KR20140077658A - 포스포다이에스터라제-5 억제제를 포함하는 츄정 - Google Patents
포스포다이에스터라제-5 억제제를 포함하는 츄정 Download PDFInfo
- Publication number
- KR20140077658A KR20140077658A KR1020120146705A KR20120146705A KR20140077658A KR 20140077658 A KR20140077658 A KR 20140077658A KR 1020120146705 A KR1020120146705 A KR 1020120146705A KR 20120146705 A KR20120146705 A KR 20120146705A KR 20140077658 A KR20140077658 A KR 20140077658A
- Authority
- KR
- South Korea
- Prior art keywords
- disintegrant
- subject
- inhibitor
- disintegration
- crospovidone
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0053—Mouth and digestive tract, i.e. intraoral and peroral administration
- A61K9/0056—Mouth soluble or dispersible forms; Suckable, eatable, chewable coherent forms; Forms rapidly disintegrating in the mouth; Lozenges; Lollipops; Bite capsules; Baked products; Baits or other oral forms for animals
- A61K9/0058—Chewing gums
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/2027—Organic macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyvinyl pyrrolidone, poly(meth)acrylates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/519—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0053—Mouth and digestive tract, i.e. intraoral and peroral administration
- A61K9/0056—Mouth soluble or dispersible forms; Suckable, eatable, chewable coherent forms; Forms rapidly disintegrating in the mouth; Lozenges; Lollipops; Bite capsules; Baked products; Baits or other oral forms for animals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2013—Organic compounds, e.g. phospholipids, fats
- A61K9/2018—Sugars, or sugar alcohols, e.g. lactose, mannitol; Derivatives thereof, e.g. polysorbates
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Nutrition Science (AREA)
- Physiology (AREA)
- Zoology (AREA)
- Biophysics (AREA)
- Molecular Biology (AREA)
- Medicinal Preparation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
| 실시예 1 | 비교예 1 | 비교예 2 | |
| 피험자 1 | 1 | 4 | 3 |
| 피험자 2 | 1 | 5 | 4 |
| 피험자 3 | 0 | 3 | 2 |
| 피험자 4 | 0 | 4 | 2 |
| 피험자 5 | 1 | 4 | 3 |
| 피험자 6 | 1 | 5 | 3 |
| 피험자 7 | 1 | 4 | 2 |
| 피험자 8 | 2 | 5 | 3 |
| 피험자 9 | 1 | 4 | 3 |
| 피험자 10 | 1 | 5 | 4 |
| 평균 | 0.9 | 4.3 | 2.9 |
