EP0322564B1 - Détergents stables contenant des azurants optiques - Google Patents
Détergents stables contenant des azurants optiques Download PDFInfo
- Publication number
- EP0322564B1 EP0322564B1 EP88119486A EP88119486A EP0322564B1 EP 0322564 B1 EP0322564 B1 EP 0322564B1 EP 88119486 A EP88119486 A EP 88119486A EP 88119486 A EP88119486 A EP 88119486A EP 0322564 B1 EP0322564 B1 EP 0322564B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- fluorescent whitening
- formula
- detergent
- so3m
- hydrogen
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 239000003599 detergent Substances 0.000 title claims description 28
- 230000003287 optical effect Effects 0.000 title description 9
- 150000003839 salts Chemical class 0.000 claims description 12
- 229910052736 halogen Inorganic materials 0.000 claims description 11
- 150000002367 halogens Chemical class 0.000 claims description 11
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 10
- 229910052739 hydrogen Inorganic materials 0.000 claims description 10
- 239000001257 hydrogen Substances 0.000 claims description 10
- 239000000203 mixture Substances 0.000 claims description 10
- 150000004965 peroxy acids Chemical class 0.000 claims description 10
- 238000005406 washing Methods 0.000 claims description 9
- -1 phenylpyrazol-1-yl Chemical group 0.000 claims description 8
- 150000001875 compounds Chemical class 0.000 claims description 7
- 150000004967 organic peroxy acids Chemical class 0.000 claims description 7
- 239000007844 bleaching agent Substances 0.000 claims description 6
- JHUXOSATQXGREM-UHFFFAOYSA-N dodecanediperoxoic acid Chemical compound OOC(=O)CCCCCCCCCCC(=O)OO JHUXOSATQXGREM-UHFFFAOYSA-N 0.000 claims description 6
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 6
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 claims description 5
- 239000004753 textile Substances 0.000 claims description 5
- 150000002431 hydrogen Chemical class 0.000 claims description 4
- 125000000229 (C1-C4)alkoxy group Chemical group 0.000 claims description 3
- 150000001768 cations Chemical class 0.000 claims description 2
- 238000007580 dry-mixing Methods 0.000 claims description 2
- JRKICGRDRMAZLK-UHFFFAOYSA-L peroxydisulfate Chemical compound [O-]S(=O)(=O)OOS([O-])(=O)=O JRKICGRDRMAZLK-UHFFFAOYSA-L 0.000 claims description 2
- 238000002360 preparation method Methods 0.000 claims description 2
- MWNQXXOSWHCCOZ-UHFFFAOYSA-L sodium;oxido carbonate Chemical compound [Na+].[O-]OC([O-])=O MWNQXXOSWHCCOZ-UHFFFAOYSA-L 0.000 claims description 2
- 239000006081 fluorescent whitening agent Substances 0.000 claims 8
- 125000004244 benzofuran-2-yl group Chemical group [H]C1=C(*)OC2=C([H])C([H])=C([H])C([H])=C12 0.000 claims 1
- 239000003054 catalyst Substances 0.000 claims 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims 1
- 239000000463 material Substances 0.000 claims 1
- 239000008187 granular material Substances 0.000 description 5
- 239000011734 sodium Substances 0.000 description 5
- 150000004966 inorganic peroxy acids Chemical class 0.000 description 4
- 238000004061 bleaching Methods 0.000 description 3
- 239000001768 carboxy methyl cellulose Substances 0.000 description 3
- PJANXHGTPQOBST-UHFFFAOYSA-N stilbene Chemical compound C=1C=CC=CC=1C=CC1=CC=CC=C1 PJANXHGTPQOBST-UHFFFAOYSA-N 0.000 description 3
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 2
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 2
- BPQQTUXANYXVAA-UHFFFAOYSA-N Orthosilicate Chemical compound [O-][Si]([O-])([O-])[O-] BPQQTUXANYXVAA-UHFFFAOYSA-N 0.000 description 2
- PJANXHGTPQOBST-VAWYXSNFSA-N Stilbene Natural products C=1C=CC=CC=1/C=C/C1=CC=CC=C1 PJANXHGTPQOBST-VAWYXSNFSA-N 0.000 description 2
