methods 0.000 description 1
- 230000005593 dissociations Effects 0.000 description 1
- 229940115080 doxil Drugs 0.000 description 1
- 229960004679 doxorubicin Drugs 0.000 description 1
- 239000008298 dragée Substances 0.000 description 1
- 210000003743 erythrocyte Anatomy 0.000 description 1
- 230000005284 excitation Effects 0.000 description 1
- 125000005313 fatty acid group Chemical group 0.000 description 1
- 230000002538 fungal effect Effects 0.000 description 1
- 239000000417 fungicide Substances 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 125000001183 hydrocarbyl group Chemical group 0.000 description 1
- 210000000987 immune system Anatomy 0.000 description 1
- 230000002163 immunogen Effects 0.000 description 1
- 239000002955 immunomodulating agent Substances 0.000 description 1
- 229940121354 immunomodulator Drugs 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 230000002458 infectious effect Effects 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 230000000968 intestinal effect Effects 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 210000004962 mammalian cell Anatomy 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 230000010534 mechanism of action Effects 0.000 description 1
- 210000004400 mucous membrane Anatomy 0.000 description 1
- 210000005170 neoplastic cell Anatomy 0.000 description 1
- 231100000252 nontoxic Toxicity 0.000 description 1
- 230000003000 nontoxic effect Effects 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 239000006186 oral dosage form Substances 0.000 description 1
- 230000008520 organization Effects 0.000 description 1
- 238000007911 parenteral administration Methods 0.000 description 1
- 230000035699 permeability Effects 0.000 description 1
- WTJKGGKOPKCXLL-RRHRGVEJSA-N phosphatidylcholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCC=CCCCCCCCC WTJKGGKOPKCXLL-RRHRGVEJSA-N 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 150000003384 small molecules Chemical class 0.000 description 1
- 235000019980 sodium acid phosphate Nutrition 0.000 description 1
- AJPJDKMHJJGVTQ-UHFFFAOYSA-M sodium dihydrogen phosphate Chemical compound [Na+].OP(O)([O-])=O AJPJDKMHJJGVTQ-UHFFFAOYSA-M 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- 238000000527 sonication Methods 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 238000002798 spectrophotometry method Methods 0.000 description 1
- 239000006208 topical dosage form Substances 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- 239000002691 unilamellar liposome Substances 0.000 description 1
- 231100000925 very toxic Toxicity 0.000 description 1
- 230000000007 visual effect Effects 0.000 description 1
Landscapes
- Medicinal Preparation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
Claims (7)
- FORMULAÇÃO LIPOSSOMAL COM O FÁRMACO NISTATINA caracterizada por ser composta em percentagem molar de 70% de Fosfatidilcolina hidrogenada (HSPC); 20% de Colesterol; 10% de Diestearoilfosfatidilglicerol (DSPG), 1,3% de 2,6 - bis (1,1 -dimetil) - 4 - metil fenol (BHT) em relação à massa de fármaco na formulação e 7% de Nistatina em relação à concentração de lipídio, em tampão succinato 0,02 mol.L-1; pH 5,6 em salina.
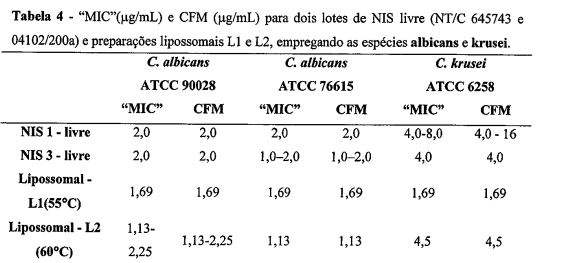
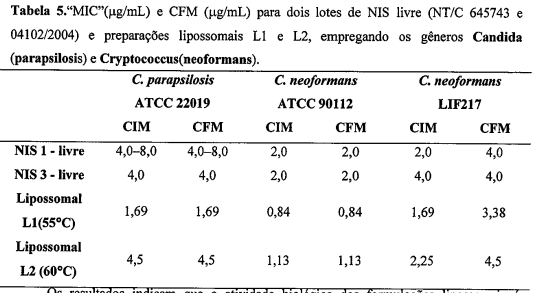
- FORMULAÇÃO LIPOSSOMAL COM O FÁRMACO NISTATINA, de acordo com a reivindicação 1, deu origem a preparação designadas Ll, caracterizada por ser hidratada com tampão succinato 0,02 mol.L-1; pH 5,6 em salina a 55°C, apresentando uma eficiência de encapsulação de Nistatina de 28,9 + 1,0%, teor (%) fosfolipídios totais de; 63,3 ± 0,1%, diâmetro médio das vesículas lipossomais de 64,3 nm e potencial zeta médio de 35,31 ± 1,54 mV.
