KR20180098705A - Vegf를 억제하는 안정하고 가용성인 항체 - Google Patents
Vegf를 억제하는 안정하고 가용성인 항체 Download PDFInfo
- Publication number
- KR20180098705A KR20180098705A KR1020187024733A KR20187024733A KR20180098705A KR 20180098705 A KR20180098705 A KR 20180098705A KR 1020187024733 A KR1020187024733 A KR 1020187024733A KR 20187024733 A KR20187024733 A KR 20187024733A KR 20180098705 A KR20180098705 A KR 20180098705A
- Authority
- KR
- South Korea
- Prior art keywords
- ser
- gly
- thr
- seq
- leu
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 0 C*CC(C(*C)C(*)CN=O)N=O Chemical compound C*CC(C(*C)C(*)CN=O)N=O 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H21/00—Compounds containing two or more mononucleotide units having separate phosphate or polyphosphate groups linked by saccharide radicals of nucleoside groups, e.g. nucleic acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/22—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans against growth factors ; against growth regulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
- A61K39/39533—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum against materials from animals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P15/00—Drugs for genital or sexual disorders; Contraceptives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P15/00—Drugs for genital or sexual disorders; Contraceptives
- A61P15/08—Drugs for genital or sexual disorders; Contraceptives for gonadal disorders or for enhancing fertility, e.g. inducers of ovulation or of spermatogenesis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/06—Antipsoriatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
- A61P27/06—Antiglaucoma agents or miotics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
- A61P33/14—Ectoparasiticides, e.g. scabicides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/10—Antioedematous agents; Diuretics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K1/00—General methods for the preparation of peptides, i.e. processes for the organic chemical preparation of peptides or proteins of any length
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies
- C07K16/46—Hybrid immunoglobulins
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K7/00—Peptides having 5 to 20 amino acids in a fully defined sequence; Derivatives thereof
- C07K7/04—Linear peptides containing only normal peptide links
- C07K7/06—Linear peptides containing only normal peptide links having 5 to 11 amino acids
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/63—Introduction of foreign genetic material using vectors; Vectors; Use of hosts therefor; Regulation of expression
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/24—Immunoglobulins specific features characterized by taxonomic origin containing regions, domains or residues from different species, e.g. chimeric, humanized or veneered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/31—Immunoglobulins specific features characterized by aspects of specificity or valency multispecific
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/54—F(ab')2
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/55—Fab or Fab'
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
- C07K2317/565—Complementarity determining region [CDR]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
- C07K2317/567—Framework region [FR]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/60—Immunoglobulins specific features characterized by non-natural combinations of immunoglobulin fragments
- C07K2317/62—Immunoglobulins specific features characterized by non-natural combinations of immunoglobulin fragments comprising only variable region components
- C07K2317/622—Single chain antibody (scFv)
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/73—Inducing cell death, e.g. apoptosis, necrosis or inhibition of cell proliferation
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Immunology (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Reproductive Health (AREA)
- Ophthalmology & Optometry (AREA)
- Rheumatology (AREA)
- Endocrinology (AREA)
- Microbiology (AREA)
- Biotechnology (AREA)
- Epidemiology (AREA)
- Mycology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Zoology (AREA)
- Dermatology (AREA)
- Vascular Medicine (AREA)
- Pain & Pain Management (AREA)
- Gynecology & Obstetrics (AREA)
- Pregnancy & Childbirth (AREA)
- Cardiology (AREA)
- Physical Education & Sports Medicine (AREA)
- Urology & Nephrology (AREA)
- Tropical Medicine & Parasitology (AREA)
Abstract
Description
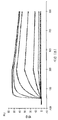
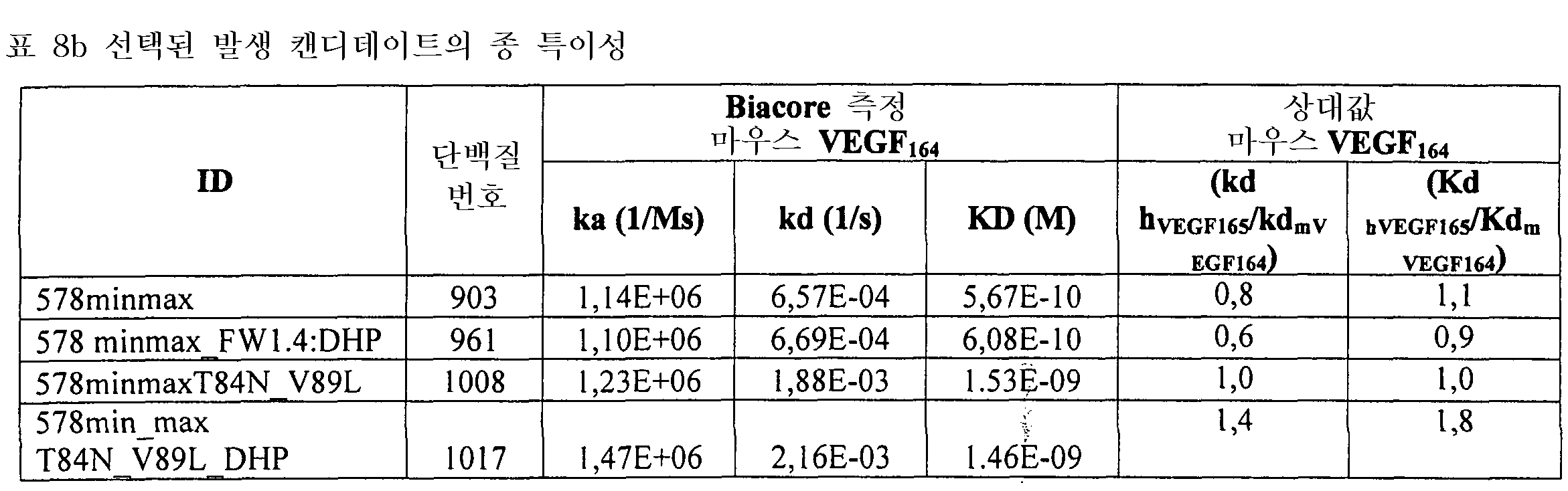
도 2는 578max의 사람, 마우스 및 래트 VEGF에 대한 결합 반응속도를 확인하여 종 특이성을 나타낸 것이다. 도 2a는 사람 VEGF165에 대해 얻어진 데이터를 나타낸다: Ka(1/Ms): 7,00E+05; SE (ka): 1.40E+03; kd(l/s): 3,07E-04; SE(kd): 8,50E-07; KD(M): 4,39E-IO. 도 2b는 마우스 VEGF164에 대해 얻어진 데이터를 나타낸다: Ka(1/Ms): l,03E+06; SE (ka): 2,30E+03; kd(l/s): 4,40E-04; SE(kd): 9,40E-07; KD(M): 4,29E-IO. 도 2c는 래트 VEGF164에 대해 얻어진 데이터를 나타낸다: Ka(1/Ms): 8,83E+05; SE (ka): 2,50E+03; kd(l/s): 5,28E-04; SE(kd): l,20E-06; KD(M): 5,98E-10.
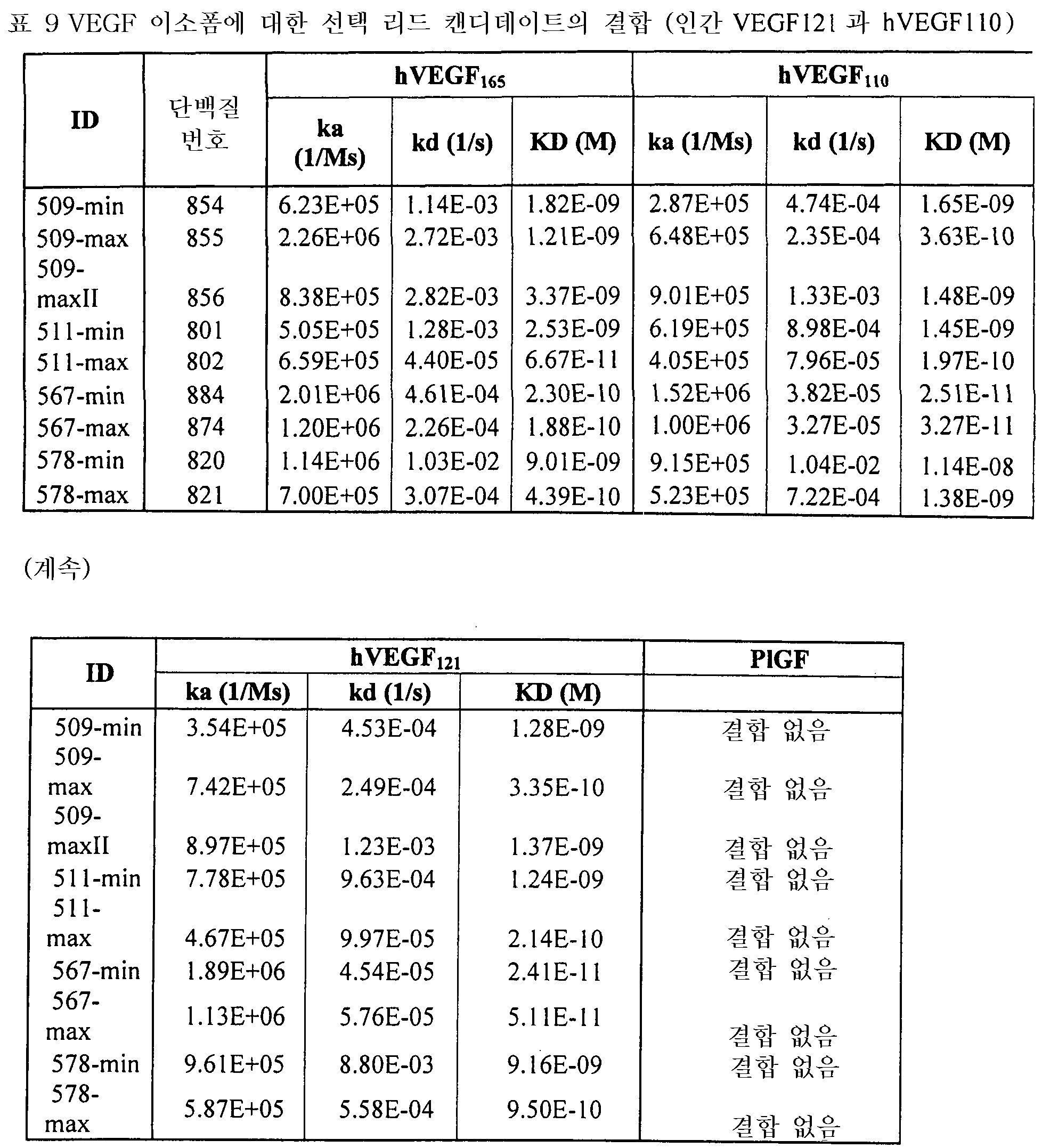
도 3은 578max의 VEGF 아이소폼(isoforms) (hVEGFl21 및 hVEGFl10)에 대한 결합 반응속도를 나타낸 것이다. 도 3a는 사람 VEGF 165에 대해 얻어진 데이터를 나타낸다: Ka(1/Ms): 7,00E+05; SE (ka): 1.4E+03; kd(l/s): 3,07E-04; SE(kd): 8,50E-07; KD(M): 4,39E-1O. 도 3b는 사람 VEGF121에 대해 얻어진 데이터를 나타낸다: Ka(1/Ms): 5,87E+05; SE (ka): 1.20E+03; kd(l/s): 5,58E-04; SE(kd): 9,60E-07; KD(M): 9,50E-11. 도 3c는 사람 VEGFl10에 대해 얻어진 데이터를 나타낸다: Ka (I/Ms): 5,23E+05; SE (ka): 1.3OE+O3; kd(l/s): 7,22E-04; SE(kd): 8,10E-07; KD(M): 1.38E-09.
