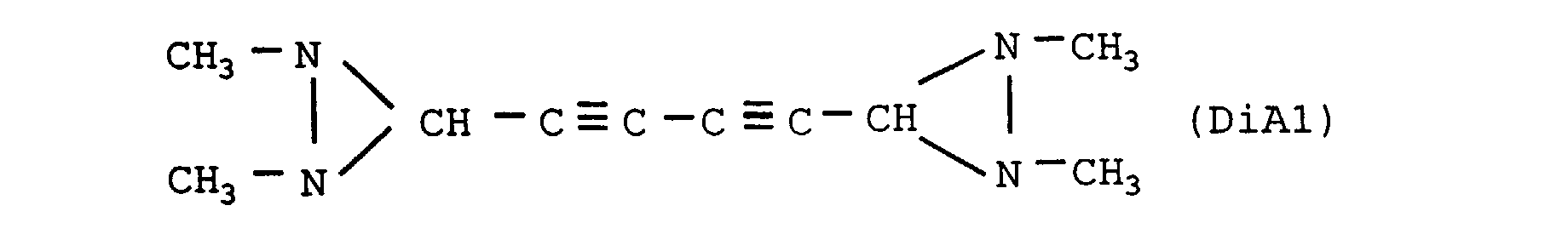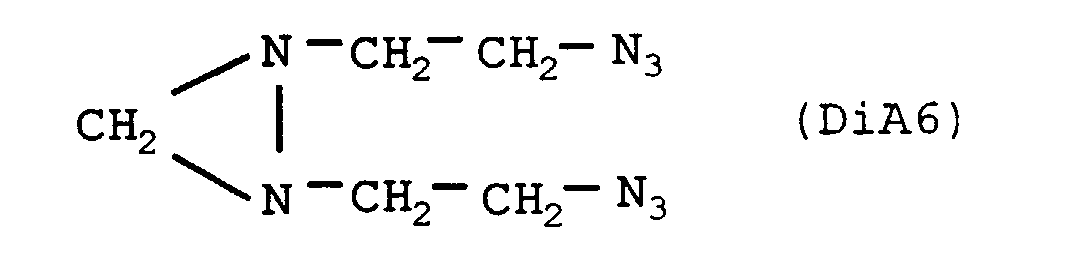EP1364931A2 - Compositions de poudres propulsives pour armes à tube à force élevée et effet érosif réduit - Google Patents
Compositions de poudres propulsives pour armes à tube à force élevée et effet érosif réduit Download PDFInfo
- Publication number
- EP1364931A2 EP1364931A2 EP03291108A EP03291108A EP1364931A2 EP 1364931 A2 EP1364931 A2 EP 1364931A2 EP 03291108 A EP03291108 A EP 03291108A EP 03291108 A EP03291108 A EP 03291108A EP 1364931 A2 EP1364931 A2 EP 1364931A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- weight
- nitrocellulose
- nitroglycerin
- powder
- nitrogenous
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000000843 powder Substances 0.000 title claims abstract description 81
- 239000000203 mixture Substances 0.000 title claims abstract description 56
- 239000003380 propellant Substances 0.000 title claims abstract description 30
- 230000003628 erosive effect Effects 0.000 title claims abstract description 15
- 230000002829 reductive effect Effects 0.000 title description 5
- 239000000020 Nitrocellulose Substances 0.000 claims abstract description 33
- 229920001220 nitrocellulos Polymers 0.000 claims abstract description 33
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims abstract description 32
- 229960003711 glyceryl trinitrate Drugs 0.000 claims abstract description 29
- SNIOPGDIGTZGOP-UHFFFAOYSA-N Nitroglycerin Chemical compound [O-][N+](=O)OCC(O[N+]([O-])=O)CO[N+]([O-])=O SNIOPGDIGTZGOP-UHFFFAOYSA-N 0.000 claims abstract description 27
- 229910052757 nitrogen Inorganic materials 0.000 claims abstract description 27
- 238000000034 method Methods 0.000 claims abstract description 20
- 150000002894 organic compounds Chemical class 0.000 claims abstract description 15
- 229910052799 carbon Inorganic materials 0.000 claims abstract description 12
- 229910052739 hydrogen Inorganic materials 0.000 claims abstract description 12
- 150000001875 compounds Chemical class 0.000 claims abstract description 11
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims abstract description 7
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims abstract description 6
- 239000001257 hydrogen Substances 0.000 claims abstract description 6
- 239000000006 Nitroglycerin Substances 0.000 claims description 28
- 230000015572 biosynthetic process Effects 0.000 claims description 14
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 claims description 11
- POCJOGNVFHPZNS-ZJUUUORDSA-N (6S,7R)-2-azaspiro[5.5]undecan-7-ol Chemical compound O[C@@H]1CCCC[C@]11CNCCC1 POCJOGNVFHPZNS-ZJUUUORDSA-N 0.000 claims description 6
- BSPUVYFGURDFHE-UHFFFAOYSA-N Nitramine Natural products CC1C(O)CCC2CCCNC12 BSPUVYFGURDFHE-UHFFFAOYSA-N 0.000 claims description 6
- POCJOGNVFHPZNS-UHFFFAOYSA-N isonitramine Natural products OC1CCCCC11CNCCC1 POCJOGNVFHPZNS-UHFFFAOYSA-N 0.000 claims description 6
- 239000000945 filler Substances 0.000 claims description 3
- 150000003536 tetrazoles Chemical class 0.000 claims description 3
- KJUGUADJHNHALS-UHFFFAOYSA-N 1H-tetrazole Substances C=1N=NNN=1 KJUGUADJHNHALS-UHFFFAOYSA-N 0.000 claims description 2
- 150000004905 tetrazines Chemical class 0.000 claims description 2
- 150000003918 triazines Chemical class 0.000 claims description 2
- 150000003852 triazoles Chemical class 0.000 claims description 2
- 230000000052 comparative effect Effects 0.000 description 34
- 238000002485 combustion reaction Methods 0.000 description 18
- -1 tetrazole Chemical compound 0.000 description 17
- ULRPISSMEBPJLN-UHFFFAOYSA-N 2h-tetrazol-5-amine Chemical compound NC1=NN=NN1 ULRPISSMEBPJLN-UHFFFAOYSA-N 0.000 description 11
- IDCPFAYURAQKDZ-UHFFFAOYSA-N 1-nitroguanidine Chemical compound NC(=N)N[N+]([O-])=O IDCPFAYURAQKDZ-UHFFFAOYSA-N 0.000 description 9
- XTFIVUDBNACUBN-UHFFFAOYSA-N 1,3,5-trinitro-1,3,5-triazinane Chemical compound [O-][N+](=O)N1CN([N+]([O-])=O)CN([N+]([O-])=O)C1 XTFIVUDBNACUBN-UHFFFAOYSA-N 0.000 description 8
- 239000000470 constituent Substances 0.000 description 8
- 239000002904 solvent Substances 0.000 description 7
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 6
- DIXBSCZRIZDQGC-UHFFFAOYSA-N diaziridine Chemical compound C1NN1 DIXBSCZRIZDQGC-UHFFFAOYSA-N 0.000 description 6
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 5
- 230000007423 decrease Effects 0.000 description 5
- ZIBGPFATKBEMQZ-UHFFFAOYSA-N triethylene glycol Chemical compound OCCOCCOCCO ZIBGPFATKBEMQZ-UHFFFAOYSA-N 0.000 description 5
- NLZBZABIJAZRIZ-UHFFFAOYSA-N (6-hydrazinyl-1,2,4,5-tetrazin-3-yl)hydrazine Chemical compound NNC1=NN=C(NN)N=N1 NLZBZABIJAZRIZ-UHFFFAOYSA-N 0.000 description 4
- 238000005520 cutting process Methods 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 4
- 239000002131 composite material Substances 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 239000011159 matrix material Substances 0.000 description 3