- BGRWYDHXPHLNKA-UHFFFAOYSA-N Tetraacetylethylenediamine Chemical compound CC(=O)N(C(C)=O)CCN(C(C)=O)C(C)=O BGRWYDHXPHLNKA-UHFFFAOYSA-N 0.000 description 2
- 238000002835 absorbance Methods 0.000 description 2
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 2
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 2
- UKMSUNONTOPOIO-UHFFFAOYSA-N docosanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCCCC(O)=O UKMSUNONTOPOIO-UHFFFAOYSA-N 0.000 description 2
- YRIUSKIDOIARQF-UHFFFAOYSA-N dodecyl benzenesulfonate Chemical compound CCCCCCCCCCCCOS(=O)(=O)C1=CC=CC=C1 YRIUSKIDOIARQF-UHFFFAOYSA-N 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 239000000835 fiber Substances 0.000 description 2
- 230000009021 linear effect Effects 0.000 description 2
- 239000011777 magnesium Substances 0.000 description 2
- 229910052751 metal Inorganic materials 0.000 description 2
- 239000002184 metal Substances 0.000 description 2
- 238000000034 method Methods 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 239000000344 soap Substances 0.000 description 2
- 235000019832 sodium triphosphate Nutrition 0.000 description 2
- 235000021286 stilbenes Nutrition 0.000 description 2
- UNXRWKVEANCORM-UHFFFAOYSA-I triphosphate(5-) Chemical compound [O-]P([O-])(=O)OP([O-])(=O)OP([O-])([O-])=O UNXRWKVEANCORM-UHFFFAOYSA-I 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 1
- 235000021357 Behenic acid Nutrition 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical class [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 description 1
- ABLZXFCXXLZCGV-UHFFFAOYSA-N Phosphorous acid Chemical class OP(O)=O ABLZXFCXXLZCGV-UHFFFAOYSA-N 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 150000001342 alkaline earth metals Chemical class 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 239000002280 amphoteric surfactant Substances 0.000 description 1
- 125000000129 anionic group Chemical group 0.000 description 1
- 239000003945 anionic surfactant Substances 0.000 description 1
- 239000012752 auxiliary agent Substances 0.000 description 1
- 229940116226 behenic acid Drugs 0.000 description 1
- 230000008033 biological extinction Effects 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 239000003093 cationic surfactant Substances 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 238000010411 cooking Methods 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 229940071161 dodecylbenzenesulfonate Drugs 0.000 description 1
- CQLRGDLBSOUXNO-UHFFFAOYSA-N ethane-1,2-diol;tetradecane Chemical compound OCCO.CCCCCCCCCCCCCC CQLRGDLBSOUXNO-UHFFFAOYSA-N 0.000 description 1
- RTZKZFJDLAIYFH-UHFFFAOYSA-N ether Substances CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 1
- 229940071106 ethylenediaminetetraacetate Drugs 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 239000006260 foam Substances 0.000 description 1
- 238000000227 grinding Methods 0.000 description 1
- 150000002484 inorganic compounds Chemical class 0.000 description 1
- 229910010272 inorganic material Inorganic materials 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 229910052748 manganese Inorganic materials 0.000 description 1
- 239000011572 manganese Substances 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- MGFYIUFZLHCRTH-UHFFFAOYSA-N nitrilotriacetic acid Chemical compound OC(=O)CN(CC(O)=O)CC(O)=O MGFYIUFZLHCRTH-UHFFFAOYSA-N 0.000 description 1
- 239000002736 nonionic surfactant Substances 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 239000002304 perfume Substances 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 239000004014 plasticizer Substances 0.000 description 1