- FORMULAÇÃO LIPOSSOMAL COM O FÁRMACO NISTATINA, de acordo com a reivindicação 1, deu origem a preparação designadas L2, caracterizada por ser hidratada com tampão succinato 0,02 mol.L-1; pH 5,6 em salina a 60°C, apresentando uma eficiência de encapsulação de Nistatina de 37, 3 ± 1,7%, teor (%) fosfolipídios totais de 57,7 ± 0,1, diâmetro médio das vesículas lipossomais de 68,1 nm e potencial zeta médio de 42,50 ± 0,8 mV.
- FORMULAÇÃO LIPOSSOMAL COM O FÁRMACO NISTATINA, de acordo com as reivindicações 1, 2 e 3, obtida pelo método do filme seco, caracterizada por ser a Nistatina e os lipídios HSPC, colesterol e DSPG dissolvidos em clorofórmio:metanol (1:1; v:v), numa razão molar entre os lipídios de 7:2:1, numa concentração de 15 mM e 1 mM de fármaco; com a utilização do conservante BHT, numa concentração de 1,3% em relação à massa de fármaco utilizado na formulação e os solventes evaporados em rotaevaporador à pressão reduzida, em temperatura de 55°C.
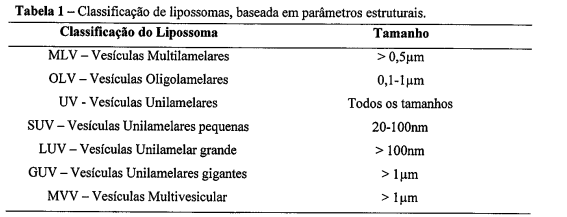
- FORMULAÇÃO LIPOSSOMAL COM O FÁRMACO NISTATINA, sendo o processo para obtenção dos lipossomas multilamelares caracterizado por ser o filme seco obtido na reivindicação 4, mantido por pelo menos 1 hora sob vácuo, para eliminação de resíduos de solventes orgânicos e hidratado nas temperaturas 55°C a 60°C, sendo empregado o tampão succinato (0,02 mol.L-1; pH 5,6) em salina como meio de hidratação. O referido meio de hidratação é adicionado no filme seco em três etapas, com velocidade de agitação designadas: lenta (20rpm), média (160rpm) e rápida (280rpm), cada uma com uma duração de 20 minutos.
- FORMULAÇÃO LIPOSSOMAL COM O FÁRMACO NISTATINA, de acordo com as reivindicações 4 e 5, os lipossomas multilamelares tratados para obtenção de lipossomas de menores tamanhos ou unilamelares caracterizado pelo processo descritos pelos itens abaixo:
- - ultracentigugação a 112.240,71 x g (quando usados 5ml) a 20°C/2h, para eliminação de eventuais compostos de degradação do fármaco (sobrenadante);
- - em seguida, o pélete resultante é ressuspenso a 5ml;
- - a suspensão lipossomal é sonicada em sonicadore de ponte por 10 minutos (3, 3 e 4 min, com pausa de lmin) a uma potência de 40W, mantendo as amostras em banho de gelo, obtendo-se a suspensão lipossomal unilamelar;
- - os lipossomas unilamelares são centrifugados a 9438,65 x g/10min/10°C para retirada das partículas de titânio e fármaco livre;
- - o sobrenadante contendo a suspensão de lipossomas de diâmetro pequeno, com o fármaco encapsulado passa por recozimento a 60°C/20 min.