- 125000001477 organic nitrogen group Chemical group 0.000 description 3
- 101710180978 D-alanine aminotransferase Proteins 0.000 description 2
- KAESVJOAVNADME-UHFFFAOYSA-N Pyrrole Chemical compound C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 239000011230 binding agent Substances 0.000 description 2
- 239000000567 combustion gas Substances 0.000 description 2
- 238000000265 homogenisation Methods 0.000 description 2
- 238000004519 manufacturing process Methods 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 239000000725 suspension Substances 0.000 description 2
- CZGWDPMDAIPURF-UHFFFAOYSA-N (4,6-dihydrazinyl-1,3,5-triazin-2-yl)hydrazine Chemical compound NNC1=NC(NN)=NC(NN)=N1 CZGWDPMDAIPURF-UHFFFAOYSA-N 0.000 description 1
- PZJFUNZDCRKXPZ-UHFFFAOYSA-N 2,5-dihydro-1h-tetrazole Chemical compound C1NNN=N1 PZJFUNZDCRKXPZ-UHFFFAOYSA-N 0.000 description 1
- NOWKCMXCCJGMRR-UHFFFAOYSA-N Aziridine Chemical compound C1CN1 NOWKCMXCCJGMRR-UHFFFAOYSA-N 0.000 description 1
- NDYLCHGXSQOGMS-UHFFFAOYSA-N CL-20 Chemical compound [O-][N+](=O)N1C2N([N+]([O-])=O)C3N([N+](=O)[O-])C2N([N+]([O-])=O)C2N([N+]([O-])=O)C3N([N+]([O-])=O)C21 NDYLCHGXSQOGMS-UHFFFAOYSA-N 0.000 description 1
- 239000000028 HMX Substances 0.000 description 1
- FJWGYAHXMCUOOM-QHOUIDNNSA-N [(2s,3r,4s,5r,6r)-2-[(2r,3r,4s,5r,6s)-4,5-dinitrooxy-2-(nitrooxymethyl)-6-[(2r,3r,4s,5r,6s)-4,5,6-trinitrooxy-2-(nitrooxymethyl)oxan-3-yl]oxyoxan-3-yl]oxy-3,5-dinitrooxy-6-(nitrooxymethyl)oxan-4-yl] nitrate Chemical compound O([C@@H]1O[C@@H]([C@H]([C@H](O[N+]([O-])=O)[C@H]1O[N+]([O-])=O)O[C@H]1[C@@H]([C@@H](O[N+]([O-])=O)[C@H](O[N+]([O-])=O)[C@@H](CO[N+]([O-])=O)O1)O[N+]([O-])=O)CO[N+](=O)[O-])[C@@H]1[C@@H](CO[N+]([O-])=O)O[C@@H](O[N+]([O-])=O)[C@H](O[N+]([O-])=O)[C@H]1O[N+]([O-])=O FJWGYAHXMCUOOM-QHOUIDNNSA-N 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 230000000772 anti-erosive effect Effects 0.000 description 1
- 150000001540 azides Chemical class 0.000 description 1
- 238000003490 calendering Methods 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- LYAGTVMJGHTIDH-UHFFFAOYSA-N diethylene glycol dinitrate Chemical compound [O-][N+](=O)OCCOCCO[N+]([O-])=O LYAGTVMJGHTIDH-UHFFFAOYSA-N 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 230000005611 electricity Effects 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 239000002360 explosive Substances 0.000 description 1
- 238000001125 extrusion Methods 0.000 description 1
- 239000004744 fabric Substances 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 229910002804 graphite Inorganic materials 0.000 description 1
- 239000010439 graphite Substances 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 238000004898 kneading Methods 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 150000002829 nitrogen Chemical class 0.000 description 1
- UZGLIIJVICEWHF-UHFFFAOYSA-N octogen Chemical compound [O-][N+](=O)N1CN([N+]([O-])=O)CN([N+]([O-])=O)CN([N+]([O-])=O)C1 UZGLIIJVICEWHF-UHFFFAOYSA-N 0.000 description 1
- 150000002897 organic nitrogen compounds Chemical class 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 229920002635 polyurethane Polymers 0.000 description 1
- 239000004814 polyurethane Substances 0.000 description 1
- 229920003225 polyurethane elastomer Polymers 0.000 description 1
- XAEFZNCEHLXOMS-UHFFFAOYSA-M potassium benzoate Chemical compound [K+].[O-]C(=O)C1=CC=CC=C1 XAEFZNCEHLXOMS-UHFFFAOYSA-M 0.000 description 1
- 238000005096 rolling process Methods 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 230000003068 static effect Effects 0.000 description 1
- 238000010025 steaming Methods 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- WEWFIUPOLKEEJP-UHFFFAOYSA-N triazine-4,6-diamine Chemical compound NC1=CC(N)=NN=N1 WEWFIUPOLKEEJP-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C06—EXPLOSIVES; MATCHES
- C06B—EXPLOSIVES OR THERMIC COMPOSITIONS; MANUFACTURE THEREOF; USE OF SINGLE SUBSTANCES AS EXPLOSIVES
- C06B25/00—Compositions containing a nitrated organic compound
- C06B25/34—Compositions containing a nitrated organic compound the compound being a nitrated acyclic, alicyclic or heterocyclic amine
-
- C—CHEMISTRY; METALLURGY
- C06—EXPLOSIVES; MATCHES
- C06B—EXPLOSIVES OR THERMIC COMPOSITIONS; MANUFACTURE THEREOF; USE OF SINGLE SUBSTANCES AS EXPLOSIVES
- C06B23/00—Compositions characterised by non-explosive or non-thermic constituents
- C06B23/04—Compositions characterised by non-explosive or non-thermic constituents for cooling the explosion gases including antifouling and flash suppressing agents
-
- C—CHEMISTRY; METALLURGY
- C06—EXPLOSIVES; MATCHES
- C06B—EXPLOSIVES OR THERMIC COMPOSITIONS; MANUFACTURE THEREOF; USE OF SINGLE SUBSTANCES AS EXPLOSIVES
- C06B25/00—Compositions containing a nitrated organic compound
- C06B25/18—Compositions containing a nitrated organic compound the compound being nitrocellulose present as 10% or more by weight of the total composition