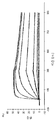
도 4는 578max, 578minmax 및 578wt의 hVEGF165에 대한 결합 반응속도를 나타낸 것이다. 도 4a는 578max에 대해 얻어진 데이터를 나타낸다: Ka(1/Ms): 7,00E+05; SE (ka): 1.40E+03; kd(l/s): 3,07E-04; SE(kd): 8,50E-07; KD(M): 4,39E-IO. 도 4b는 578minmax에 대해 얻어진 데이터를 나타낸다: Ka(1/Ms): 8,06E+05; SE (ka): 2,10E+03; kd(l/s): 5,04E-04; SE(kd): l,10E-06; KD(M): 6,25E-IO. 도 4c는 578wt-His에 대해 얻어진 데이터를 나타낸다: Ka(1/Ms): 8,45E+05; SE (ka): 1.60E+03; kd(l/s): l,69E-04; SE(kd): 7,60E-07; KD(M): 2,00E-IO.
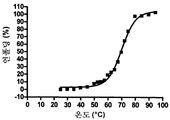
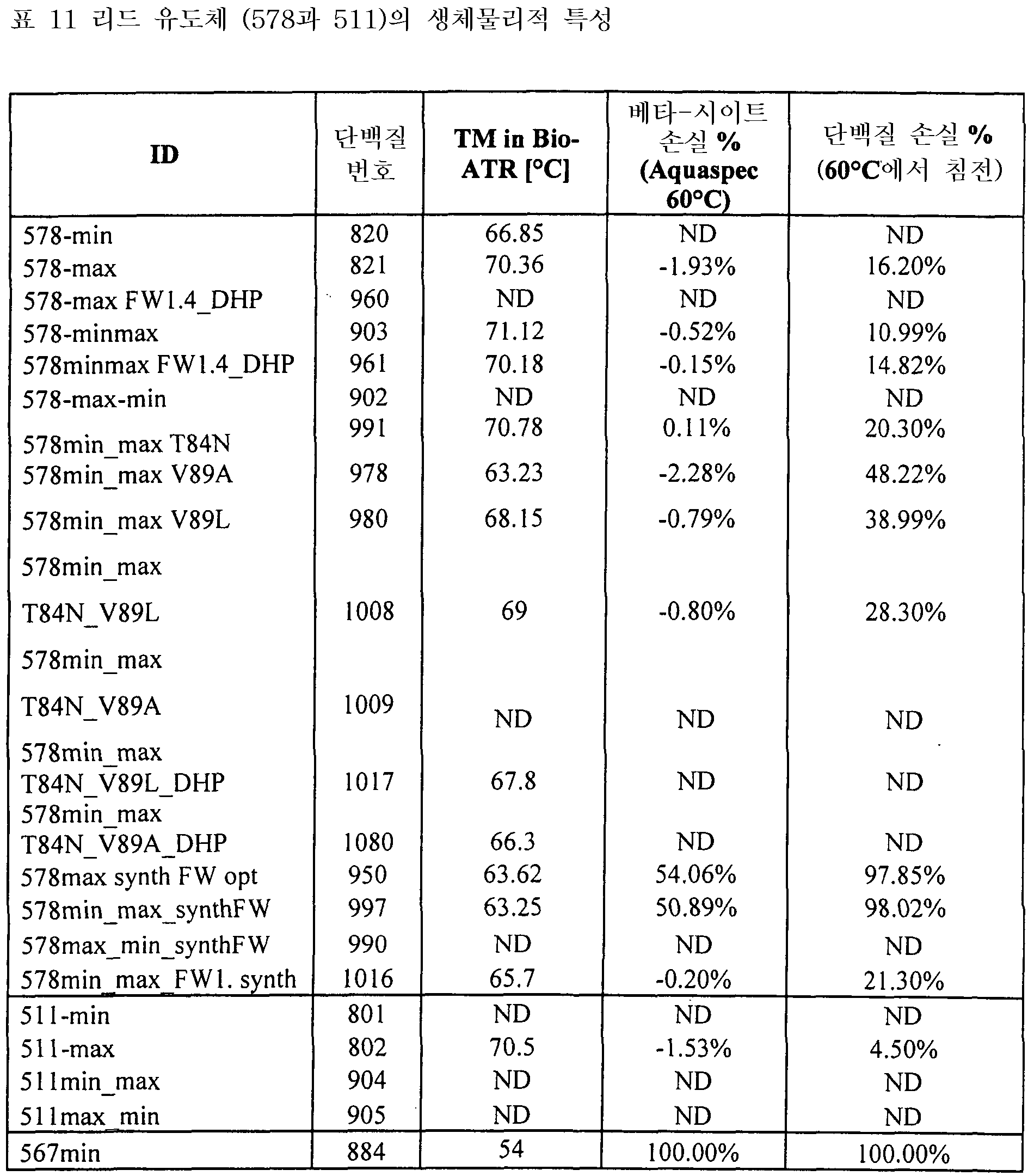
도 5는 578max, 578minmax 및 578minmax_DHP의 열 안정성을 나타낸 것이다(FT-IR에 의해 측정된 언폴딩). 도 5a: 578minmax (ESBA903): Tm = 71.1 ℃; 도 5b: 578minmax_DHP (#961): Tm = 70.2 ℃; 도 5c: 578max (#821): Tm = 70.4 ℃.
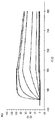
도 6은 30분 동안의 열적 스트레스(도 6a: 50 ℃, 도 6b: 60 ℃, 도 6c: 70 ℃) 후 578 유도체의 변성 및 침전을 나타낸 것이다.
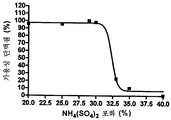
도 7은 578max, 578minmax 및 578minmax_DHP의 용해도(암모늄 설페이트 침전에 의해 측정)를 나타낸 것이다. 도 7a: 578max (#821). V50은 27,24 %였다. 도 7b: 578minmax (ESBA903). V50은 28,13 %였다. 도 7c: 578minmax_DHP (#961). V50은 32,36 %였다.