- FORMULAÇÃO LIPOSSOMAL COM O FÁRMACO NISTATINA, de acordo com as reivindicações 1, 2, 3, 4, 5 e 6, caracterizada pelo uso da Nistatina lipossomada, aplicada na forma parenteral, aos indivíduos portadores de micoses sistêmicas.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BRPI0704816A BRPI0704816B8 (pt) | 2007-12-17 | 2007-12-17 | formulação lipossomal com o fármaco nistatina |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BRPI0704816A BRPI0704816B8 (pt) | 2007-12-17 | 2007-12-17 | formulação lipossomal com o fármaco nistatina |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| BRPI0704816A2 BRPI0704816A2 (pt) | 2009-08-11 |
| BRPI0704816B1 true BRPI0704816B1 (pt) | 2020-09-24 |
| BRPI0704816B8 BRPI0704816B8 (pt) | 2022-07-19 |
Family
ID=40942033
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| BRPI0704816A BRPI0704816B8 (pt) | 2007-12-17 | 2007-12-17 | formulação lipossomal com o fármaco nistatina |
Country Status (1)
| Country | Link |
|---|---|
| BR (1) | BRPI0704816B8 (pt) |
-
2007
- 2007-12-17 BR BRPI0704816A patent/BRPI0704816B8/pt active IP Right Grant
Also Published As
| Publication number | Publication date |
|---|---|
| BRPI0704816A2 (pt) | 2009-08-11 |
| BRPI0704816B8 (pt) | 2022-07-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CA1339008C (en) | Amphotericin b liposome preparation | |
| Shah et al. | LeciPlex, invasomes, and liposomes: A skin penetration study | |
| US5874104A (en) | Treatment of systemic fungal infections with phospholipid particles encapsulating polyene antibiotics | |
| JPH0920652A (ja) | リポソーム中に被包されたイオン化可能な生物学的活性剤を含有する医薬製剤 | |
| Darole et al. | Formulation and evaluation of microemulsion based delivery system for amphotericin B | |
| JP2015502400A (ja) | リポソームの亜塩素酸塩または塩素酸塩組成物 | |
| US20110020428A1 (en) | Gel-stabilized liposome compositions, methods for their preparation and uses thereof | |
| ES2825802T3 (es) | Formulación de anfotericina liposomal que comprende colesterol para tratar infecciones fúngicas | |
| US20050129753A1 (en) | Method for drug loading in liposomes | |
| ES2477233T3 (es) | Liposomas que contienen prostaglandina E1 (PGE1), formulaciones que los contienen y su uso | |
| US20120207821A1 (en) | Liposomal formulation and use thereof | |
| US20040175417A1 (en) | Amphotericin B liposome preparation | |
| BRPI0704816B1 (pt) | Formulação lipossomal com o fármaco nistatina | |
| Gursoy et al. | Characterization of ciprofloxacin liposomes: derivative ultraviolet spectrophotometric determinations | |
| Deĝim et al. | The use of liposomal enrofloxacin for intracellular infections in Kangal dogs and visualization of phagocytosis of liposomes | |
| Larabi et al. | An Original Lipid Complex System for Amphotericin B | |
| US20050169978A1 (en) | Wet-micro grinding | |
| Aramwit | The effect of serum albumin and Pluronics® on the aggregation state and toxicity of polyene macrolides | |
| HK1233914B (zh) | 用於治疗真菌感染的含胆固醇的脂质体两性霉素的制剂 | |
| Karanth | Action of Paclitaxel pH-Sensitive Liposomes on B16F1 Melanoma Cells | |
| BRPI0903009A2 (pt) | vesÍculas lipÍdicas de carotenàides, composiÇço, processo de preparaÇço de vesÍculas lipÍdicas de carotenàides e uso de vesÍculas lipÍdicas de carotenàides |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| B03A | Publication of a patent application or of a certificate of addition of invention [chapter 3.1 patent gazette] | ||