- C06B25/24—Compositions containing a nitrated organic compound the compound being nitrocellulose present as 10% or more by weight of the total composition with nitroglycerine
- C06B25/26—Compositions containing a nitrated organic compound the compound being nitrocellulose present as 10% or more by weight of the total composition with nitroglycerine with an organic non-explosive or an organic non-thermic component
Definitions
- the present invention is in the field general of tube weapons, and concerns more particularly new powder compositions propellants intended to ensure the propulsion of a projectile with this type of weapon.
- Tube weapons which are well known to man of the trade, make it possible to launch a projectile, in general a bullet or a shell, in one direction determined, with a determined force obtained by the rapid production of a large quantity of resulting gas from the combustion of a propellant powder.
- tube weapons As examples of tube weapons one can cite the handguns such as pistols and revolvers, long guns such as rifles and shotguns, and lookout weapons such as machine guns and cannons.
- combustion temperature the factor predominant of the erosivity of a powder is the combustion temperature. The lower this temperature the higher the powder is erosive, whatever the composition of the powder. The determination of the combustion temperature therefore makes it easy to compare the erosivity of various powders.
- US patent 4,098,193 proposes, to decrease the erosion of the wall of the weapon tube, to incorporate, in ammunition containing a propellant powder double-base, an anti-erosion sleeve between the envelope and propellant powder.
- This anti-erosion sleeve is consisting of a fabric support impregnated with a compound decomposable organic rich in nitrogen such as tetrazole, a polyvinyltetrazole or an azide organic.
- the present invention provides a solution to this problem.
- New propellant powder compositions for tube weapons according to the invention are characterized in that this other organic compound nitrogen energy consists only of carbon, nitrogen and hydrogen and has at least one nitrogen heterocycle.
- the enthalpy of formation of this nitrogen organic energy compound is greater than + 100cal / g, better still greater than + 400cal / g, better even more than + 600cal / g and even better greater than + 900cal / g.
- This nitrogenous energetic organic compound which, at the room temperature, about 20 ° C, may occur both in the solid state and in the liquid state, is preference chosen from the group made up of diaziridines, triazoles, triazines, tetrazoles and tetrazines.
- diaminotriazine dimethylbistetrazole, trihydrazinotriazine, 5-amino-tetrazole, aminotetrazolotetrazine, 3,3'-azobis (6-amino-s-tetrazine) and 3,6-dihydrazino-s-tetrazine.
- the weight content of the nitrogenous organic energy compound is between 5% and 50% relative to the weight total of the composition, preferably between 10% and 30 %.
- the temperature of combustion of the powder therefore its erosivity, decreases all the more so as the weight content of compound organic nitrogen energy increases, and secondly that the strength of the powder increases, reaches a maximum, then generally decreases, when the content is increased by weight of energetic nitrogenous organic compound.
- the overall weight content of nitrocellulose and nitroglycerin is between 50% and 95% by relative to the total weight of the composition.
- the nitrocellulose / nitroglycerin weight ratio is preferably between 2.5 and 1.
- any nitrocellulose can be used usually used in double-base powders.
- compositions according to the invention can also understand the additives usually present in double-base powders, for example a stabilizer like centrality, graphite to make powders that conduct static electricity and facilitate loading and / or an anti-glare agent such as a potassium salt.
- a stabilizer like centrality, graphite to make powders that conduct static electricity and facilitate loading
- an anti-glare agent such as a potassium salt.
- propellant powder compositions include also a powdery nitramine filler, preferably hexogen, octogen or hexanitrohexaazaisowurtzitane.
- the weight content of nitramine powder charge is preferably included between 5% and 40% relative to the total weight of the composition.
- the new powder compositions for handguns tube according to the invention can be obtained according to processes analogous to those used in the state of the technique for obtaining double-base nitrocellulose / nitroglycerin powders or triple base powders nitrocellulose / nitroglycerin / nitroguanidine, optionally "doped" with a nitramine charge.
- a first process well known to those skilled in the art is a solventless process. According to this process, first prepare a patty from a aqueous nitrocellulose suspension in which add an aqueous nitroglycerin emulsion, suspension which is filtered after homogenization with press dough.