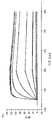
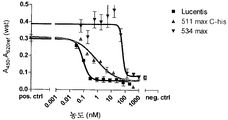
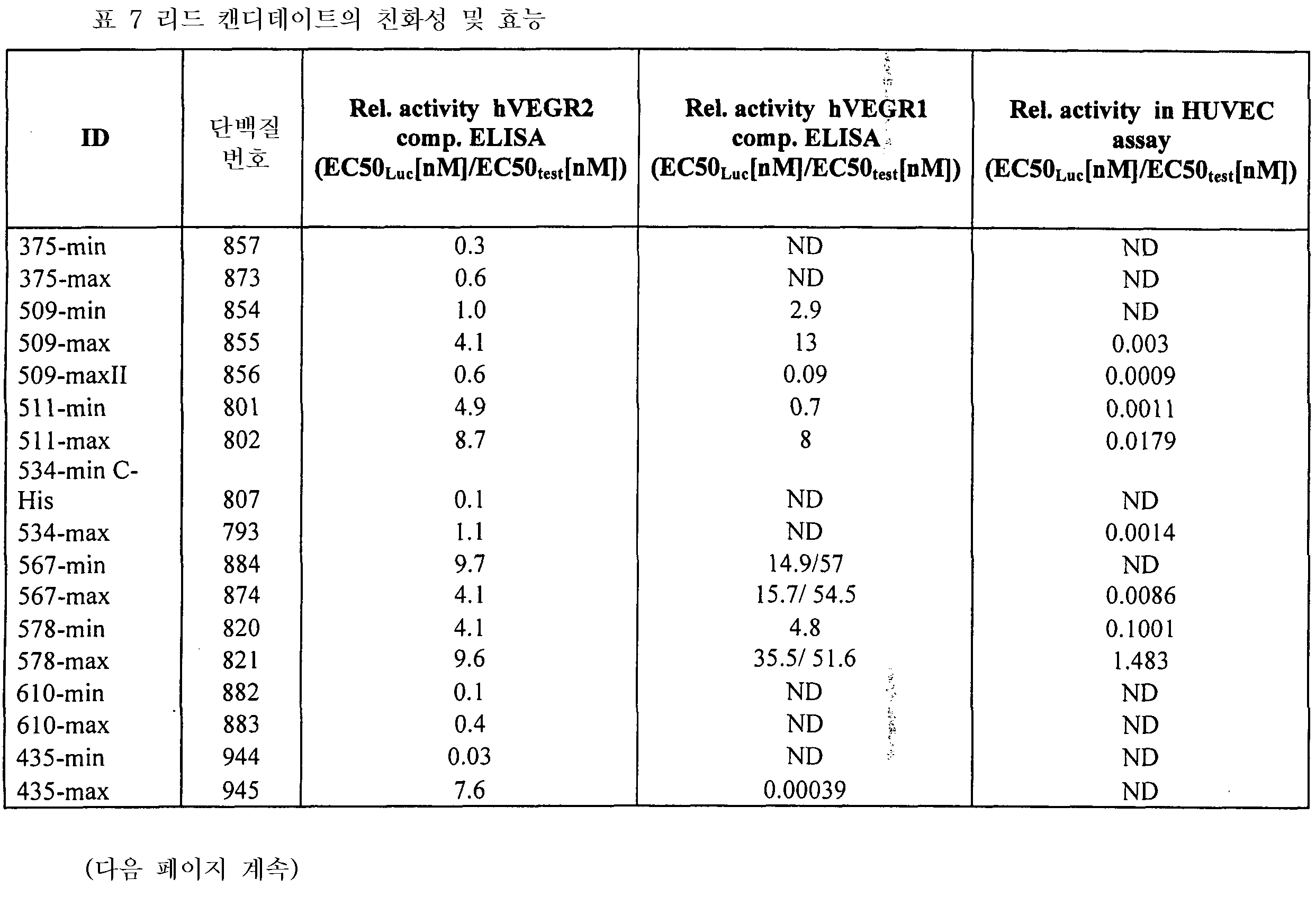
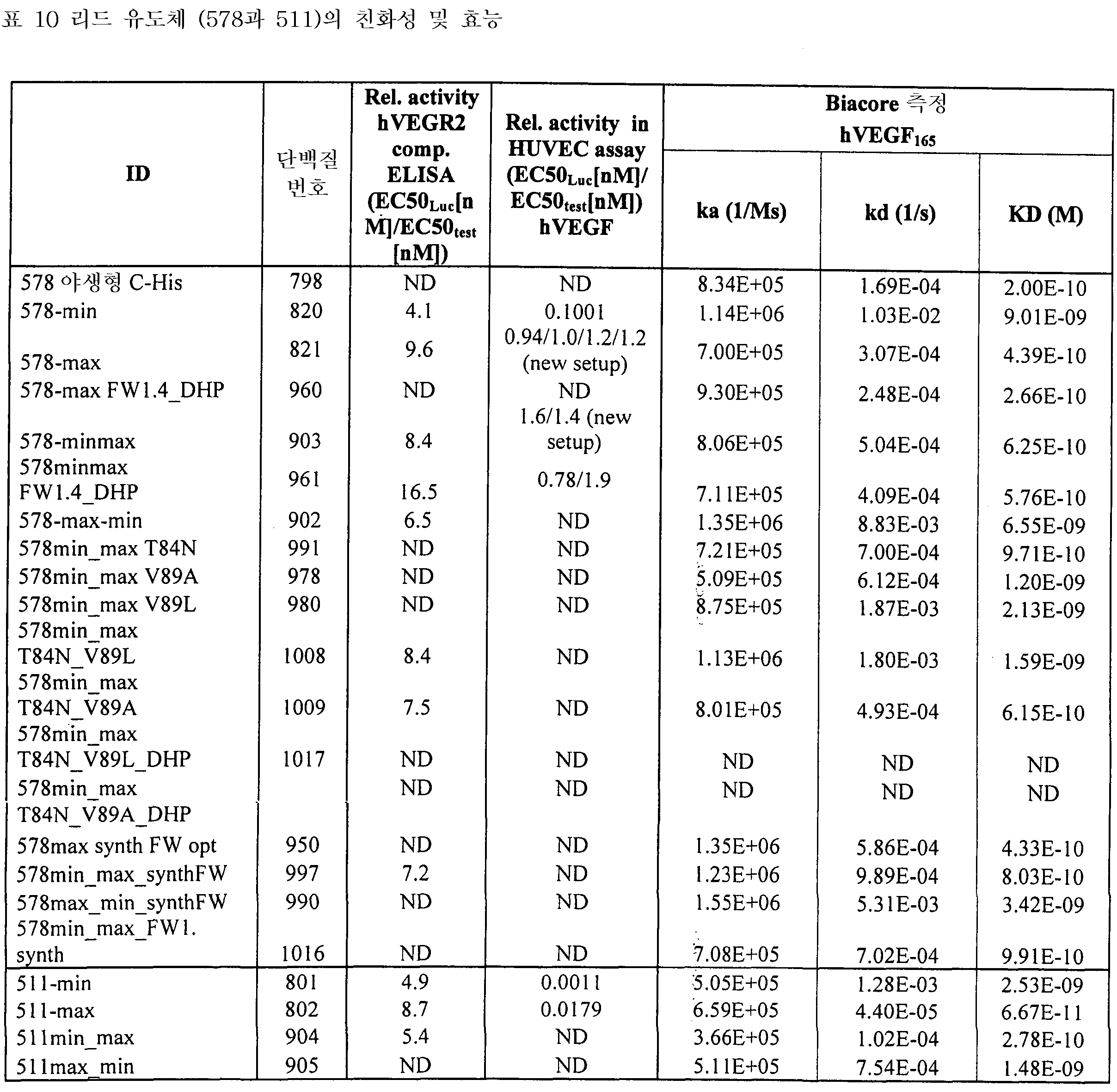
도 8은 VEGFR2 경쟁적 ELISA 대 포텐시(potency)를 측정하는 방법인 HUVEC 에세이를 나타낸 것이다. 도 8a: VEGFR2 경쟁적 ELISA에서 Lucentis와 511max (#802)의 비교. Lucentis의 R2: 0,9417; ESBA802의 R2: 0,9700. Lucentis의 EC50: 7,137 nM; #802의 EC50: 0.8221 nM. 도 8b: VEGFR2 경쟁적 ELISA에서 Lucentis와 578max (#821)의 비교. 도 8c: HUVEC 에세이에서 Lucentis, 511maxC-his 및 534max의 비교. Lucentis의 R2: 0.9399; EP511maxC-his의 R2: 0.9313, EP534max의 R2: 0.7391. Lucetins의 EC50: 0.08825 nM, 51lmaxC-his의 EC50: 0,7646 nM, 534max의EC50: 63.49 nM. 도 8d: HUVEC 에세이에서 Lucentis, 578min 및 578max의 비교. Lucentis의 R2: 0,9419, EP578min의 R2: 0,8886, EP578max의 R2: 0,9274. Lucentis 의 EC50: 0,1529 nM, 578min의 EC50: 1,528 nM, 578max의 EC50: 0,1031 nM.
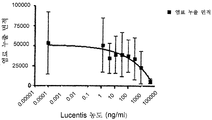
도 9는 hVEGF165에 의해 유도된 HUVEC 증식에 대한 578minmax 의 효과를 나타낸 것이다. 이 에세이의 매개변수들은 다음과 같다: hVEGF165 농도: 0,08nM (3ng/ml); VEGF와 테스트 항목과의 인큐베이션: 96h. Lucentis에 대한 EC50은 0,08959 nM였고 578minmax에 대하여는 0,05516 nM였으며, Lucentis의 R2는 0,9066 이었고 578minmax의 R2는 0,9622였다.
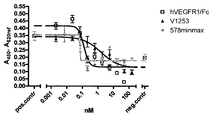
도 10은 마우스 VEGF 164와 래트 VEGF 164에 의해 유도된 HUVEC 증식에 대한 578minmax의 효과를 나타낸 것이다. 이 에세이의 매개변수들은 다음과 같다: 마우스 VEGF 164의 농도: 0,08nM (3ng/ml); 래트 VEGF 164의 농도: 0,3nM (11.3 ng/ml). 2개의 농도는 모두 VEGF로 유도된 HUVEC 증식에 대한 EC90에서 선택되었다. VEGF와 테스트 항목과의 인큐베이션: 96h. 도 10a는 마우스 VEGF에 대해 얻어진 데이터를 나타낸 것이다. EC50은 V1253에 대하여는 0,1196 nM였고 578minmax에 대하여는 0,06309 nM였으며, R2는 Lucetins에 대하여는 0.02744, V1253에 대하여는 0.9348 및 EP578minmax에 대하여는 0.9767이었다. Lucentis는 마우스 VEGF로 유도된 HUVEC 증식을 억제하지 않았다. 도 10b는 래트 VEGF에 대해 얻어진 데이터를 나타낸 것이다. EC50은 V1253에 대하여는 1,597nM였고 578minmax에 대하여는 0,06974였으며, R2는 Vl253에 대하여는 0,7664였고 578minmax에 대하여는 0,6635였다.