| 실시예 1 | 비교예 1 | 비교예 2 | |
| 1 | 12 | 10 | 32 |
| 2 | 11 | 10 | 31 |
| 3 | 12 | 11 | 33 |
| 4 | 13 | 10 | 29 |
| 5 | 10 | 12 | 30 |
| 6 | 13 | 12 | 27 |
| 평균 (초) | 11.8 | 10.8 | 30.3 |
| 실시예 1 | 실시예 2 | 실시예 3 | 실시예 4 | |
| 피험자 1 | 1 | 1 | 1 | 2 |
| 피험자 2 | 1 | 2 | 2 | 2 |
| 피험자 3 | 0 | 1 | 1 | 2 |
| 피험자 4 | 0 | 1 | 1 | 1 |
| 피험자 5 | 1 | 1 | 1 | 2 |
| 피험자 6 | 1 | 2 | 2 | 3 |
| 피험자 7 | 1 | 1 | 1 | 2 |
| 피험자 8 | 2 | 2 | 1 | 3 |
| 피험자 9 | 1 | 2 | 1 | 3 |
| 피험자 10 | 1 | 1 | 1 | 2 |
| 평균 | 0.9 | 1.4 | 1.2 | 2.2 |
| 실시예 1 | 실시예 2 | 실시예 3 | 실시예 4 | |
| 1 | 12 | 8 | 15 | 11 |
| 2 | 11 | 7 | 18 | 11 |
| 3 | 12 | 8 | 19 | 10 |
| 4 | 13 | 7 | 17 | 12 |
| 5 | 10 | 8 | 17 | 11 |
| 6 | 13 | 9 | 19 | 10 |
| 평균 (초) | 11.8 | 7.8 | 17.5 | 10.8 |
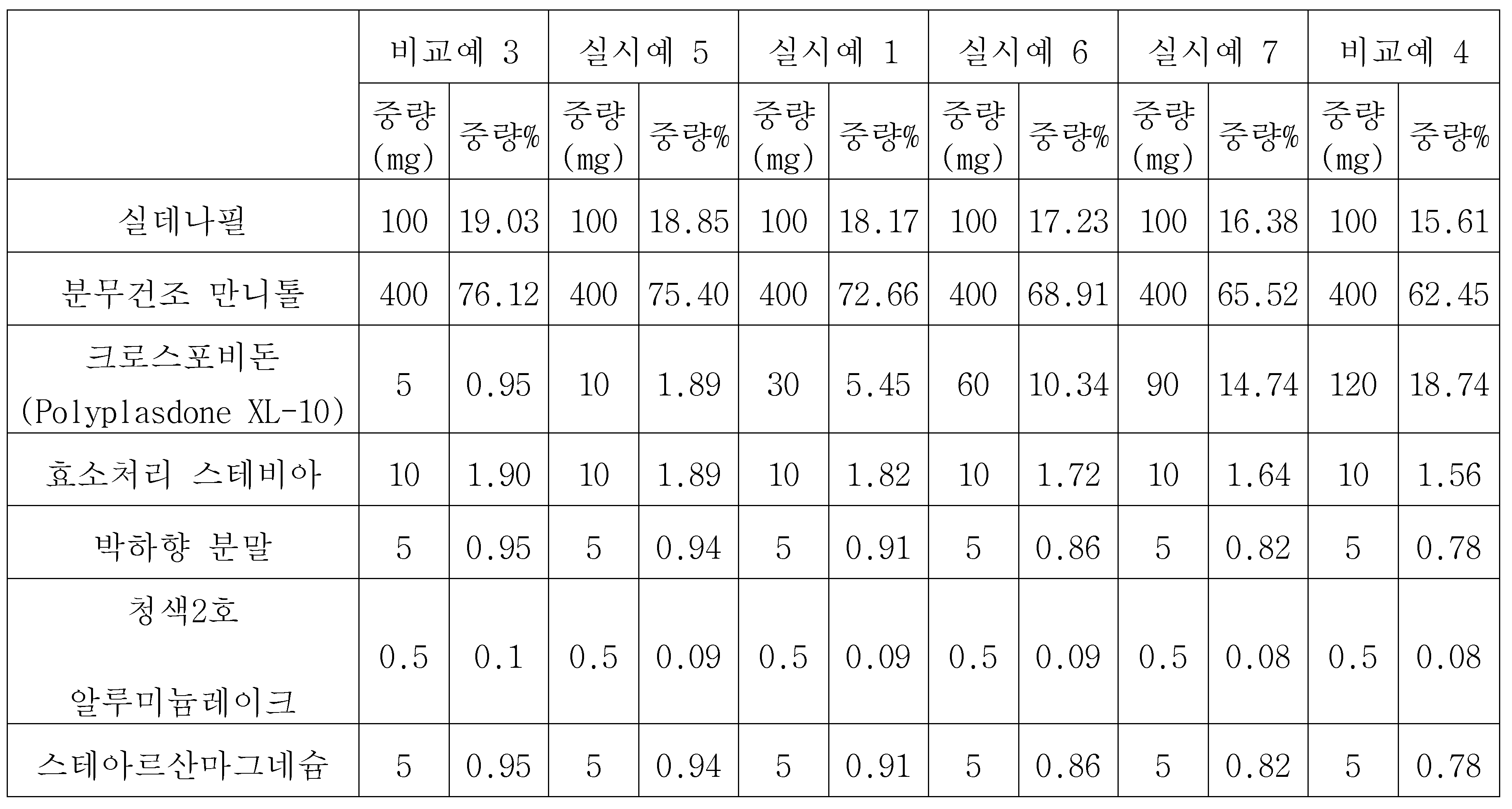
| 비교예 3 | 실시예 5 | 실시예 1 | 실시예 6 | 실시예 7 | 비교예 4 | |
| 피험자 1 | 3 | 2 | 1 | 2 | 3 | 3 |
| 피험자 2 | 2 | 1 | 1 | 2 | 3 | 4 |
| 피험자 3 | 1 | 1 | 0 | 1 | 2 | 3 |
| 피험자 4 | 1 | 0 | 0 | 2 | 2 | 4 |
| 피험자 5 | 2 | 1 | 1 | 2 | 3 | 4 |
| 피험자 6 | 2 | 2 | 1 | 2 | 3 | 4 |
| 피험자 7 | 3 | 0 | 1 | 2 | 3 | 3 |
| 피험자 8 | 2 | 1 | 2 | 3 | 3 | 4 |