- This cake is moistened for reasons of security for storage and transport.
- the moist cake (35 to 40% water in general) is first wrung to bring the water content to around 20%, then underwent a humidity kneading in a paddle mixer to better homogenize the water in the product.
- the powder is then shaped by extrusion to press and cutting or by calendering and cutting according to the desired shape and dimensions.
- the nitrogenous energetic organic compound only consisting of carbon, nitrogen and hydrogen and containing a nitrogen heterocycle is added to the paddle mixer as other constituents than nitrocellulose and nitroglycerin.
- a second process well known to those skilled in the art is a semi-solvent process. According to this process, first mix nitrocellulose in a mixer dehydrated in alcohol with dissolved nitroglycerin in acetone as well as with the other constituents of the powder.
- the powder is then shaped according to the same operations than those mentioned above for the process without solvent, then the solvents are removed by steaming.
- the nitrogenous energetic organic compound is added to the mixer as other constituents of the powder.
- the present invention also relates to a process for reducing the erosive effect and increase the strength of a propellant powder for weapons with nitrocellulose and nitroglycerin-based tubes.
- composition of this powder an organic compound made up only of carbon, nitrogen and hydrogen and comprising at least minus one nitrogen heterocycle.
- the powder samples were obtained according to the aforementioned semi-solvent process.
- the constituents are introduced into a mixer horizontal, first of all nitrocellulose (13.15% nitrogen) in the form roughened to 25% ethanol then the nitroglycerin phlegmatized with 30% acetone. We add then gradually the other constituents.
- the level of acetone and ethanol is adjusted for get a good homogenization of the dough.
- the rate watering is variable depending on the composition.
- the mixing time is approximately 3 hours.
- the dough is then spun in a press pot.
- the die geometry is a 7-hole die with a 2.7 mm outer diameter, a hole diameter of 0.3 mm and a web of 0.45 mm.
- the spun strands are left at temperature ambient (around 20 ° C) to allow a slight evaporation of the solvents and obtaining a consistency favorable to cutting.
- the strands are then cut with a cutting machine sash to a length such that the ratio length / diameter is equal to 1.1.
- the powder grains are then dried in an oven several days at a temperature between 40 ° C and 70 ° C.
- the powder combustion temperatures were determined by calculation, according to a well-known method of the skilled person.
- the strength of the powders was determined according to the examples, either by calculation or from measurements experimental manometric bomb shots according to methods also well known to those skilled in the art.
- Examples 1 to 23 Propellant powder compositions consisting of nitrocellulose / nitroglycerin 60/40 by weight and of a nitrogenous organic compound solely consisting of C, H, N and comprising at least one nitrogenous heterocycle, at various weight contents
- compositions if we denote by x the weight content in the compound compositions organic nitrogen only consisting of C, H, N and comprising at least one nitrogen heterocycle, the content nitrocellulose by weight is 0.6 (100-x) and that by 0.4 nitroglycerin (100-x).
- the nitrogenous organic compound is dimethylbistetrazole (DMBT), of crude formula C 4 H 6 N 8 , the heat of formation of which is + 750 cal / g.
- DMBT dimethylbistetrazole
- the nitrogenous organic compound is 5-aminotetrazole (5AT), with the crude formula CH 3 N 5 , the heat of formation of which is +584 cal / g.
- the nitrogenous organic compound is 3,6-dihydrazino-s-tetrazine (DHT), of crude formula C 2 H 6 N 8 , the heat of formation of which is +901 cal / g.
- DHT 3,6-dihydrazino-s-tetrazine
- the nitrogenous organic compound is diaziridine of crude formula C 10 H 14 N 4 , the heat of formation of which is +1087 cal / g, of structural formula:
- the organic nitrogen compound is 3,3'-azobis (6-amino-s-tetrazine) (DAAT), with the crude formula C 4 H 4 N 12 , the heat of formation of which is +937 cal / g.
- DAAT 3,3'-azobis (6-amino-s-tetrazine)
- the nitrogenous organic compound is aminotetrazolotetrazine (AAT), of crude formula C 2 H 2 N 8 , the heat of formation of which is +1290 cal / g.
- AAT aminotetrazolotetrazine
- the NGU does not make it possible to increase the strength of the compositions, only for NGU contents less than about 20%, the drop in erosivity is very minimal with maintenance of strength and that drop is more consistent for NGU contents between 30% and 50%, but the strength is then markedly decreased.
- Examples 24 to 39 - Propellant powder compositions consisting of 60% by weight of nitrocellulose, of nitroglycerin and of a nitrogenous organic compound consisting solely of C, H, N and comprising at least one nitrogenous heterocycle, with various weight contents
- compositions which all comprise 60% by weight of nitrocellulose, if we denote by x the weight content in the compound compositions organic nitrogen only consisting of C, H, N and comprising at least one nitrogen heterocycle, the content Nitroglycerin weight is (40-x).