- 229920005646 polycarboxylate Polymers 0.000 description 1
- 229920005996 polystyrene-poly(ethylene-butylene)-polystyrene Polymers 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 239000010453 quartz Substances 0.000 description 1
- 150000004760 silicates Chemical class 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N silicon dioxide Inorganic materials O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 1
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 1
- 229910052938 sodium sulfate Inorganic materials 0.000 description 1
- 235000011152 sodium sulphate Nutrition 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 238000001694 spray drying Methods 0.000 description 1
- 239000012086 standard solution Substances 0.000 description 1
- 239000000375 suspending agent Substances 0.000 description 1
- 239000003760 tallow Substances 0.000 description 1
- QEMXHQIAXOOASZ-UHFFFAOYSA-N tetramethylammonium Chemical compound C[N+](C)(C)C QEMXHQIAXOOASZ-UHFFFAOYSA-N 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-O triethanolammonium Chemical compound OCC[NH+](CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-O 0.000 description 1
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 1
- 239000010457 zeolite Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/39—Organic or inorganic per-compounds
- C11D3/3942—Inorganic per-compounds
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/39—Organic or inorganic per-compounds
- C11D3/3945—Organic per-compounds
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/40—Dyes ; Pigments
- C11D3/42—Brightening agents ; Blueing agents
Definitions
- the present invention relates to storage-stable detergents which, in addition to at least diperoxydodecanedioic acid or its salts, contain at least one specific optical brightener of the stilbene type, and to their use for washing textiles.
- M in formula (I) is, for example, alkaline earth metal such as magnesium or calcium, but preferably alkali metal such as lithium, sodium, potassium and optionally substituted ammonium such as ammonium, mono-, di- or triethanol-ammonium, mono-, di- or tripropanol -ammonium or tri- or tetramethyl-ammonium.
- alkaline earth metal such as magnesium or calcium
- alkali metal such as lithium, sodium, potassium
- ammonium such as ammonium, mono-, di- or triethanol-ammonium, mono-, di- or tripropanol -ammonium or tri- or tetramethyl-ammonium.
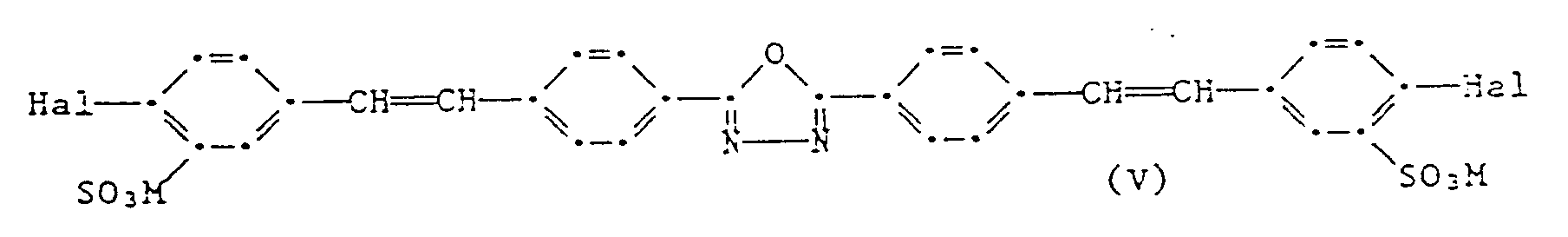
- optical brighteners of the formulas I to V are known and can be prepared by known methods.
- the peracids or their salts are organic or inorganic compounds described in the literature or on the market, which preferably bleach textiles even at temperatures from 20 ° C.
- Diperoxydodecanedioic acid (DPDDA) can be used as the organic peracid.
- the amount of organic peracid to be used is preferably 0.5% to 10%, in particular 1% to 5%.
- very effective inorganic peracids such as persulfate and / or percarbonate can also be used.
- the amount of these inorganic peracids is preferably 1% to 20%, in particular 10% to 20%, based on the total detergent weight. If appropriate and advantageously, they are used in combination with small amounts of catalytically active bivalent metal salts, as are described in US Pat. Nos. 4,655,782 and 4,655,953.
- Metal salts of copper and / or manganese are preferably used.
- the peracids are added to the detergent by dry mixing e.g. with the help of automatic screw dosing systems and / or fluidized bed mixers.
- the detergents are dry detergents of conventional compositions.