| 피험자 9 | 2 | 1 | 1 | 2 | 2 | 3 |
| 피험자 10 | 3 | 1 | 1 | 3 | 3 | 4 |
| 평균 | 2.1 | 1.0 | 0.9 | 2.1 | 2.7 | 3.6 |
| 비교예 3 | 실시예 5 | 실시예 1 | 실시예 6 | 실시예 7 | 비교예 4 | |
| 1 | 35 | 16 | 12 | 10 | 8 | 6 |
| 2 | 33 | 18 | 11 | 7 | 8 | 5 |
| 3 | 34 | 20 | 12 | 11 | 6 | 6 |
| 4 | 34 | 23 | 13 | 12 | 9 | 7 |
| 5 | 39 | 22 | 10 | 9 | 10 | 7 |
| 6 | 43 | 19 | 13 | 10 | 7 | 6 |
| 평균 (초) | 36.3 | 19.7 | 11.8 | 9.8 | 8.0 | 6.2 |
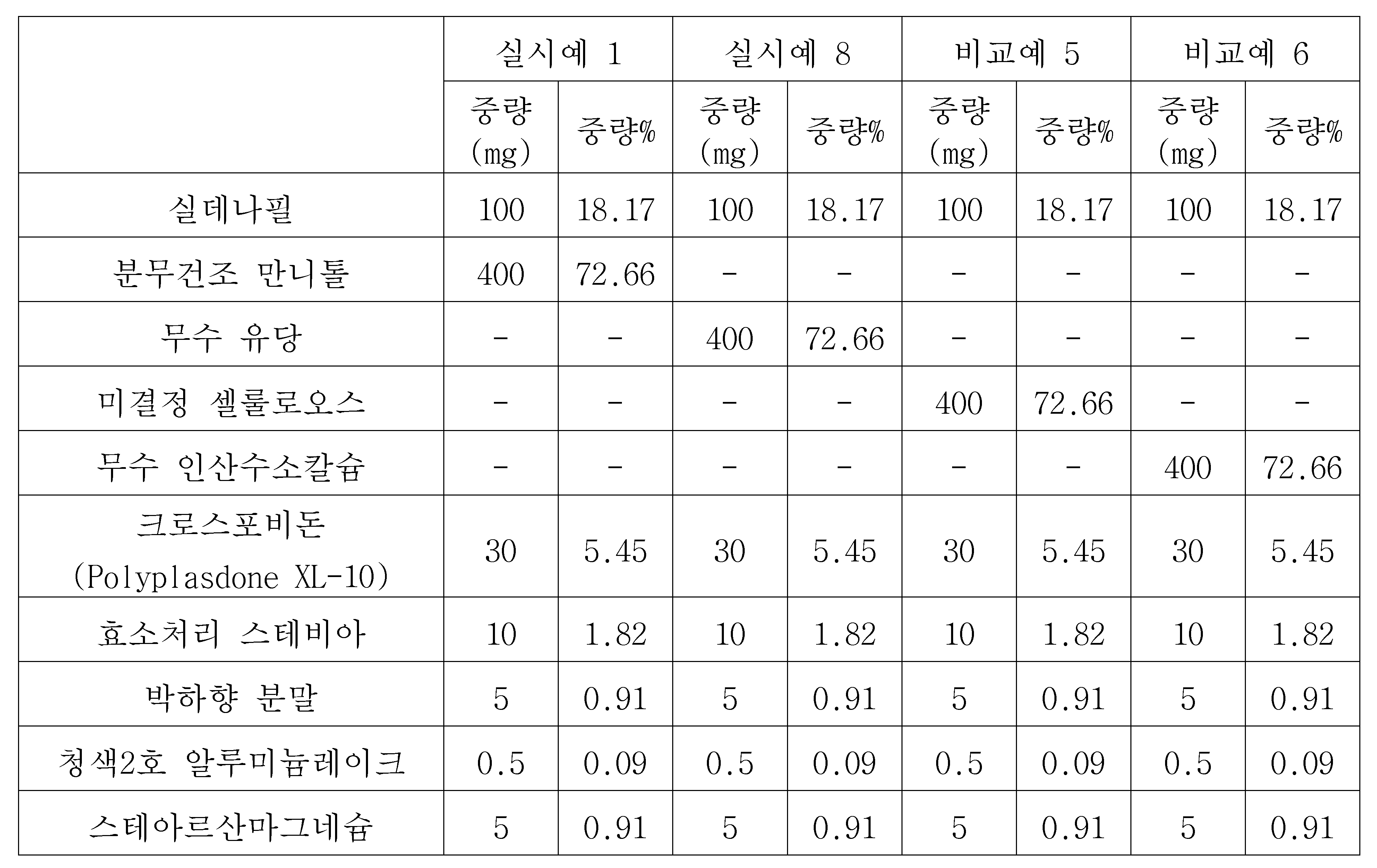
| 실시예 1 | 실시예 8 | 비교예 5 | 비교예 6 | |
| 피험자 1 | 1 | 1 | 3 | 4 |
| 피험자 2 | 1 | 2 | 4 | 4 |
| 피험자 3 | 0 | 1 | 3 | 3 |
| 피험자 4 | 0 | 1 | 3 | 3 |
| 피험자 5 | 1 | 2 | 3 | 4 |
| 피험자 6 | 1 | 2 | 4 | 4 |
| 피험자 7 | 1 | 1 | 3 | 3 |
| 피험자 8 | 2 | 2 | 4 | 4 |
| 피험자 9 | 1 | 1 | 3 | 3 |
| 피험자 10 | 1 | 2 | 3 | 4 |
| 평균 | 0.9 | 1.5 | 3.3 | 3.6 |
| 실시예 1 | 실시예 8 | 비교예 5 | 비교예 6 | |
| 1 | 12 | 12 | 39 | 42 |
| 2 | 11 | 14 | 42 | 45 |
| 3 | 12 | 15 | 38 | 39 |
| 4 | 13 | 14 | 38 | 46 |
| 5 | 10 | 12 | 44 | 48 |
| 6 | 13 | 15 | 36 | 41 |
| 평균 (초) | 11.8 | 13.7 | 39.5 | 43.5 |
Claims (11)
- PDE-5 억제제 또는 이의 약학적으로 허용 가능한 염; 및 팽창계수가 4 내지 8 L/kg인 붕해제를 포함하는 츄정.