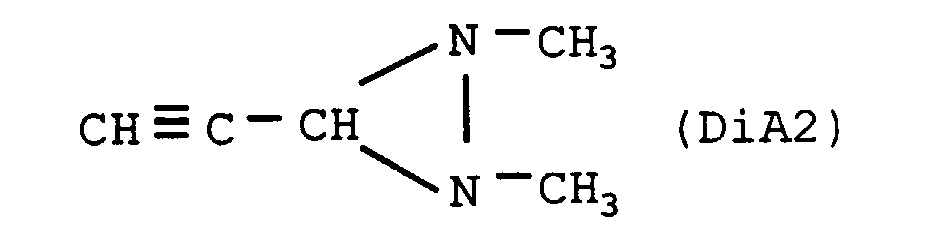
- the nitrogenous organic compound is diaziridine of crude formula C 5 H 8 N 2 , the heat of formation of which is +1082 cal / g, of structural formula:
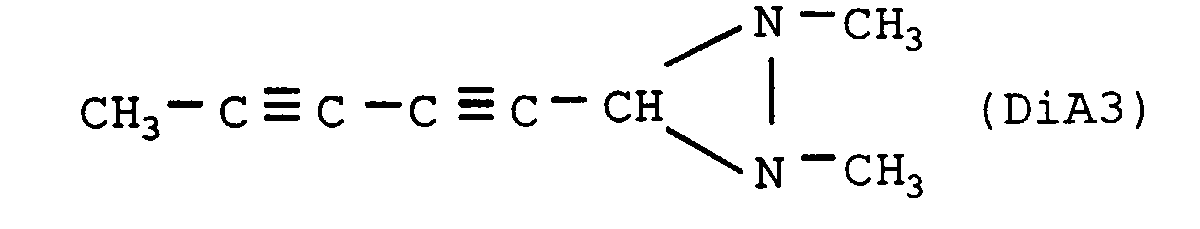
- the nitrogenous organic compound is diaziridine of crude formula C 8 H 10 N 2 , the heat of formation of which is +1180 cal / g, of structural formula:
- the nitrogenous organic compound is diaziridine of crude formula C 7 H 8 N 2 , the heat of formation of which is +1374 cal / g, of structural formula:
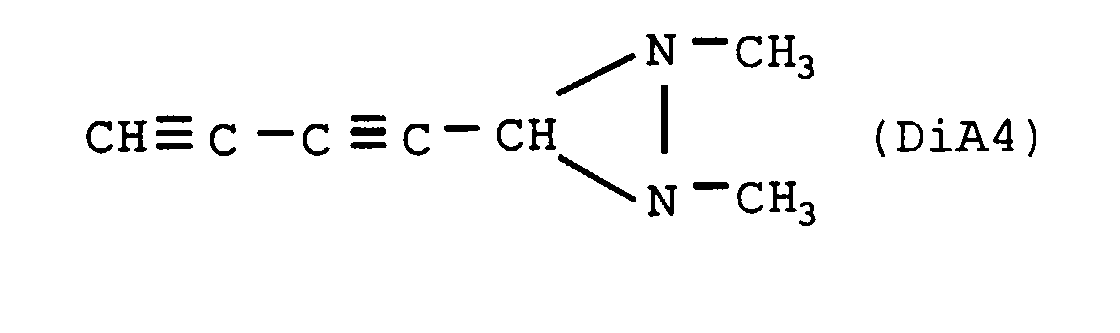
- the nitrogenous organic compound is diaziridine of crude formula C 6 H 12 N 2 , the heat of formation of which is +517 cal / g, of structural formula:
- the nitrogenous organic compound is diaziridine of crude formula C5H10N8, the heat of formation of which is +1061 cal / g, of structural formula:
- compositions if we denote by x the weight content in the compound compositions organic nitrogen only consisting of C, H, N and comprising at least one nitrogen heterocycle, the content nitrocellulose weight is 0.6 (75 - x) and that in 0.4 nitroglycerin (75 - x).
- the organic compound nitrogen is the dimethylbistetrazole (DMBT) already used for examples 1 to 4.
- DMBT dimethylbistetrazole
- the organic compound nitrogen is 3,6-dihydrazino-s-tetrazine (DHT) already used for examples 9 to 13.
- DHT 3,6-dihydrazino-s-tetrazine
- these comparative examples G to O, P 1 to P 9 and Q 1 to Q 7 are not part of the invention. They were carried out with the aim of showing that the double technical effect observed according to the invention, namely reduction in erosivity and increase in strength, occurs surprisingly only with double-base nitrocellulose / nitroglycerin powders and is not observed for other types of powders, in particular for single-base nitrocellulosic powders, for double-base powders in which the nitroglycerin is replaced by another nitrated oil, as well as for composite powders with inert binder.
- the powder does not contain than these 2 constituents.
- the powder also contains DMBT in various strengths by weight, as for examples 1 to 4 according to the invention.
- the powder also contains 5AT, as for Examples 5 to 8 according to the invention.
- the powder contains only nitrocellulose.
- Table 5 specifies, for these comparative examples P 1 to P 9 , the combustion strength and temperature of the compositions.
- Comparative example DMBT (%) 5AT (%) Force (MJ / kg) Combustion temperature (K) P 1 - - 1.10 (m) 3,324 P 2 5 - 1.09 (m) 3,185 P 3 10 - 1.07 (m) 3,041 P 4 20 - 1.04 (m) 2,750 P 5 30 - 0.99 (m) 2,475 P 6 - 5 1.09 (m) 3,220 P 7 - 10 1.08 (m) 3,113 F 8 - 20 1.05 (m) 2,895 P 9 - 30 1.02 (m) 2,678
- the powder consists of 10% by weight of inert matrix and 90% by weight of hexogen.
- the powder consists of 10% by weight of inert matrix, x% by weight of DMBT or 5AT and (90-x)% by weight of hexogen.