도 11은 누드 기니아 피그에서 Miles 에세이를 사용한 효능 시험을 나타낸 것이다(파트 I). 알마 블루 1 염료를 누드 기니아 피그의 정맥 내로 투여하였다. 염료를 주사하고 1시간 후에 hVEGF (2,61nM)와 Lucentis, ESBA903 또는 #802 각각의 예비혼합물 2를 기니아 피그 3의 피부에 주사하였다. 용액을 주사한 1시간 후에 상기 동물 3을 안락사시키고 가죽을 수집하여 세척한 다음, 입사 및 투과광을 사용하여 디지털사진을 촬영하였다. 주사 부위 내로 혈관외 유출된 Evans Blue 염료의 면적을 이미지 J를 사용하여 평가하고 투약 면적 정체를 표시하였다.
도 12는 누드 기니아 피그에서 Miles 에세이를 사용한 효능 시험을 나타낸 것이다(파트 I). 도 12a는 #803 (511max)에 대해 얻어진 결과를 나타낸다. EC50은 5,99O nM였고 2,060과 17,41 nM 사이에서 통계적 분포를 나타내었고 R2는 0.5800이었다. 도 12b는 ESBA903 (578minmax)에 대한 결과를 나타낸다. EC50은 3,989였고 1,456과 10,93 nM 사이에서 통계적 분포를 나타내었고 R2는 0.3920이었다. 도 12c는 Lucentis의 염료 유출 영역을 나타낸다. 명확하지 않은 곡선으로 인하여 Lucentis에 대한 EC50은 계산할 수 없었다.
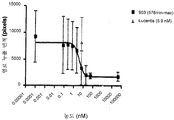
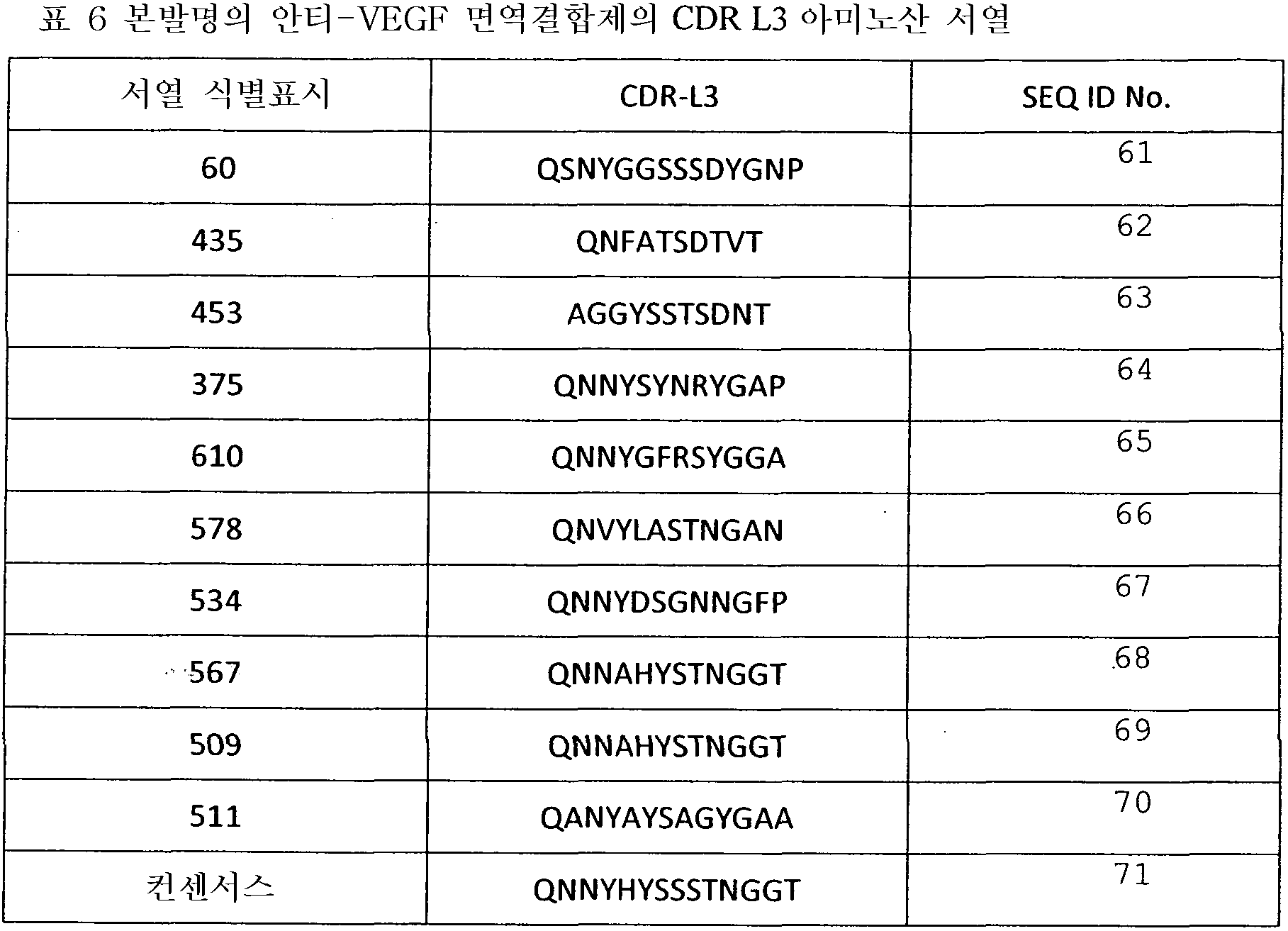
도 13은 래트에서 수정된 Miles 에세이를 사용한 효능 시험을 나타낸 것이다(hVEGF 165와 578minmax (ESBA903)를 미리 혼합). 도 13a는 래트-투약 반응에서 VEGF로 유도된 망막혈관누출에 대한 Avastin의 항투과성 효과를 나타낸 것이다. Avastin은 hVEGF로 유도된 망막 혈관 투과성을 억제한다. 주사 전에 미리 혼합. 대략 등몰(equimolar), 3배 또는 10배 과량. *p<0,05 (VEGF s. BSA), ** p<0,05 (Avastin 처리 대 VEGF). 도 13b는 래트에서 VEGF로 유도된 망막혈관누출에 대한 ESBA903의 항투과성 효과를 나타낸다. 투약 반응(예비혼합, ivt). ESBA903에 의한 hVEGF로 유도된 망막혈관투과성의 완전한 억제. 주사 전 예비혼합. 대략 등몰, 3배 또는 10배 과량. *p<0,05 (VEGF s. BSA), ** p<0,05 (ESBA903 처리 대 VEGF).
도 14는 래트에서 수정된 Miles 에세이를 사용한 효능 시험을 나타낸 것이다 (578minmax (ESBA903)의 국소 투여). 래트에서 VEGF로 유도된 망막혈관누출에 대한 AL-51287 (ESBA903)의 항투과성 효능을 국소 투여에 대하여 시험하였다. 5일의 예비처리, 10 ng/ml ESBA903 제제 4 drops/일. *p<0,05 (VEGF s. BSA), ** p<0,05 (VEGF 대 AL-51287), ***p=0,060 (AL-51287 대 AL-52667), ****(VEGF 대 AL-39324); p<0,05 (AL-39324 대 비히클 참조 대조군). AL-51287: ESBA903; AL-52657: 국소 비히클 참조 대조군; AL-39324: 소형 분자 RTK 억제제.
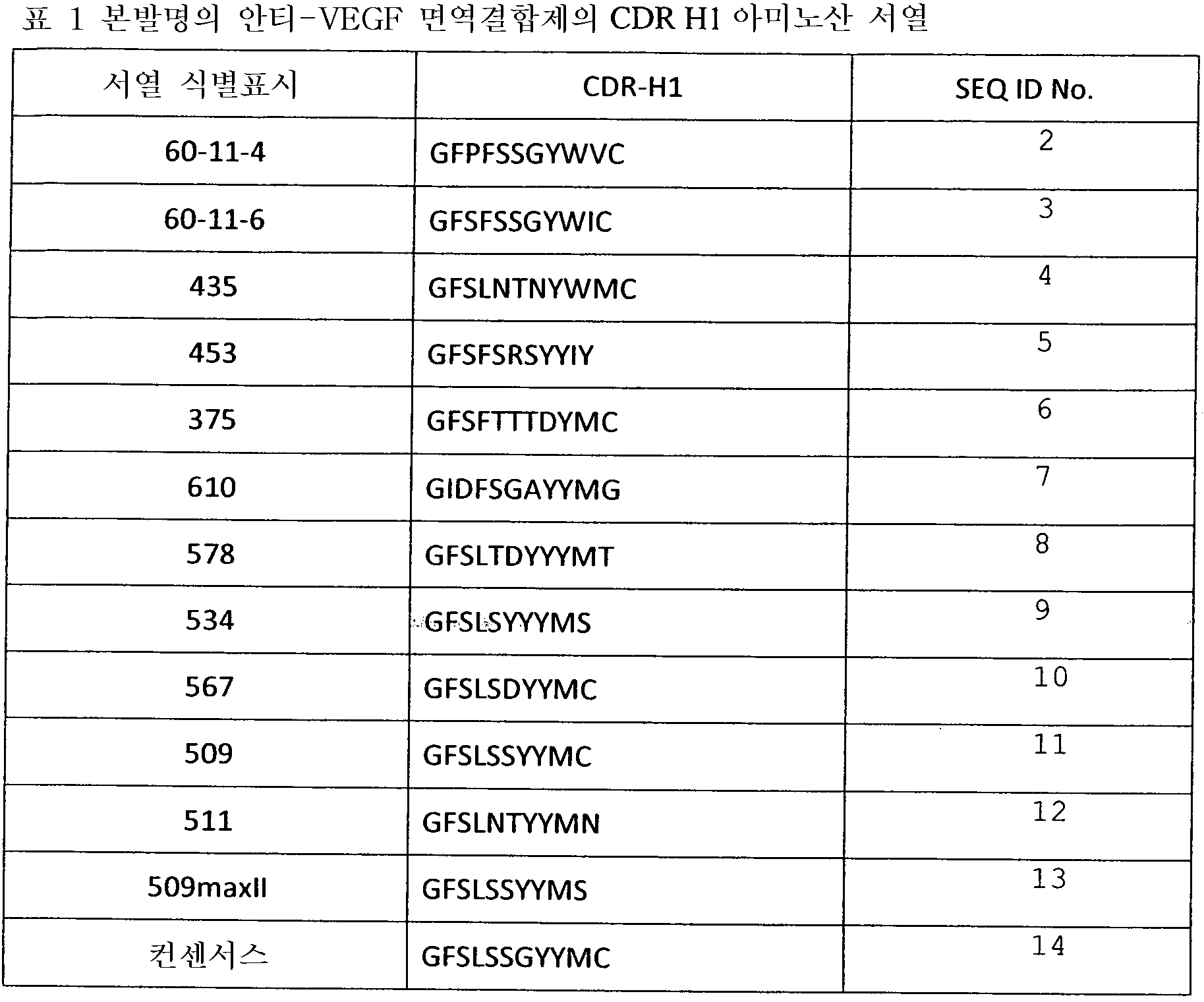
도 15는 여기에서 사용된 VH의 CDRl의 정의를 나타낸 것이다.