- they contain, for example, anionic, nonionic, amphoteric and / or cationic surfactants, builders such as, for example, pentasodium tripolyphosphate or substitute products such as phosphonates, polycarboxylates, acrylic-maleic copolymers, zeolites, nitrilotriacetate, ethylenediaminotetraacetate , Dirt suspending agents such as sodium carboxymethyl cellulose, salts for adjusting the pH such as alkali or alkaline earth silicates, foam regulators such as soap, salts for adjusting the spray drying and granulate properties such as sodium sulfate, perfumes, and optionally antistatic and plasticizers, enzymes, photobleaches, pigments and / or shading agents.
- these components should be stable with respect to the bleaching system used.
- the peracid B (15 g DPDDA) is mixed homogeneously into 500 g of this granulate A, which has a residual moisture of ⁇ 5% after drying.
- the FWA determination mentioned above is carried out as follows: The washing powder is homogenized well by grinding and 1 g of it is mixed with 200 ml of solvent consisting of 9 parts of dimethyl sulfoxide and 1 part of water and stirred at room temperature for 30 minutes. Then it is centrifuged for 30 minutes. A sample of the clear solution thus obtained is transferred with a pipette into a 1 cm quartz cuvette and its absorbance in the UV range is measured at the absorption maximum against a standard solution of the respective FWA. The absorbance is proportional to the FWA concentration. The reproducibility of the results is approx. ⁇ 1% if the test conditions are observed exactly the same.
- Example According to the conditions described above, the percentage FWA loss is determined. Storage is in a closed package, at 20 to 25 ° C. After 6 months, the FWA loss is:
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Wood Science & Technology (AREA)
- Organic Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Detergent Compositions (AREA)
Claims (9)
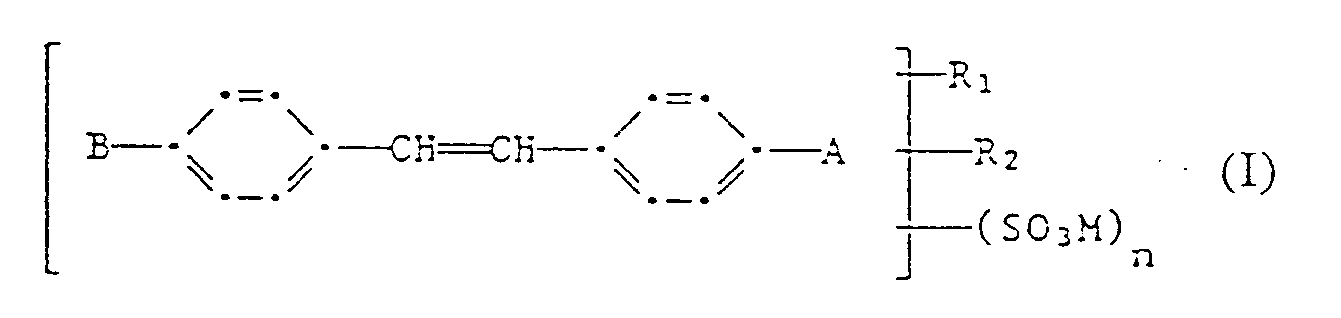
- Détergents secs stables contenant 0,5 à 30 % d'un peracide organique ou de ses sels conjugués ou des mélanges de ceux-ci avec d'autres peracides ou leur sels, ainsi qu'un azurant optique ou un mélange d'azurants optiques, caractérisés en ce qu'ils contiennenta) comme peracide organique, 0,5 à 30 % d'acide diperoxydodécanedioïque ou de ses sels ou 0,5 à 30 % de mélanges d'acide diperoxydodécanedioïque ou de ses sels avec d'autres peracides et leurs sels, etb) 0,03 % à 0,5 % d'un azurant optique ou d'un mélange d'azurants optiques de formule (I)
A représente un atome d'hydrogène ou halogène, un groupe -SO₃M,phényle ou phénylpyrazolyle-1;
B représente un groupe benzofuranyle, dans le cas où A est un atome d'hydrogène,
un groupe 2-(stilbényl-4)-oxadiazolyle-5, dans le cas ou A est un halogène,
un groupe phénylpyrazolyle-1, dans le cas où A est un résidu phénylpyrazolyle-1,
un groupe benzoxazolyle-2, dans le cas où A est un groupe SO₃M ou phény le,
un groupe naphtoxazolyle-2, dans le cas où A est un atome d'hydrogène;
R₁ et R₂ représentent, indépendamment un atome d'hydrogène ou d'halogène, un groupe alkyle en C₁-C₄ ou alkoxy en C₁-C₄;
M représente un atome d'hydrogène ou un équivalent d'un cation non chromophore et
n est un nombre entier compris entre 0 et 2, le composé de formule (I) contenant toutefois au moins un groupe SO₃M. - Détergent conforme à la revendication 1, contenant des peracides ou leurs sels ayant un effet blanchissant des textiles à partir d'une température de 20 °C.