- 제1항에 있어서,
상기 PDE-5 억제제가 실데나필 유리 염기인 츄정. - 제1항에 있어서,
상기 츄정이 당류, 당알콜류 또는 이들의 혼합물을 더 포함하는, 츄정. - 제3항에 있어서,
상기 당류가 유당, 글루코오스, 수크로오스, 과당, 레불로오스, 말토덱스트린 및 파라티노오스로 이루어진 군으로부터 선택되고, 상기 당알콜류가 만니톨, 솔비톨, 자일리톨 및 에리스리톨로 이루어진 군으로부터 선택되는, 츄정. - 제3항에 있어서,
상기 당알콜류가 분무건조 만니톨인 츄정. - 제3항에 있어서,
상기 당류, 당알콜류 또는 이들의 혼합물이 제제의 총 중량을 기준으로 10 내지 98 중량%의 양으로 포함되는 츄정. - 제1항에 있어서,
상기 붕해제의 입경이 5 내지 250 ㎛인 츄정. - 제1항에 있어서,
상기 붕해제가 제제의 총 중량을 기준으로 1.5 내지 15 중량%의 양으로 포함되는 츄정. - 제1항에 있어서,
상기 츄정의 평균 경도가 2 내지 17 kp 범위인 것을 특징으로 하는, 츄정. - 제3항에 있어서,
상기 츄정이 실데나필 유리 염기, 분무건조 만니톨 및 팽창계수가 4 내지 8 L/kg인 붕해제를 포함하는 츄정. - 제1항에 있어서,
상기 츄정이 감미제, 착향제, 착색제 및 활택제로 이루어진 군으로부터 선택되는 어느 하나 이상을 추가로 포함하는 츄정.
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020120146705A KR101953735B1 (ko) | 2012-12-14 | 2012-12-14 | 포스포다이에스터라제-5 억제제를 포함하는 츄정 |
| PCT/KR2013/010739 WO2014092358A1 (en) | 2012-12-14 | 2013-11-25 | Chewable tablet comprising a phosphodiesterase-5 inhibitor |
| ARP130104461A AR093707A1 (es) | 2012-12-14 | 2013-12-03 | Comprimido masticable que comprende un inhibidor de la fosfodiesterasa-5 |
| TW102146092A TW201427721A (zh) | 2012-12-14 | 2013-12-13 | 包含磷酸二酯酶-5抑制劑的咀嚼錠 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020120146705A KR101953735B1 (ko) | 2012-12-14 | 2012-12-14 | 포스포다이에스터라제-5 억제제를 포함하는 츄정 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20140077658A true KR20140077658A (ko) | 2014-06-24 |
| KR101953735B1 KR101953735B1 (ko) | 2019-03-04 |
Family
ID=50934596
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020120146705A Active KR101953735B1 (ko) | 2012-12-14 | 2012-12-14 | 포스포다이에스터라제-5 억제제를 포함하는 츄정 |
Country Status (4)
| Country | Link |
|---|---|
| KR (1) | KR101953735B1 (ko) |
| AR (1) | AR093707A1 (ko) |
| TW (1) | TW201427721A (ko) |
| WO (1) | WO2014092358A1 (ko) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN112826802B (zh) * | 2021-01-22 | 2021-09-28 | 上海普康药业有限公司 | 一种枸橼酸西地那非咀嚼片及其制备方法 |
| CN115337276B (zh) * | 2021-05-14 | 2024-02-09 | 南京科默生物医药有限公司 | 一种枸橼酸西地那非口腔崩解片剂的制备方法 |
Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR100262926B1 (ko) | 1993-06-09 | 2000-09-01 | 디. 제이. 우드, 무어 제임스 더블유 | 임포텐스 치료용 피라졸로피리미디논 |
| KR100353014B1 (ko) | 1998-11-11 | 2002-09-18 | 동아제약 주식회사 | 발기부전 치료에 효과를 갖는 피라졸로피리미디논 화합물 |
| KR100357411B1 (ko) | 1994-01-21 | 2003-02-05 | 아이코스 코포레이션 | 테트라시클릭유도체,제조방법및용도 |
| KR20030076051A (ko) * | 2002-03-22 | 2003-09-26 | 한미약품 주식회사 | 실데나필 젖산염의 속효제형 |
| KR100430355B1 (ko) | 1997-11-12 | 2004-05-04 | 바이엘 악티엔게젤샤프트 | 포스포디에스테라제 저해제로서의 2-페닐 치환된이미다조트리아지논 |
| JP2008127320A (ja) * | 2006-11-20 | 2008-06-05 | Zensei Yakuhin Kogyo Kk | 口腔内速崩壊性固形製剤 |
| WO2009074995A1 (en) * | 2007-12-10 | 2009-06-18 | Ajanta Pharma Limited | Taste masked chewable compositions of sildenafil citrate |