- Comparative example DMBT (%) 5AT (%) Force (MJ / kg) Combustion temperature (K) Q 1 - - 1.31 (m) 3,440 Q 2 5 - 1.28 (m) 3,265 Q 3 10 - 1.25 (m) 3,086 Q 4 20 - 1.17 (m) 2,735 Q 5 - 5 1.28 (m) 3,298 Q 6 - 10 1.25 (m) 3,153 Q 7 - 20 1.19 (m) 2,859
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Emergency Medicine (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Description
| Exemple N° | DMBT (%) | 5AT (%) | DHT (%) | DiA1 (%) | DAAT (%) | AAT (%) | NGU (%) | Force (MJ/kg) | Température de combustion (K) |
| Comparatif A | - | - | - | - | - | - | - | 1,17 (m) (c) | 3 884 |
| 1 | 4,8 | - | - | - | - | - | - | 1,19 (m) | 3 794 |
| 2 | 9,1 | - | - | - | - | - | - | 1,20 (m) | 3 685 |
| 3 | 16,7 | - | - | - | - | - | - | 1,19 (m) | 3 453 |
| 4 | 23,1 | - | - | - | - | - | - | 1,18 (m) | 3 233 |
| 5 | - | 4,8 | - | - | - | - | - | 1,18 (m) | 3 809 |
| 6 | - | 9,1 | - | - | - | - | - | 1,19 (m) | 3 727 |
| 7 | - | 16,7 | - | - | - | - | - | 1,19 (m) | 3 553 |
| 8 | - | 23,1 | - | - | - | - | - | 1,18 (m) | 3 387 |
| 9 | - | - | 9,1 | - | - | - | - | 1,22 (c) | 3 763 |
| 10 | - | - | 20,0 | - | - | - | - | 1,24 (c) | 3 556 |
| 11 | - | - | 28,6 | - | - | - | - | 1,25 (c) | 3 371 |
| 12 | - | - | 35,5 | - | - | - | - | 1,25 (c) | 3 216 |
| 13 | - | - | 41,2 | - | - | - | - | 1,25 (c) | 3 087 |
| 14 | - | - | - | 4,8 | - | - | - | 1,22 (c) | 3 710 |
| 15 | - | - | - | 9,1 | - | - | - | 1,23 (c) | 3 483 |
| 16 | - | - | - | 16,7 | - | - | - | 1,21 (c) | 3 047 |
| 17 | - | - | - | 23,1 | - | - | - | 1,17 (c) | 2 700 |
| 18 | - | - | - | - | 9,1 | - | - | 1,20 (c) | 3 791 |
| 19 | - | - | - | - | 25,9 | - | - | 1,21 (c) | 3 510 |
| 20 | - | - | - | - | - | 13,0 | - | 1,23 (c) | 3 859 |
| 21 | - | - | - | - | - | 23,1 | - | 1,27 (c) | 3 818 |
| 22 | - | - | - | - | - | 35,5 | - | 1,31 (c) | 3 746 |
| 23 | - | - | - | - | - | 50,0 | - | 1,36 (c) | 3 640 |
| Comparatif B | - | - | - | - | - | - | 9,1 | 1,17 (m) | 3 781 |
| Comparatif C | - | - | - | - | - | - | 16,7 | 1,17 (m) | 3 685 |
| Comparatif D | - | - | - | - | - | - | 30,0 | 1,15 (m) | 3 492 |
| Comparatif E | - | - | - | - | - | - | 50,0 | 1,11 (m) | 3 170 |
| Exemple N° | DiA2 (%) | DiA3 (%) | DiA4 (%) | DiA5 (%) | DiA6 (%) | Force (MJ/kg) | Température de combustion (K) |
| Comparatif A | - | - | - | - | - | 1,17 (m) (c) | 3 884 |
| 24 | 5 | - | - | - | - | 1,21 (c) | 3 645 |
| 25 | 10 | - | - | - | - | 1,21 (c) | 3 312 |
| 26 | 15 | - | - | - | - | 1,18 (c) | 2 962 |
| 27 | - | 5 | - | - | - | 1,21 (c) | 3 638 |
| 28 | - | 10 | - | - | - | 1,21 (c) | 3 290 |
| 29 | - | 15 | - | - | - | 1,17 (c) | 2 924 |
| 30 | - | - | 5 | - | - | 1,21 (c) | 3 638 |
| 31 | - | - | 10 | - | - | 1,21 (c) | 3 290 |
| 32 | - | - | 15 | - | - | 1,17 (c) | 2 924 |
| 33 | - | - | - | 5 | - | 1,20 (c) | 3 547 |
| 34 | - | - | - | 10 | - | 1,17 (c) | 3 104 |
| 35 | - | - | - | - | 5 | 1,21 (c) | 3 755 |
| 36 | - | - | - | - | 10 | 1,22 (c) | 3 573 |
| 37 | - | - | - | - | 15 | 1,22 (c) | 3 365 |
| 38 | - | - | - | - | 20 | 1,21 (c) | 3 149 |
| 39 | - | - | - | - | 25 | 1,19 (c) | 2 931 |
| Exemple N° | DMBT (%) | DHT (%) | Force (MJ/kg) | Température de combustion (K) |
| Comparatif F | - | - | 1,22 (m) (c) | 3 932 |
| 40 | 10 | - | 1,25 (m) | 3 706 |
| 41 | 20 | - | 1,24 (m) | 3 388 |
| 42 | - | 10 | 1,27 (c) | 3 794 |
| 43 | - | 20 | 1,30 (c) | 3 603 |
| 44 | - | 30 | 1,31 (c) | 3 388 |
| Exemple Comparatif | DMBT (%) | 5AT (%) | Force (MJ/kg) | Température de combustion (K) |
| G | - | - | 1,06 (m) | 2 764 |
| H | 4,8 | - | 1,04 (m) | 2 660 |
| I | 9,1 | - | 1,02 (m) | 2 566 |
| J | 13,0 | - | 1,01 (m) | 2 482 |
| K | 16,7 | - | 0,99 (m) | 2 410 |
| L | - | 4,8 | 1,04 (m) | 2 692 |
| M | - | 9,1 | 1,03 (m) | 2 626 |
| N | - | 13,0 | 1,02 (m) | 2 567 |
| O | - | 16,7 | 1,01 (m) | 2 513 |
| Exemple Comparatif | DMBT (%) | 5AT (%) | Force (MJ/kg) | Température de combustion (K) |
| P1 | - | - | 1,10 (m) | 3 324 |
| P2 | 5 | - | 1,09 (m) | 3 185 |
| P3 | 10 | - | 1,07 (m) | 3 041 |
| P4 | 20 | - | 1,04 (m) | 2 750 |
| P5 | 30 | - | 0,99 (m) | 2 475 |
| P6 | - | 5 | 1,09 (m) | 3 220 |
| P7 | - | 10 | 1,08 (m) | 3 113 |
| F8 | - | 20 | 1,05 (m) | 2 895 |
| P9 | - | 30 | 1,02 (m) | 2 678 |
| Exemple Comparatif | DMBT (%) | 5AT (%) | Force (MJ/kg) | Température de combustion (K) |
| Q1 | - | - | 1,31 (m) | 3 440 |
| Q2 | 5 | - | 1,28 (m) | 3 265 |
| Q3 | 10 | - | 1,25 (m) | 3 086 |
| Q4 | 20 | - | 1,17 (m) | 2 735 |
| Q5 | - | 5 | 1,28 (m) | 3 298 |
| Q6 | - | 10 | 1,25 (m) | 3 153 |
| Q7 | - | 20 | 1,19 (m) | 2 859 |
Claims (8)
- Composition de poudre propulsive pour armes à tube, à base de nitrocellulose et de nitroglycérine et comprenant un autre composé organique énergétique azoté uniquement constitué de carbone, d'azote et d'hydrogène et comportant au moins un hétérocycle azoté, caractérisée en ce que la teneur pondérale dudit composé organique énergétique azoté est comprise entre 5 % et 50 % par rapport au poids total de la composition.