Claims (12)
- 가변 중쇄 (VH) 및 가변 경쇄 (VL)를 포함하는 재조합 항체로서, 여기서
a. 가변 중쇄가 SEQ ID NO: 2, SEQ ID NO: 15 및 SEQ ID NO: 27로 이루어지는 군의 상보성 결정 영역 (CDR)을 포함하고, 가변 경쇄가 SEQ ID NO: 38, SEQ ID NO: 50 및 SEQ ID NO: 61로 이루어지는 군의 CDR을 포함하거나;
b. 가변 중쇄가 SEQ ID NO: 3, SEQ ID NO: 15 및 SEQ ID NO: 27로 이루어지는 군의 CDR을 포함하고, 가변 경쇄가 SEQ ID NO: 38, SEQ ID NO: 50 및 SEQ ID NO: 61로 이루어지는 군의 CDR을 포함하거나;
c. 가변 중쇄가 SEQ ID NO: 4, SEQ ID NO: 16 및 SEQ ID NO: 28로 이루어지는 군의 CDR을 포함하고, 가변 경쇄가 SEQ ID NO: 39, SEQ ID NO: 51 및 SEQ ID NO: 62로 이루어지는 군의 CDR을 포함하거나;
d. 가변 중쇄가 SEQ ID NO: 5, SEQ ID NO: 17 및 SEQ ID NO: 29로 이루어지는 군의 CDR을 포함하고, 가변 경쇄가 SEQ ID NO: 40, SEQ ID NO: 52 및 SEQ ID NO: 63로 이루어지는 군의 CDR을 포함하거나;
e. 가변 중쇄가 SEQ ID NO: 6, SEQ ID NO: 18 및 SEQ ID NO: 30 로 이루어지는 군의 CDR을 포함하고, 가변 경쇄가 SEQ ID NO: 41, SEQ ID NO: 53 및 SEQ ID NO: 64로 이루어지는 군의 CDR을 포함하거나;
f. 가변 중쇄가 SEQ ID NO: 7, SEQ ID NO: 19 및 SEQ ID NO: 31로 이루어지는 군의 CDR을 포함하고, 가변 경쇄가 SEQ ID NO: 42, SEQ ID NO: 54 및 SEQ ID NO: 65로 이루어지는 군의 CDR을 포함하거나;
g. 가변 중쇄가 SEQ ID NO: 9, SEQ ID NO: 21 및 SEQ ID NO: 33로 이루어지는 군의 CDR을 포함하고, 가변 경쇄가 SEQ ID NO: 44, SEQ ID NO: 56 및 SEQ ID NO: 67로 이루어지는 군의 CDR을 포함하거나;
h. 가변 중쇄가 SEQ ID NO: 10, SEQ ID NO: 22 및 SEQ ID NO: 34로 이루어지는 군의 CDR을 포함하고, 가변 경쇄가 SEQ ID NO: 45, SEQ ID NO: 57 및 SEQ ID NO: 68로 이루어지는 군의 CDR을 포함하거나;
i. 가변 중쇄가 (i) SEQ ID NO: 11, SEQ ID NO: 23 및 SEQ ID NO: 35로 이루어지는 군; (ii) SEQ ID NO: 11, SEQ ID NO: 25 및 SEQ ID NO: 35로 이루어지는 군; (iii) SEQ ID NO: 13, SEQ ID NO: 23 및 SEQ ID NO: 35로 이루어지는 군; 또는 (iv) SEQ ID NO: 13, SEQ ID NO: 25 및 SEQ ID NO: 35로 이루어지는 군의 CDR을 포함하고, 가변 경쇄가 SEQ ID NO: 46, SEQ ID NO: 58 및 SEQ ID NO: 69로 이루어지는 군의 CDR을 포함하거나; 또는
j. 가변 중쇄가 SEQ ID NO: 12, SEQ ID NO: 24 및 SEQ ID NO: 36로 이루어지는 군의 CDR을 포함하고, 가변 경쇄가 (i) SEQ ID NO: 47, SEQ ID NO: 59 및 SEQ ID NO: 70로 이루어지는 군의 CDR; 또는 (ii) SEQ ID NO: 48, SEQ ID NO: 59 및 SEQ ID NO: 70로 이루어지는 군의 CDR을 포함하는, 재조합 항체. - 제 1 항의 재조합 항체의 항원-결합 단편으로서, 상기 단편이 scFv, Fab, F(ab')2 또는 Fab'인, 항원-결합 단편.
- 제 2 항에 있어서, 상기 단편이 scFv이고 여기서 중쇄 가변 영역 및 경쇄 가변 영역이 SEQ ID NO: 181의 서열에 의해 연결된 것인, 항원-결합 단편.
- 제 1 항의 재조합 항체를 포함하는 약학 조성물.
- 제 2 항의 항원-결합 단편을 포함하는 약학 조성물.
- 제 3 항의 항원-결합 단편을 포함하는 약학 조성물.
- 대상에서 VEGF-매개 질환이 치료되도록 제 1 항의 항체의 치료적 유효량을 대상에 투여하는 것을 포함하는, 대상에서 인간 VEGF-매개 질환을 치료하는 방법.
- 제 7 항에 있어서, VEGF-매개 질환이 연령과 관련된 황반 퇴화, 신생혈관 녹내장, 당뇨망막병증, 미숙아 망막증, 후수정체 섬유증식증, 유방암, 폐암, 위암, 식도암, 대장암, 간암, 난소암, 혼수, 남성화세포종, 자궁경부암, 자궁내막암, 자궁내막증식, 자궁내막증, 섬유육종, 융모암, 머리와 목의 암, 상인두암, 후두암, 간아세포종, 카포시 육종, 악성 흑색종, 피부암, 혈관종, 해면상 혈관종, 혈관모 세포종, 췌장암, 망막아세포종, 성상세포종, 교모세포종, 슈반종, 핍지교종, 수모세포종, 신경아세포종, 횡문근육종, 골육종, 평활근육종, 요로암, 갑상선암, 빌름스 종양, 신세포암, 전립선암, 모반증과 연관된 비정상 혈관 증식, 부종 (뇌종양과 연관된 부종 등), 메이그 신드롬, 류마티스 관절염, 건선 및 동맥경화증으로 이루어지는 군으로부터 선택되는 것인, 방법.
- 대상에서 VEGF-매개 질환이 치료되도록 제 2 항의 항원-결합 단편의 치료적 유효량을 대상에 투여하는 것을 포함하는, 대상에서 인간 VEGF-매개 질환을 치료하는 방법.
- 제 9 항에 있어서, VEGF-매개 질환이 연령과 관련된 황반 퇴화, 신생혈관 녹내장, 당뇨망막병증, 미숙아 망막증, 후수정체 섬유증식증, 유방암, 폐암, 위암, 식도암, 대장암, 간암, 난소암, 혼수, 남성화세포종, 자궁경부암, 자궁내막암, 자궁내막증식, 자궁내막증, 섬유육종, 융모암, 머리와 목의 암, 상인두암, 후두암, 간아세포종, 카포시 육종, 악성 흑색종, 피부암, 혈관종, 해면상 혈관종, 혈관모 세포종, 췌장암, 망막아세포종, 성상세포종, 교모세포종, 슈반종, 핍지교종, 수모세포종, 신경아세포종, 횡문근육종, 골육종, 평활근육종, 요로암, 갑상선암, 빌름스 종양, 신세포암, 전립선암, 모반증과 연관된 비정상 혈관 증식, 부종 (뇌종양과 연관된 부종 등), 메이그 신드롬, 류마티스 관절염, 건선 및 동맥경화증으로 이루어지는 군으로부터 선택되는 것인, 방법.
- 제 1 항의 항체 또는 제 2 항의 항원-결합 단편을 포함하는 이중특이성 분자.
- 제 11 항에 있어서, 다중특이성인, 이중특이성 분자.