- Détergent conforme à la revendication 2, contenant comme autre peracide ou autre sel de peracide du persulfate et/ou du percarbonate additionné de catalyseur.
- Emploi des détergents conformes aux revendications 1 à 7 pour le lavage de textiles à des températures comprises entre 20 et 60°C.
- Préparation des détergents conformes aux revendications 1 à 7 par mélangeage à l'état sec des composants à l'aide de systèmes doseurs à vis sans fin et/ou de mélangeurs à lit fluidisé.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CH500987 | 1987-12-23 | ||
| CH5009/87 | 1987-12-23 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP0322564A2 EP0322564A2 (fr) | 1989-07-05 |
| EP0322564A3 EP0322564A3 (en) | 1989-08-30 |
| EP0322564B1 true EP0322564B1 (fr) | 1994-06-15 |
Family
ID=4286370
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP88119486A Expired - Lifetime EP0322564B1 (fr) | 1987-12-23 | 1988-11-23 | Détergents stables contenant des azurants optiques |
Country Status (7)
| Country | Link |
|---|---|
| EP (1) | EP0322564B1 (fr) |
| JP (1) | JP2634453B2 (fr) |
| AU (1) | AU624587B2 (fr) |
| BR (1) | BR8806215A (fr) |
| DE (1) | DE3850234D1 (fr) |
| ES (1) | ES2054774T3 (fr) |
| ZA (1) | ZA888793B (fr) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6991747B2 (en) | 1999-10-19 | 2006-01-31 | Procter & Gamble Company | Radical scavenger |
Families Citing this family (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0317979B1 (fr) * | 1987-11-26 | 1994-12-07 | Ciba-Geigy Ag | Détergents stables contenant des azurants optiques |
| US4970019A (en) * | 1988-10-27 | 1990-11-13 | Fmc Corporation | Particulate composition containing bleach and optical brightener and process for its manufacture |
| EP0724012A1 (fr) * | 1995-01-27 | 1996-07-31 | The Procter & Gamble Company | Composition détergente contenant un agent de blanchiment et un agent d'azurage |
| US6566320B1 (en) | 1999-10-19 | 2003-05-20 | The Procter & Gamble Company | Bleaching composition containing chromotropic compound |
| KR100351295B1 (ko) * | 2000-03-29 | 2002-09-05 | 김기식 | 압축프레스 안전점검장치 거치대 |
| ATE520767T1 (de) | 2008-04-25 | 2011-09-15 | Procter & Gamble | Gefärbte bleichmittelzusammensetzung |
| CN104046350B (zh) * | 2013-03-15 | 2018-08-17 | 广州熵能创新材料股份有限公司 | 一种荧光微球及制备方法和应用 |
| ES2940468T3 (es) | 2015-06-22 | 2023-05-08 | Fater Spa | Composición blanqueadora estable |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0317979A2 (fr) * | 1987-11-26 | 1989-05-31 | Ciba-Geigy Ag | Détergents stables contenant des azurants optiques |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4028263A (en) * | 1973-08-24 | 1977-06-07 | Colgate-Palmolive Company | Bleaching and brightening detergent composition |
| US4177347A (en) * | 1974-06-12 | 1979-12-04 | Ciba-Geigy Corporation | Distilbenyl-oxdiazoles |
| CH610478GA3 (en) * | 1974-06-12 | 1979-04-30 | Use of distilbenyloxadiazoles for the optical brightening of organic textile materials | |
| CH612817GA3 (en) * | 1974-06-12 | 1979-08-31 | Use of stilbene compounds for the fluorescent brightening of textile organic materials | |
| DE2525637A1 (de) * | 1974-06-12 | 1976-01-02 | Ciba Geigy Ag | Distilbenyl-oxdiazole |
| DE2525683A1 (de) * | 1974-06-12 | 1976-01-02 | Ciba Geigy Ag | Sulfogruppenhaltige heterocyclen |
| CH603733A5 (en) * | 1975-05-02 | 1978-08-31 | Ciba Geigy Ag | Stilbene cpds. used as optical brightening agents |
| DE2756583A1 (de) * | 1977-12-19 | 1979-06-21 | Henkel Kgaa | Zur textilbehandlung geeignetes, perverbindungen und optische aufheller enthaltendes bleichmittel |