| WO2011030351A2 (en) * | 2009-09-03 | 2011-03-17 | Rubicon Research Private Limited | Taste - masked pharmaceutical compositions |
| JP2012007320A (ja) * | 2010-06-23 | 2012-01-12 | Hitachi Constr Mach Co Ltd | 作業機械 |
| WO2012120522A1 (en) * | 2011-03-04 | 2012-09-13 | Genepharm India Private Limited | A taste masked chewable tablet of sildenafil |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB0219516D0 (en) * | 2002-08-21 | 2002-10-02 | Phoqus Ltd | Fast dissolving and taste masked oral dosage form comprising sildenafil |
| EP2338474A1 (de) * | 2009-12-23 | 2011-06-29 | Ratiopharm GmbH | Oral dispersible Tablette enthaltend kompaktierte Sildenafil-Base |
-
2012
- 2012-12-14 KR KR1020120146705A patent/KR101953735B1/ko active Active
-
2013
- 2013-11-25 WO PCT/KR2013/010739 patent/WO2014092358A1/en not_active Ceased
- 2013-12-03 AR ARP130104461A patent/AR093707A1/es unknown
- 2013-12-13 TW TW102146092A patent/TW201427721A/zh unknown
Patent Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR100262926B1 (ko) | 1993-06-09 | 2000-09-01 | 디. 제이. 우드, 무어 제임스 더블유 | 임포텐스 치료용 피라졸로피리미디논 |
| KR100357411B1 (ko) | 1994-01-21 | 2003-02-05 | 아이코스 코포레이션 | 테트라시클릭유도체,제조방법및용도 |
| KR100430355B1 (ko) | 1997-11-12 | 2004-05-04 | 바이엘 악티엔게젤샤프트 | 포스포디에스테라제 저해제로서의 2-페닐 치환된이미다조트리아지논 |
| KR100353014B1 (ko) | 1998-11-11 | 2002-09-18 | 동아제약 주식회사 | 발기부전 치료에 효과를 갖는 피라졸로피리미디논 화합물 |
| KR20030076051A (ko) * | 2002-03-22 | 2003-09-26 | 한미약품 주식회사 | 실데나필 젖산염의 속효제형 |
| JP2008127320A (ja) * | 2006-11-20 | 2008-06-05 | Zensei Yakuhin Kogyo Kk | 口腔内速崩壊性固形製剤 |
| WO2009074995A1 (en) * | 2007-12-10 | 2009-06-18 | Ajanta Pharma Limited | Taste masked chewable compositions of sildenafil citrate |
| WO2011030351A2 (en) * | 2009-09-03 | 2011-03-17 | Rubicon Research Private Limited | Taste - masked pharmaceutical compositions |
| JP2012007320A (ja) * | 2010-06-23 | 2012-01-12 | Hitachi Constr Mach Co Ltd | 作業機械 |
| WO2012120522A1 (en) * | 2011-03-04 | 2012-09-13 | Genepharm India Private Limited | A taste masked chewable tablet of sildenafil |
Non-Patent Citations (1)
| Title |
|---|
| 삭제 |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2014092358A1 (en) | 2014-06-19 |
| KR101953735B1 (ko) | 2019-03-04 |
| AR093707A1 (es) | 2015-06-17 |
| TW201427721A (zh) | 2014-07-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7398374B2 (ja) | 固形経口ニコチン製剤 | |
| CN111447921B (zh) | 烟碱片剂 | |
| ES2393640T3 (es) | Comprimidos bucodisgregables | |
| EP2698145B1 (en) | Pharmaceutical Formulations comprising dapoxetine and a PDE5 inhibitor | |
| Bhatt | Mouth dissolving tablets challenges, preparation strategies with a special emphasis on Losartan potassium–A review | |
| US20040076665A1 (en) | Modified release oral dosage form | |
| JP2009114113A (ja) | 口腔内崩壊錠及びその製造方法 | |