Applications Claiming Priority (9)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US7569208P | 2008-06-25 | 2008-06-25 | |
| US7569708P | 2008-06-25 | 2008-06-25 | |
| US13321208P | 2008-06-25 | 2008-06-25 | |
| US61/133,212 | 2008-06-25 | ||
| US61/075,697 | 2008-06-25 | ||
| US61/075,692 | 2008-06-25 | ||
| US15504109P | 2009-02-24 | 2009-02-24 | |
| US61/155,041 | 2009-02-24 | ||
| PCT/CH2009/000220 WO2009155724A2 (en) | 2008-06-25 | 2009-06-25 | Stable and soluble antibodies inhibiting vegf |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020187000324A Division KR20180005753A (ko) | 2008-06-25 | 2009-06-25 | Vegf를 억제하는 안정하고 가용성인 항체 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020197022648A Division KR102157097B1 (ko) | 2008-06-25 | 2009-06-25 | Vegf를 억제하는 안정하고 가용성인 항체 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20180098705A true KR20180098705A (ko) | 2018-09-04 |
| KR102095257B1 KR102095257B1 (ko) | 2020-04-01 |
Family
ID=41209803
Family Applications (8)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020187024733A Active KR102095257B1 (ko) | 2008-06-25 | 2009-06-25 | Vegf를 억제하는 안정하고 가용성인 항체 |
| KR1020187000324A Abandoned KR20180005753A (ko) | 2008-06-25 | 2009-06-25 | Vegf를 억제하는 안정하고 가용성인 항체 |
| KR1020177012884A Active KR101817284B1 (ko) | 2008-06-25 | 2009-06-25 | Vegf를 억제하는 안정하고 가용성인 항체 |
| KR1020157032314A Active KR101671886B1 (ko) | 2008-06-25 | 2009-06-25 | Vegf를 억제하는 안정하고 가용성인 항체 |
| KR1020197022648A Active KR102157097B1 (ko) | 2008-06-25 | 2009-06-25 | Vegf를 억제하는 안정하고 가용성인 항체 |
| KR1020117001961A Active KR102013220B1 (ko) | 2008-06-25 | 2009-06-25 | Vegf를 억제하는 안정하고 가용성인 항체 |
| KR1020207026266A Active KR102296443B1 (ko) | 2008-06-25 | 2009-06-25 | Vegf를 억제하는 안정하고 가용성인 항체 |
| KR1020167030125A Active KR101737466B1 (ko) | 2008-06-25 | 2009-06-25 | Vegf를 억제하는 안정하고 가용성인 항체 |
Family Applications After (7)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020187000324A Abandoned KR20180005753A (ko) | 2008-06-25 | 2009-06-25 | Vegf를 억제하는 안정하고 가용성인 항체 |
| KR1020177012884A Active KR101817284B1 (ko) | 2008-06-25 | 2009-06-25 | Vegf를 억제하는 안정하고 가용성인 항체 |
| KR1020157032314A Active KR101671886B1 (ko) | 2008-06-25 | 2009-06-25 | Vegf를 억제하는 안정하고 가용성인 항체 |
| KR1020197022648A Active KR102157097B1 (ko) | 2008-06-25 | 2009-06-25 | Vegf를 억제하는 안정하고 가용성인 항체 |
| KR1020117001961A Active KR102013220B1 (ko) | 2008-06-25 | 2009-06-25 | Vegf를 억제하는 안정하고 가용성인 항체 |
| KR1020207026266A Active KR102296443B1 (ko) | 2008-06-25 | 2009-06-25 | Vegf를 억제하는 안정하고 가용성인 항체 |
| KR1020167030125A Active KR101737466B1 (ko) | 2008-06-25 | 2009-06-25 | Vegf를 억제하는 안정하고 가용성인 항체 |
Country Status (26)
| Country | Link |
|---|---|
| US (6) | US8349322B2 (ko) |
| EP (3) | EP2307454B1 (ko) |
| JP (7) | JP5956752B2 (ko) |
| KR (8) | KR102095257B1 (ko) |
| CN (3) | CN104961828B (ko) |
| AU (1) | AU2009264565C1 (ko) |
| BR (1) | BRPI0914251B1 (ko) |
| CA (2) | CA2727839C (ko) |
| CL (1) | CL2010001544A1 (ko) |
| CY (3) | CY1118829T1 (ko) |
| DK (3) | DK3216803T3 (ko) |
| ES (3) | ES2998307T3 (ko) |
| FI (1) | FI3722310T3 (ko) |
| HR (3) | HRP20170615T1 (ko) |
| HU (4) | HUE032894T2 (ko) |
| LT (4) | LT2307454T (ko) |
| MX (3) | MX347972B (ko) |
| NL (1) | NL301058I2 (ko) |
| NO (1) | NO2020021I1 (ko) |
| PH (1) | PH12015501593A1 (ko) |
| PL (3) | PL2307454T3 (ko) |
| PT (3) | PT3216803T (ko) |
| RU (6) | RU2531523C3 (ko) |
| SI (3) | SI3216803T1 (ko) |
| WO (1) | WO2009155724A2 (ko) |
| ZA (1) | ZA201008594B (ko) |
Families Citing this family (104)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3252079B8 (en) | 2006-04-07 | 2020-09-09 | Aerpio Therapeutics LLC | Antibodies that bind human protein tyrosine phosphatase beta (hptp-beta) and uses thereof |
| HUE032894T2 (hu) | 2008-06-25 | 2017-11-28 | Esbatech Alcon Biomed Res Unit | VEGF-gátló, stabilis és oldható antitestek |
| RU2653753C1 (ru) | 2008-06-25 | 2018-05-14 | ИЭсБиЭйТЕК, ЭН АЛЬКОН БАЙОМЕДИКАЛ РИСЕРЧ ЮНИТ ЭлЭлСи | СТАБИЛЬНЫЕ И РАСТВОРИМЫЕ АНТИТЕЛА, ИНГИБИРУЮЩИЕ TNFα |
| CN104004094B (zh) | 2008-06-25 | 2017-09-22 | 艾斯巴技术-诺华有限责任公司 | 使用通用抗体构架进行的兔抗体的人源化 |
| MX344076B (es) | 2008-06-30 | 2016-12-05 | Esbatech Alcon Biomed Res Unit | Polipeptidos funcionalizados. |
| US8268314B2 (en) | 2008-10-08 | 2012-09-18 | Hoffmann-La Roche Inc. | Bispecific anti-VEGF/anti-ANG-2 antibodies |
| CA2744103C (en) | 2009-02-24 | 2018-01-02 | Esbatech, An Alcon Biomedical Research Unit Llc | Methods for identifying immunobinders of cell-surface antigens |
| CN102482321B (zh) | 2009-06-22 | 2015-06-17 | 安姆根有限公司 | 使用化学控制的氧化还原态重折叠蛋白质 |
| WO2010151688A2 (en) | 2009-06-25 | 2010-12-29 | Amgen Inc. | Capture purification processes for proteins expressed in a non-mammalian system |
| WO2012054077A2 (en) * | 2010-10-19 | 2012-04-26 | Mayo Foundation For Medical Education And Research | Human antibodies and diagnostic and therapeutic uses thereof for the treatment of neurological disease |
| AR083495A1 (es) | 2010-10-22 | 2013-02-27 | Esbatech Alcon Biomed Res Unit | Anticuerpos estables y solubles |
| US9527925B2 (en) | 2011-04-01 | 2016-12-27 | Boehringer Ingelheim International Gmbh | Bispecific binding molecules binding to VEGF and ANG2 |
| US10752676B2 (en) * | 2011-09-16 | 2020-08-25 | Ucb Biopharma Sprl | Neutralising antibodies to the major exotoxins TCDA and TCDB of Clostridium difficile |
| CN104039351A (zh) | 2011-10-13 | 2014-09-10 | 阿尔皮奥治疗学股份有限公司 | 用于治疗血管渗漏综合征和癌症的方法 |
| JP6184982B2 (ja) | 2012-02-02 | 2017-08-23 | エスバテック − ア ノバルティス カンパニー エルエルシー | 点眼用抗体含有徐放性製剤 |
| EP2872534B1 (en) | 2012-07-13 | 2018-08-08 | Roche Glycart AG | Bispecific anti-vegf/anti-ang-2 antibodies and their use in the treatment of ocular vascular diseases |
| MX2015007931A (es) | 2012-12-18 | 2015-10-05 | Novartis Ag | Composiciones y metodos que utilizan una etiqueta de peptido que se une a hialuronano. |
| SMT202000088T1 (it) | 2013-02-18 | 2020-03-13 | Vegenics Pty Ltd | Molecole leganti ligandi e relativi usi |
| KR101541478B1 (ko) * | 2013-05-31 | 2015-08-05 | 동아쏘시오홀딩스 주식회사 | 항-vegf 항체 및 이를 포함하는 암 또는 신생혈관형성관련 질환의 예방, 진단 또는 치료용 약학 조성물 |
| IL243311B (en) * | 2013-06-26 | 2022-07-01 | Numab Therapeutics AG | New frameworks for antibodies |
| EA201690212A8 (ru) | 2013-07-12 | 2016-08-31 | Офтотек Корпорейшн | Способы лечения или профилактики офтальмологических патологических состояний |
| WO2015076425A1 (ja) * | 2013-11-25 | 2015-05-28 | リンク・ジェノミクス株式会社 | 新規モノクローナル抗体 |
| EP3828202A1 (en) | 2014-05-12 | 2021-06-02 | Formycon AG | Pre-filled plastic syringe containing a vegf antagonist |
| EP3160991A2 (en) | 2014-06-25 | 2017-05-03 | Novartis AG | Compositions and methods for long acting proteins |
| EP3160990A2 (en) | 2014-06-25 | 2017-05-03 | Novartis AG | Compositions and methods for long acting proteins |
| US9840553B2 (en) | 2014-06-28 | 2017-12-12 | Kodiak Sciences Inc. | Dual PDGF/VEGF antagonists |
| GB201412658D0 (en) | 2014-07-16 | 2014-08-27 | Ucb Biopharma Sprl | Molecules |
| KR102390359B1 (ko) | 2014-09-29 | 2022-04-22 | 삼성전자주식회사 | 폴리펩타이드, 이를 포함하는 항 VEGF 항체 및 항 c-Met/항 VEGF 이중 특이 항체 |