| ES2001074A6 (es) * | 1985-08-21 | 1988-04-16 | Clorox Co | Perfeccionamientos en la fabricacion de productos blanqueantes secos basados en diperacidos. |
-
1988
- 1988-11-23 EP EP88119486A patent/EP0322564B1/fr not_active Expired - Lifetime
- 1988-11-23 DE DE3850234T patent/DE3850234D1/de not_active Expired - Fee Related
- 1988-11-23 ES ES88119486T patent/ES2054774T3/es not_active Expired - Lifetime
- 1988-11-24 JP JP63294819A patent/JP2634453B2/ja not_active Expired - Lifetime
- 1988-11-24 ZA ZA888793A patent/ZA888793B/xx unknown
- 1988-11-25 AU AU25935/88A patent/AU624587B2/en not_active Ceased
- 1988-11-25 BR BR888806215A patent/BR8806215A/pt unknown
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0317979A2 (fr) * | 1987-11-26 | 1989-05-31 | Ciba-Geigy Ag | Détergents stables contenant des azurants optiques |
Non-Patent Citations (1)
| Title |
|---|
| Ullmann's Encyclopedia of Industrial Chemistry , 5th ed., vol. A8, pp. 360-362 * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6991747B2 (en) | 1999-10-19 | 2006-01-31 | Procter & Gamble Company | Radical scavenger |
Also Published As
| Publication number | Publication date |
|---|---|
| AU624587B2 (en) | 1992-06-18 |
| JPH01172497A (ja) | 1989-07-07 |
| EP0322564A3 (en) | 1989-08-30 |
| AU2593588A (en) | 1989-07-06 |
| ES2054774T3 (es) | 1994-08-16 |
| JP2634453B2 (ja) | 1997-07-23 |
| DE3850234D1 (de) | 1994-07-21 |
| EP0322564A2 (fr) | 1989-07-05 |
| ZA888793B (en) | 1989-08-30 |
| BR8806215A (pt) | 1989-08-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| DE68901888T2 (de) | Waessrige, lagerbestaendige bleich- und waschmittel und waschverfahren. | |
| EP0026744B1 (fr) | Moyen et procédé pour le traitement et l'amélioration de l'apparence de textiles | |
| EP0322564B1 (fr) | Détergents stables contenant des azurants optiques | |
| DE68901737T2 (de) | Alkylsulphonylpercarbonsaeuren und die sie enthaltenden bleichmittel- und reinigungszusammensetzungen. | |
| DE1961775B2 (de) | Wasch- und Reinigungsmittel | |
| EP0321715B1 (fr) | Détergents stables contenant des azurants optiques | |
| EP0317979B1 (fr) | Détergents stables contenant des azurants optiques | |
| US5035825A (en) | Stable bleaching detergents containing stilbene fluorescent whitening agents | |
| EP0317978B1 (fr) | Détergents stables contenant des azurants optiques | |
| EP0002746A1 (fr) | Agent de blanchiment contenant des composés "per" et des azurants optiques et son application comme agent de traitement de textiles | |
| EP0332050A1 (fr) | Activateurs pour composés per minéraux | |
| DE2616350C3 (fr) | ||
| EP0116887A2 (fr) | Composés cyanamides organiques comme activeurs pour composés péroxidés | |
| US5230820A (en) | Storage-stable bleaching detergents containing bis-benzofuranyl fluoescent whitening agents | |
| DE2301437C3 (de) | Mit optischen Aufhellern verträgliche peraeylierte Polyamine als Aktivatoren für anorganische Perverbindungen | |
| DE2242093C2 (de) | Phosphatfreie alkyläthersulfathaltige Waschmittel | |
| CH601486A5 (en) | Sulphonate detergent | |
| DE2242157C3 (de) | Phosphatfreie olefinsulfonathaltige Waschmittel | |
| DE4231466A1 (de) | Aktivatoren für anorganische Perverbindungen | |
| DE1419307A1 (de) | Bleichmittel | |
| DE69617170T2 (de) | Waschmittelzusammensetzungen | |
| DE1617112B2 (de) | Weißwaschmittel | |
| AT287636B (de) | Optische Aufhellungsmittel und Komplexierungsmittel enthaltendes Waschmittel | |
| EP0395588A1 (fr) | Dibenzofuranylbiphényles | |