| B08F | Application dismissed because of non-payment of annual fees [chapter 8.6 patent gazette] |
Free format text: REFERENTE A 7A ANUIDADE. |
|
| B08G | Application fees: restoration [chapter 8.7 patent gazette] | ||
| B07D | Technical examination (opinion) related to article 229 of industrial property law [chapter 7.4 patent gazette] | ||
| B06F | Objections, documents and/or translations needed after an examination request according [chapter 6.6 patent gazette] | ||
| B07E | Notification of approval relating to section 229 industrial property law [chapter 7.5 patent gazette] | ||
| B06V | Preliminary requirement: patent application procedure suspended [chapter 6.22 patent gazette] | ||
| B09A | Decision: intention to grant [chapter 9.1 patent gazette] | ||
| B16A | Patent or certificate of addition of invention granted [chapter 16.1 patent gazette] |
Free format text: PRAZO DE VALIDADE: 10 (DEZ) ANOS CONTADOS A PARTIR DE 24/09/2020, OBSERVADAS AS CONDICOES LEGAIS. |
|
| B16C | Correction of notification of the grant [chapter 16.3 patent gazette] |
Free format text: PRAZO DE VALIDADE: 20 (VINTE) ANOS CONTADOS A PARTIR DE 17/12/2007 OBSERVADAS AS CONDICOES LEGAIS. PATENTE CONCEDIDA CONFORME ADI 5.529/DF |
|
| B16C | Correction of notification of the grant [chapter 16.3 patent gazette] |
Free format text: REF. RPI 2594 DE 24/09/2020 QUANTO AO INVENTOR. |
|
| B25C | Requirement related to requested transfer of rights |
Owner name: UNIVERSIDADE ESTADUAL DE MARINGA (BR/PR) Free format text: A FIM DE ATENDER A TRANSFERENCIA DE PARTE DOS DIREITOS, REQUERIDA ATRAVES DA PETICAO NO 870220097689 DE 24/09/2020, E NECESSARIO APRESENTAR DOCUMENTO QUE COMPROVE QUE O REPRESENTANTE DA CEDENTE TEM PODERES PARA REALIZAR TAL ATO. ALEM DISSO, E PRECISO APRESENTAR A GUIA DE CUMPRIMENTO DE EXIGENCIA. |
|
| B25K | Entry of change of name and/or headquarter and transfer of application, patent and certificate of addition of invention: republication |
Owner name: UNIVERSIDADE ESTADUAL DE MARINGA (BR/PR) Free format text: RETIFICACAO DO DESPACHO 25.3 ? TRANSFERENCIA EM EXIGENCIA PUBLICADO NA RPI NO 2719, DE 14/02/2023.ONDE SE LE: REQUERIDA ATRAVES DA PETICAO NO 870220097689 DE 24/09/2020.LEIA-SE: REQUERIDA ATRAVES DA PETICAO NO 870220097689 DE 21/10/2022. |
|
| B25C | Requirement related to requested transfer of rights |
Owner name: UNIVERSIDADE ESTADUAL DE MARINGA (BR/PR) Free format text: A FIM DE ATENDER A TRANSFERENCIA DE PARTE DOS DIREITOS, REQUERIDA ATRAVES DA PETICAO NO 870230026274 DE 29/03/2023, E NECESSARIO APRESENTAR DOCUMENTO QUE COMPROVE QUE A PESSOA QUE ASSINOU O DOCUMENTO DE CESSAO TINHA PODERES PARA REALIZAR TAL ATO, UMA VEZ QUE O NOME DO REITOR QUE ASSINOU O DOCUMENTO DE CESSAO NA PETICAO 870220097689 DE 21/10/2022 E DIFERENTE DONOME DO REITOR QUE CONSTA NO DOCUMENTO APRESENTADO NA PETICAO 870230026274 DE 29/03/2023. ALEM DISSO, E PRECISO APRESENTAR A GUIA DE CUMPRIMENTO DE EXIGENCIA. |
|
| B25A | Requested transfer of rights approved |
Owner name: UNIVERSIDADE ESTADUAL DE MARINGA (BR/PR) ; UNIVERSIDADE ESTADUAL DE CAMPINAS (BR/SP) |