| TWI761959B (zh) * | 2014-11-07 | 2022-04-21 | 瑞士商諾華公司 | 治療眼部疾病之方法 |
| CA2960297A1 (en) | 2014-11-10 | 2016-05-19 | Genentech, Inc. | Anti-interleukin-33 antibodies and uses thereof |
| EP3250596A1 (en) * | 2015-01-28 | 2017-12-06 | Pfizer Inc | Stable aqueous anti- vascular endothelial growth factor (vegf) antibody formulation |
| JP6752223B2 (ja) | 2015-04-16 | 2020-09-09 | アルダー・バイオファーマシューティカルズ・インコーポレーテッド | 羞明を処置、予防、または阻害するための抗pacap抗体及びそれらの抗原結合性断片の使用 |
| US10851399B2 (en) * | 2015-06-25 | 2020-12-01 | Native Microbials, Inc. | Methods, apparatuses, and systems for microorganism strain analysis of complex heterogeneous communities, predicting and identifying functional relationships and interactions thereof, and selecting and synthesizing microbial ensembles based thereon |
| GB201601077D0 (en) | 2016-01-20 | 2016-03-02 | Ucb Biopharma Sprl | Antibody molecule |
| GB201601073D0 (en) | 2016-01-20 | 2016-03-02 | Ucb Biopharma Sprl | Antibodies |
| CN107922975B (zh) | 2015-08-12 | 2022-06-28 | 诺华股份有限公司 | 治疗眼科病症的方法 |
| NZ741780A (en) | 2015-10-30 | 2019-11-29 | Genentech Inc | Anti-htra1 antibodies and methods of use thereof |
| US11136609B2 (en) * | 2015-11-09 | 2021-10-05 | National University Corporation Kyoto Institute Of Technology | Separating agent for human serum-derived IgG polyclonal antibodies, and method for separating human serum-derived IgG polyclonal antibodies using same |
| WO2018218013A2 (en) | 2017-05-24 | 2018-11-29 | Sio2 Medical Products, Inc. | Sterilizable pharmaceutical package for ophthalmic formulations |
| CN108883172A (zh) | 2015-11-18 | 2018-11-23 | 福迈康股份公司 | 包含vegf拮抗剂的液体配制品的预填充药物包装 |
| RU2751510C2 (ru) | 2015-11-18 | 2021-07-14 | Сио2 Медикал Продактс, Инк. | Фармацевтическая упаковка для офтальмологических составов |
| US20180326126A1 (en) | 2015-11-18 | 2018-11-15 | Formycon Ag | Pre-filled plastic syringe containing a vegf antagonist |
| GB201521382D0 (en) | 2015-12-03 | 2016-01-20 | Ucb Biopharma Sprl | Antibodies |
| GB201521389D0 (en) | 2015-12-03 | 2016-01-20 | Ucb Biopharma Sprl | Method |
| BR112018013407A2 (en) | 2015-12-30 | 2018-12-18 | Kodiak Sciences Inc. | antibodies and conjugates thereof |
| WO2017119435A1 (ja) | 2016-01-06 | 2017-07-13 | 株式会社オーダーメードメディカルリサーチ | 高親和性抗vegf抗体 |
| KR102293753B1 (ko) | 2016-01-06 | 2021-08-24 | 오더-메이드 메디컬 리서치 인코포레이티드 | Vegf 와 nrp1 의 결합을 저해하는 항체 |
| USD789171S1 (en) | 2016-01-21 | 2017-06-13 | Nomis Llc | Right angle drive |
| AU2017213103B2 (en) | 2016-01-26 | 2022-08-11 | Formycon Ag | Liquid formulation of a VEGF antagonist |
| CN109562163B (zh) | 2016-02-05 | 2022-04-08 | 得克萨斯大学体系董事会 | Egfl6特异性单克隆抗体及其使用方法 |
| WO2017156423A2 (en) * | 2016-03-11 | 2017-09-14 | Integrated Biotherapeutics, Inc. | Broadly protective antibody cocktails for treatment of filovirus hemorrhagic fever |
| EP3222673A1 (de) * | 2016-03-23 | 2017-09-27 | LANXESS Deutschland GmbH | Metallazopigmente |
| CA3020839A1 (en) | 2016-04-15 | 2017-10-19 | Alder Biopharmaceuticals, Inc. | Anti-pacap antibodies and uses thereof |
| CR20180599A (es) | 2016-06-30 | 2019-04-09 | Celltrion Inc | Preparación farmacéutica líquida estable |
| EP3481963B1 (en) | 2016-07-08 | 2025-10-29 | Genentech, Inc. | Methods for diagnosing and treating cancer by means of the expression status and mutational status of nrf2 and downstream target genes of said gene. |
| WO2018162724A1 (en) * | 2017-03-09 | 2018-09-13 | Mab Discovery Gmbh | Antibodies specifically binding to human il-1r7 |
| JOP20190245A1 (ar) | 2017-04-20 | 2019-10-15 | Novartis Ag | أنظمة توصيل إطلاق مستدام تتضمن روابط بلا أثر لنقطة الربط |
| CA3063320A1 (en) | 2017-05-19 | 2018-11-22 | Council Of Scientific & Industrial Research | A method for producing refolded recombinant humanized ranibizumab |
| WO2018215580A1 (en) | 2017-05-24 | 2018-11-29 | Formycon Ag | Method for sterilizing prefilled plastic syringes containing a vegf antagonist |
| WO2018217995A1 (en) | 2017-05-24 | 2018-11-29 | Formycon Ag | Sterilizable pre-filled pharmaceutical packages comprising a liquid formulation of a vegf-antagonist |
| EP3645048A4 (en) * | 2017-06-25 | 2021-06-16 | Systimmune, Inc. | MULTISPECIFIC ANTIBODIES AND THE METHOD OF MANUFACTURING AND USING THEREOF |
| WO2019020777A1 (en) | 2017-07-26 | 2019-01-31 | Formycon Ag | LIQUID FORMULATION OF A VEGF ANTAGONIST |
| FI3717636T3 (fi) | 2017-11-27 | 2023-06-01 | 4D Molecular Therapeutics Inc | Adenoassosioidun viruksen kapsidivariantteja ja käyttö angiogeneesin estämisessä |
| WO2019129677A1 (en) | 2017-12-29 | 2019-07-04 | F. Hoffmann-La Roche Ag | Anti-vegf antibodies and methods of use |
| WO2019143837A1 (en) * | 2018-01-17 | 2019-07-25 | Apexigen, Inc. | Anti-pd-l1 antibodies and methods of use |
| SG11202008242XA (en) | 2018-03-02 | 2020-09-29 | Kodiak Sciences Inc | Il-6 antibodies and fusion constructs and conjugates thereof |
| AU2019235577B2 (en) | 2018-03-16 | 2021-09-30 | Novartis Ag | Methods for treating ocular diseases |
| CN111902189A (zh) * | 2018-04-06 | 2020-11-06 | 伊莱利利公司 | 用于治疗儿科患者中的癌症的雷莫芦单抗 |
| KR20250160211A (ko) | 2018-08-30 | 2025-11-11 | 이뮤니티바이오, 인크. | 다중-사슬 키메라 폴리펩타이드 및 이의 용도 |
| WO2020047473A1 (en) | 2018-08-30 | 2020-03-05 | HCW Biologics, Inc. | Single-chain and multi-chain chimeric polypeptides and uses thereof |
| CN113365663B (zh) | 2018-08-30 | 2025-11-21 | 免疫生物公司 | 单链嵌合多肽和其用途 |
| AU2019339469A1 (en) * | 2018-09-13 | 2021-03-11 | Immune-Onc Therapeutics, Inc. | Novel LILRB4 antibodies and uses thereof |
| CN120058958A (zh) | 2018-09-24 | 2025-05-30 | 视点制药公司 | 靶向HPTP-β(VE-PTP)和VEGF的多特异性抗体 |
| MX2021007393A (es) | 2018-12-18 | 2021-09-23 | Novartis Ag | Formulacion de solucion de proteinas que contiene una alta concentracion de un anticuerpo anti-vegf. |
| USD907455S1 (en) | 2019-05-21 | 2021-01-12 | Nomis Llc | Right angle drive attachment |
| USD907456S1 (en) | 2019-05-21 | 2021-01-12 | Nomis Llc | Right angle drill attachment |
| KR20250051157A (ko) | 2019-06-05 | 2025-04-16 | 리제너론 파아마슈티컬스, 인크. | 정밀 투여량 전달을 위한 디바이스 및 방법 |
| AU2020294797B2 (en) | 2019-06-21 | 2025-12-18 | Immunitybio, Inc. | Multi-chain chimeric polypeptides and uses thereof |
| AU2020347575A1 (en) | 2019-09-13 | 2022-03-17 | Novartis Ag | Methods for treating ocular diseases |
| US11912784B2 (en) | 2019-10-10 | 2024-02-27 | Kodiak Sciences Inc. | Methods of treating an eye disorder |
| PE20221781A1 (es) * | 2020-02-07 | 2022-11-16 | Agency Science Tech & Res | Moleculas de union a antigeno contra alppl2 y/o alpp y usos de las mismas |
| CA3169243A1 (en) | 2020-02-11 | 2021-08-19 | HCW Biologics, Inc. | Methods of activating regulatory t cells |
| JP2023513573A (ja) | 2020-02-11 | 2023-03-31 | エイチシーダブリュー バイオロジックス インコーポレイテッド | 加齢性および炎症性疾患を治療する方法 |
| AU2021219720B2 (en) | 2020-02-11 | 2025-10-02 | Immunitybio, Inc. | Chromatography resin and uses thereof |
| WO2021174127A1 (en) * | 2020-02-28 | 2021-09-02 | Apexigen, Inc. | Anti-sirpa antibodies and methods of use |