| DE3514877A1 (de) | Verfahren zur aktivierung von anorganischen peroxyverbindungen |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 19881123 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): CH DE ES FR GB IT LI NL SE |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): CH DE ES FR GB IT LI NL SE |
|
| 17Q | First examination report despatched |
Effective date: 19920730 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): CH DE ES FR GB IT LI NL SE |
|
| REF | Corresponds to: |
Ref document number: 3850234 Country of ref document: DE Date of ref document: 19940721 |
|
| ET | Fr: translation filed | ||
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2054774 Country of ref document: ES Kind code of ref document: T3 |
|
| GBT | Gb: translation of ep patent filed (gb section 77(6)(a)/1977) |
Effective date: 19940802 |
|
| ITF | It: translation for a ep patent filed | ||
| EAL | Se: european patent in force in sweden |
Ref document number: 88119486.4 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 19960926 Year of fee payment: 9 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 19960930 Year of fee payment: 9 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 19961004 Year of fee payment: 9 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 19961024 Year of fee payment: 9 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PUE Owner name: CIBA-GEIGY AG TRANSFER- CIBA SC HOLDING AG |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 19961107 Year of fee payment: 9 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 19961112 Year of fee payment: 9 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SE Payment date: 19961125 Year of fee payment: 9 |
|
| NLS | Nl: assignments of ep-patents |
Owner name: CIBA SC HOLDING AG |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: TP |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PFA Free format text: CIBA SC HOLDING AG TRANSFER- CIBA SPECIALTY CHEMICALS HOLDING INC. |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: 732E |
|
| NLT1 | Nl: modifications of names registered in virtue of documents presented to the patent office pursuant to art. 16 a, paragraph 1 |
Owner name: CIBA SPECIALTY CHEMICALS HOLDING INC. |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19971123 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19971124 Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19971124 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19971130 Ref country code: FR Free format text: THE PATENT HAS BEEN ANNULLED BY A DECISION OF A NATIONAL AUTHORITY Effective date: 19971130 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19971130 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: CD |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19980601 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: PC2A |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 19971123 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19980801 |
|
| EUG | Se: european patent has lapsed |
Ref document number: 88119486.4 |
|
| NLV4 | Nl: lapsed or anulled due to non-payment of the annual fee |
Effective date: 19980601 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 19981212 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES;WARNING: LAPSES OF ITALIAN PATENTS WITH EFFECTIVE DATE BEFORE 2007 MAY HAVE OCCURRED AT ANY TIME BEFORE 2007. THE CORRECT EFFECTIVE DATE MAY BE DIFFERENT FROM THE ONE RECORDED. Effective date: 20051123 |