- Composition de poudre propulsive selon la revendication 1, caractérisée en ce que l'enthalpie de formation du composé organique énergétique azoté est supérieure à +100 cal/g.
- Composition de poudre propulsive selon la revendication 1, caractérisée en ce que le composé organique énergétique azoté est choisi dans le groupe constitué par les diaziridines, les triazoles, les triazines, les tétrazoles et les tétrazines.
- Composition de poudre propulsive selon la revendication 1, caractérisée en ce que la teneur pondérale globale nitrocellulose + nitroglycérine est comprise entre 50 % et 95 % par rapport au poids total de la composition.
- Composition de poudre propulsive selon la revendication 1, caractérisée en ce que le rapport pondéral nitrocellulose/nitroglycérine est compris entre 2,5 et 1.
- Composition de poudre propulsive selon la revendication 1, caractérisée en ce qu'elle comprend également une charge pulvérulente nitramine.
- Composition de poudre propulsive selon la revendication 6, caractérisée en ce que la teneur pondérale en charge pulvérulente nitramine est comprise entre 5 % et 40 % par rapport au poids total de la composition.
- Procédé permettant d'abaisser l'effet érosif et d'augmenter la force d'une poudre propulsive pour armes à tube à base de nitrocellulose et de nitroglycérine, caractérisé en ce qu'on incorpore, à la composition de cette poudre, à une teneur pondérale comprise entre 5 % et 50 % par rapport au poids total de la composition, un composé organique uniquement constitué de carbone, d'azote et d'hydrogène et comportant un hétérocycle azoté.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR0205904A FR2839715B1 (fr) | 2002-05-14 | 2002-05-14 | Compositions de poudres propulsives pour armes a tube a force elevee et effet erosif reduit |
| FR0205954 | 2002-05-14 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP1364931A2 true EP1364931A2 (fr) | 2003-11-26 |
| EP1364931A3 EP1364931A3 (fr) | 2010-10-06 |
| EP1364931B1 EP1364931B1 (fr) | 2014-07-09 |
Family
ID=8871535
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP03291108.3A Expired - Lifetime EP1364931B1 (fr) | 2002-05-14 | 2003-05-12 | Poudres propulsives pour armes à tube à force élevée et effet érosif réduit |
Country Status (3)
| Country | Link |
|---|---|
| EP (1) | EP1364931B1 (fr) |
| FR (1) | FR2839715B1 (fr) |
| NO (1) | NO20031945L (fr) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107151271A (zh) * | 2017-06-05 | 2017-09-12 | 常熟理工学院 | 一种高能低爆温硝化棉的制备方法 |
| US9885550B1 (en) | 2014-08-29 | 2018-02-06 | Orbital Atk, Inc. | Methods of preparing nitrocelluse based propellants and propellants made therefrom |
| US10066911B1 (en) | 2014-08-29 | 2018-09-04 | Orbital Atk, Inc. | Methods of preparing nitrocelluse based propellants and propellants made therefrom |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4098193A (en) | 1976-09-08 | 1978-07-04 | The United States Of America As Represented By The Secretary Of The Army | Wear and corrosion reducing additive for gun propellants |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE201215C (fr) * | ||||
| GB616898A (en) * | 1946-09-18 | 1949-01-28 | Stuart Gordon | Improvements in or relating to the production of "flashless" propellent powders |
| US2480852A (en) * | 1947-03-10 | 1949-09-06 | George C Hale | Propellent powders |
| US3073731A (en) * | 1956-03-29 | 1963-01-15 | Cohen Joseph | Plasticizing agents for nitrocellulose |
| FR2165093A5 (fr) * | 1971-12-17 | 1973-08-03 | Poudres & Explosifs Ste Nale | |
| DE2900020C2 (de) * | 1979-01-02 | 1982-12-09 | WNC-Nitrochemie GmbH, 8261 Aschau | Verfahren zur Herstellung eines mehrbasigen Treibladungspulvers |
| US4315785A (en) * | 1980-04-09 | 1982-02-16 | The United States Of America As Represented By The Secretary Of The Army | Propellant charge with reduced muzzle smoke and flash characteristics |
| US6231702B1 (en) * | 1998-02-20 | 2001-05-15 | Trw Inc. | Cool burning ammonium nitrate based gas generating composition |
| WO1999054270A1 (fr) * | 1998-04-20 | 1999-10-28 | Daicel Chemical Industries, Ltd. | PROCEDE DE REDUCTION DE NO¿x? |
| US6645325B1 (en) * | 1998-06-01 | 2003-11-11 | Russell R. Nickel | Fast-burning nitrocellulose compositions |
| US6692655B1 (en) * | 2000-03-10 | 2004-02-17 | Alliant Techsystems Inc. | Method of making multi-base propellants from pelletized nitrocellulose |
| US6984275B1 (en) * | 2003-02-12 | 2006-01-10 | The United States Of America As Represented By The Secretary Of The Navy | Reduced erosion additive for a propelling charge |
-
2002
- 2002-05-14 FR FR0205904A patent/FR2839715B1/fr not_active Expired - Fee Related
-
2003
- 2003-04-29 NO NO20031945A patent/NO20031945L/no not_active Application Discontinuation
- 2003-05-12 EP EP03291108.3A patent/EP1364931B1/fr not_active Expired - Lifetime
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4098193A (en) | 1976-09-08 | 1978-07-04 | The United States Of America As Represented By The Secretary Of The Army | Wear and corrosion reducing additive for gun propellants |
Non-Patent Citations (1)
| Title |
|---|
| J. QUINCHON: "les poudres pour armes, Technique et Documentation", 1986, LAVOISIER, article "les poudres, propergols et explosifs", pages: 18 - 20 |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9885550B1 (en) | 2014-08-29 | 2018-02-06 | Orbital Atk, Inc. | Methods of preparing nitrocelluse based propellants and propellants made therefrom |
| US10066911B1 (en) | 2014-08-29 | 2018-09-04 | Orbital Atk, Inc. | Methods of preparing nitrocelluse based propellants and propellants made therefrom |
| US10801819B1 (en) | 2014-08-29 | 2020-10-13 | Northrop Grumman Innovation Systems, Inc. | Methods of preparing nitrocellulose based propellants and propellants made therefrom |
| CN107151271A (zh) * | 2017-06-05 | 2017-09-12 | 常熟理工学院 | 一种高能低爆温硝化棉的制备方法 |
| CN107151271B (zh) * | 2017-06-05 | 2019-06-25 | 常熟理工学院 | 一种高能低爆温硝化棉的制备方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| NO20031945D0 (no) | 2003-04-29 |
| NO20031945L (no) | 2003-11-17 |
| EP1364931A3 (fr) | 2010-10-06 |
| FR2839715A1 (fr) | 2003-11-21 |
| FR2839715B1 (fr) | 2005-02-04 |
| EP1364931B1 (fr) | 2014-07-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0210881B1 (fr) | Utilisation du 5-oxo 3-nitro 1,2,4-triazole comme explosif secondaire et compositions pyrotechniques contenant du 5-oxo 3-nitro 1,2,4-triazole | |
| US8257522B2 (en) | Black powder substitutes for small caliber firearms | |
| FR2867469A1 (fr) | Compositions reactives contenant un metal, et leur procede de production | |
| AU2014321143B2 (en) | Burn rate modifier | |