| US20070082048A1 (en) | Sleep aid formulations | |
| WO2000020033A1 (en) | Tablets immediately disintegrating in the oral cavity | |
| RS54238B1 (sr) | Formulacije oralnog filma koje sadrže dapoksetin i tadalafil | |
| KR102239291B1 (ko) | 타다라필 또는 이의 약학적으로 허용가능한 염을 포함하는 저작정 제제 | |
| JP5656258B2 (ja) | ガランタミンを含有する口腔内崩壊錠剤 | |
| JP2008285434A (ja) | 口腔内速崩壊錠 | |
| WO2014027981A2 (en) | Effervescent tablet formulations of dapoxetine and a pde5 inhibitor | |
| KR101953735B1 (ko) | 포스포다이에스터라제-5 억제제를 포함하는 츄정 | |
| JP5848432B2 (ja) | ミルタザピンを含有する口腔内崩壊錠 | |
| KR101304343B1 (ko) | Pde-5 억제제의 경구용 속용제형 | |
| KR20130021888A (ko) | 실데나필 유리 염기를 포함하는 구강 붕해정 | |
| CN101203246B (zh) | 药物组合物 | |
| JP4944467B2 (ja) | 医薬用組成物 | |
| WO2014027978A1 (en) | Chewing gum formulations comprising dapoxetine and tadalafil | |
| Malik et al. | Recent patents on fast dissolving tablets-A Review | |
| HK40017837A (en) | Orally disintegrated tablet comprising carbamate compound | |
| HK1111619A (en) | Pharmaceutical composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0109 | Patent application |
St.27 status event code: A-0-1-A10-A12-nap-PA0109 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-3-3-R10-R18-oth-X000 |
|
| PG1501 | Laying open of application |
St.27 status event code: A-1-1-Q10-Q12-nap-PG1501 |
|
| R17-X000 | Change to representative recorded |
St.27 status event code: A-3-3-R10-R17-oth-X000 |
|
| R17-X000 | Change to representative recorded |
St.27 status event code: A-3-3-R10-R17-oth-X000 |
|
| PA0201 | Request for examination |
St.27 status event code: A-1-2-D10-D11-exm-PA0201 |
|
| R17-X000 | Change to representative recorded |
St.27 status event code: A-3-3-R10-R17-oth-X000 |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
St.27 status event code: A-1-2-D10-D21-exm-PE0902 |
|
| E13-X000 | Pre-grant limitation requested |
St.27 status event code: A-2-3-E10-E13-lim-X000 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
St.27 status event code: A-1-2-D10-D22-exm-PE0701 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
St.27 status event code: A-2-4-F10-F11-exm-PR0701 |
|
| PR1002 | Payment of registration fee |
St.27 status event code: A-2-2-U10-U11-oth-PR1002 Fee payment year number: 1 |
|
| PG1601 | Publication of registration |
St.27 status event code: A-4-4-Q10-Q13-nap-PG1601 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 4 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 5 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 6 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 7 |
|
| U11 | Full renewal or maintenance fee paid |
Free format text: ST27 STATUS EVENT CODE: A-4-4-U10-U11-OTH-PR1001 (AS PROVIDED BY THE NATIONAL OFFICE) Year of fee payment: 7 |