| NO327395B1 (no) | Framgangsmate for tilvirkning av en-, to- eller trebasige drivladningskrutt for lopsammunisjon | |
| FR2488246A1 (fr) | Composition propulsive pour fusees et armes a feu | |
| EP0219431A1 (fr) | Procédé de fabrication d'une composition propulsive double-base à faible émission de lueur de flamme, et composition propulsive ainsi obtenue | |
| US10066911B1 (en) | Methods of preparing nitrocelluse based propellants and propellants made therefrom | |
| EP1364931B1 (fr) | Poudres propulsives pour armes à tube à force élevée et effet érosif réduit | |
| FR2854889A1 (fr) | Matiere energetique a liant thermoplastique | |
| BR112016006031B1 (pt) | Propelente, utilização de um composto, método de preparação de um propelente, e cartucho de munição | |
| BE1005565A7 (fr) | Matieres energetiques. | |
| US20020043316A1 (en) | Method for producing a functional, high-energy material | |
| BE528535A (fr) | ||
| AU2014242706B2 (en) | Insensitive munition propellants | |
| EP0013850B1 (fr) | Nouvelles poudres propulsives en grains à base de nitrocellulose, d'huile nitrée et de nitrate de polyvinyle et leur procédé de fabrication | |
| EP2978731A1 (fr) | Charges propulsives non phtalate | |
| US9395164B1 (en) | Methods of preparing nitrocelluse based propellants and propellants made therefrom | |
| EP3515881B1 (fr) | Produit pyrotechnique composite renfermant un agent anti-lueur de type sel de potassium. | |
| BE902208A (fr) | Composition propulsive a base de nitrocellulose. | |
| FR2930548A1 (fr) | Composition propulsive double-base a forte impulsion specifique et vitesse de combustion elevee et procede de fabrication | |
| BE484613A (fr) | ||
| FR2936795A1 (fr) | Compositions explosives denses, chargements explosifs denses et munitions les comprenant | |
| FR2621911A1 (fr) | Poudre propulsive a base de nitrocellulose et de dinitroglycolurile et son procede de fabrication | |
| FR3013706A1 (fr) | Produit pyrotechnique composite a liant reticule et son procede de preparation |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LI LU MC NL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL LT LV MK |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: SNPE MATERIAUX ENERGETIQUES |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LI LU MC NL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL LT LV MK |
|
| 17P | Request for examination filed |
Effective date: 20110404 |
|
| AKX | Designation fees paid |
Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LI LU MC NL PT RO SE SI SK TR |
|
| 17Q | First examination report despatched |
Effective date: 20110524 |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: EURENCO |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| INTG | Intention to grant announced |
Effective date: 20131119 |
|
| GRAJ | Information related to disapproval of communication of intention to grant by the applicant or resumption of examination proceedings by the epo deleted |
Free format text: ORIGINAL CODE: EPIDOSDIGR1 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| INTG | Intention to grant announced |
Effective date: 20140207 |
|
| INTC | Intention to grant announced (deleted) | ||
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: EURENCO |
|
| INTG | Intention to grant announced |
Effective date: 20140217 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LI LU MC NL PT RO SE SI SK TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP Ref country code: CH Ref legal event code: NV Representative=s name: BOVARD AG, CH Ref country code: AT Ref legal event code: REF Ref document number: 676520 Country of ref document: AT Kind code of ref document: T Effective date: 20140715 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: LANGUAGE OF EP DOCUMENT: FRENCH |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 60346448 Country of ref document: DE Effective date: 20140814 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MK05 Ref document number: 676520 Country of ref document: AT Kind code of ref document: T Effective date: 20140709 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: VDEP Effective date: 20140709 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140709 Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141010 Ref country code: ES Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140709 Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141009 Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141110 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140709 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140709 Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140709 Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140709 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 60346448 Country of ref document: DE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140709 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140709 Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140709 Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140709 Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140709 Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140709 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed |
Effective date: 20150410 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140709 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20150512 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150512 Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140709 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20150512 Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20150512 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 14 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 15 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT; INVALID AB INITIO Effective date: 20030512 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20150531 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: TR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140709 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 16 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20190510 Year of fee payment: 17 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20190520 Year of fee payment: 17 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 20190517 Year of fee payment: 17 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 60346448 Country of ref document: DE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20200531 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20200531 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20200531 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20201201 |