| KR20220152316A (ko) | 2020-03-12 | 2022-11-15 | 이뮨-온크 테라퓨틱스, 인코포레이티드 | 신규한 항-lilrb4 항체 및 유도체 생성물 |
| CA3174680A1 (en) | 2020-03-13 | 2021-09-16 | Genentech, Inc. | Anti-interleukin-33 antibodies and uses thereof |
| US12497462B2 (en) | 2020-04-29 | 2025-12-16 | Immunitybio, Inc. | Anti-CD26 proteins and uses thereof |
| WO2021247003A1 (en) | 2020-06-01 | 2021-12-09 | HCW Biologics, Inc. | Methods of treating aging-related disorders |
| WO2021247604A1 (en) | 2020-06-01 | 2021-12-09 | HCW Biologics, Inc. | Methods of treating aging-related disorders |
| US12024545B2 (en) | 2020-06-01 | 2024-07-02 | HCW Biologics, Inc. | Methods of treating aging-related disorders |
| EP4182025A1 (en) * | 2020-07-16 | 2023-05-24 | Novartis AG | Anti-betacellulin antibodies, fragments thereof, and multi-specific binding molecules |
| EP4228706A1 (en) | 2020-10-15 | 2023-08-23 | Genentech, Inc. | Hyaluronic acid binding derivatives of versican (vg1) for long acting delivery of therapeutics |
| TW202237181A (zh) | 2020-11-25 | 2022-10-01 | 瑞士商諾華公司 | 治療眼部疾病之方法 |
| WO2022201084A1 (en) | 2021-03-26 | 2022-09-29 | Novartis Ag | Methods for treating ocular diseases |
| BR112023022439A2 (pt) | 2021-04-26 | 2023-12-26 | Celanese Eva Performance Polymers Llc | Dispositivo implantável para liberação sustentada de um composto de fármaco macromolecular |
| EP4145456A1 (en) | 2021-09-06 | 2023-03-08 | Bayer AG | Prediction of a change related to a macular fluid |
| KR102809807B1 (ko) | 2021-12-21 | 2025-05-16 | 한림대학교 산학협력단 | 항-cldn18.2를 포함하는 키메릭 항원 수용체를 유효성분으로 포함하는 암의 예방 또는 치료용 약학적 조성물 및 이의 제조 방법 |
| EP4486368A1 (en) | 2022-03-02 | 2025-01-08 | ImmunityBio, Inc. | Method of treating pancreatic cancer |
| TW202337496A (zh) | 2022-03-17 | 2023-10-01 | 瑞士商諾華公司 | 治療新生血管性年齡相關性黃斑點退化之方法 |
| CN117820474A (zh) * | 2023-04-25 | 2024-04-05 | 南京贝思奥生物科技有限公司 | 改善眼部血管新生相关疾病的药物及其应用 |
| CN117069848B (zh) * | 2023-08-02 | 2025-02-28 | 武汉爱博泰克生物科技有限公司 | 抗人cd146兔单克隆抗体及其应用 |
| US20260049118A1 (en) | 2024-08-19 | 2026-02-19 | HCW Biologics, Inc. | Single-chain chimeric polypeptides and uses thereof |
| WO2026043906A1 (en) | 2024-08-19 | 2026-02-26 | HCW Biologics, Inc. | Multi-chain chimeric polypeptides and uses thereof |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2000034337A1 (en) * | 1998-12-10 | 2000-06-15 | Tsukuba Research Laboratory, Toagosei Co., Ltd. | Humanized monoclonal antibodies against vascular endothelial cell growth factor |
| WO2000053760A2 (en) * | 1999-03-12 | 2000-09-14 | Genentech, Inc. | Method of preventing the death of retinal neurons and treating ocular diseases |
Family Cites Families (71)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4275149A (en) | 1978-11-24 | 1981-06-23 | Syva Company | Macromolecular environment control in specific receptor assays |
| US4376110A (en) | 1980-08-04 | 1983-03-08 | Hybritech, Incorporated | Immunometric assays using monoclonal antibodies |
| US4737456A (en) | 1985-05-09 | 1988-04-12 | Syntex (U.S.A.) Inc. | Reducing interference in ligand-receptor binding assays |
| US6548640B1 (en) | 1986-03-27 | 2003-04-15 | Btg International Limited | Altered antibodies |
| US5225539A (en) | 1986-03-27 | 1993-07-06 | Medical Research Council | Recombinant altered antibodies and methods of making altered antibodies |
| US4946778A (en) | 1987-09-21 | 1990-08-07 | Genex Corporation | Single polypeptide chain binding molecules |
| US5260203A (en) | 1986-09-02 | 1993-11-09 | Enzon, Inc. | Single polypeptide chain binding molecules |
| US4881175A (en) | 1986-09-02 | 1989-11-14 | Genex Corporation | Computer based system and method for determining and displaying possible chemical structures for converting double- or multiple-chain polypeptides to single-chain polypeptides |
| US5013653A (en) | 1987-03-20 | 1991-05-07 | Creative Biomolecules, Inc. | Product and process for introduction of a hinge region into a fusion protein to facilitate cleavage |
| AU612370B2 (en) | 1987-05-21 | 1991-07-11 | Micromet Ag | Targeted multifunctional proteins |
| US5132405A (en) | 1987-05-21 | 1992-07-21 | Creative Biomolecules, Inc. | Biosynthetic antibody binding sites |
| US5258498A (en) | 1987-05-21 | 1993-11-02 | Creative Biomolecules, Inc. | Polypeptide linkers for production of biosynthetic proteins |
| US5091513A (en) | 1987-05-21 | 1992-02-25 | Creative Biomolecules, Inc. | Biosynthetic antibody binding sites |
| WO1989006692A1 (en) | 1988-01-12 | 1989-07-27 | Genentech, Inc. | Method of treating tumor cells by inhibiting growth factor receptor function |
| US5530101A (en) | 1988-12-28 | 1996-06-25 | Protein Design Labs, Inc. | Humanized immunoglobulins |
| US5147638A (en) | 1988-12-30 | 1992-09-15 | Oklahoma Medical Research Foundation | Inhibition of tumor growth by blockade of the protein C system |
| US5859205A (en) | 1989-12-21 | 1999-01-12 | Celltech Limited | Humanised antibodies |
| GB8928874D0 (en) | 1989-12-21 | 1990-02-28 | Celltech Ltd | Humanised antibodies |
| US6582959B2 (en) | 1991-03-29 | 2003-06-24 | Genentech, Inc. | Antibodies to vascular endothelial cell growth factor |
| OA10149A (en) | 1991-03-29 | 1996-12-18 | Genentech Inc | Vascular endothelial cell growth factor antagonists |
| EP0940468A1 (en) | 1991-06-14 | 1999-09-08 | Genentech, Inc. | Humanized antibody variable domain |
| EP0666868B2 (en) | 1992-10-28 | 2006-06-14 | Genentech, Inc. | Use of anti-VEGF antibodies for the treatment of cancer |
| IL117645A (en) | 1995-03-30 | 2005-08-31 | Genentech Inc | Vascular endothelial cell growth factor antagonists for use as medicaments in the treatment of age-related macular degeneration |
| US5730977A (en) | 1995-08-21 | 1998-03-24 | Mitsui Toatsu Chemicals, Inc. | Anti-VEGF human monoclonal antibody |
| DE122004000004I1 (de) | 1996-02-09 | 2004-08-12 | Abott Biotechnology Ltd | Humane Antikörper welche an Humanen TNFalpha binden. |
| CA2267620A1 (en) * | 1996-07-16 | 1998-01-22 | Andreas Pluckthun | Immunoglobulin superfamily domains and fragments with increased solubility |
| US6986890B1 (en) | 1996-11-21 | 2006-01-17 | Kyowa Hakko Kogyo Co., Ltd. | Anti-human VEGF receptor Flt-1 monoclonal antibody |
| CN101665536B (zh) * | 1997-04-07 | 2013-07-03 | 基因技术股份有限公司 | 抗-血管内皮生长因子的抗体 |
| KR100870353B1 (ko) * | 1997-04-07 | 2008-11-25 | 제넨테크, 인크. | 항-vegf 항체 |
| US20020032315A1 (en) | 1997-08-06 | 2002-03-14 | Manuel Baca | Anti-vegf antibodies |
| US20070059302A1 (en) | 1997-04-07 | 2007-03-15 | Genentech, Inc. | Anti-vegf antibodies |
| US6703020B1 (en) | 1999-04-28 | 2004-03-09 | Board Of Regents, The University Of Texas System | Antibody conjugate methods for selectively inhibiting VEGF |
| ES2223705T3 (es) | 1999-04-28 | 2005-03-01 | Board Of Regents, The University Of Texas System | Composiciones y metodos para el tratamiento de cancer mediante inhibi cion selectiva del vegf. |
| WO2001030393A2 (en) | 1999-10-22 | 2001-05-03 | Ludwig Institute For Cancer Research | Methods for reducing the effects of cancers that express a33 antigen using a33 antigen specific immunoglobulin products |
| EP1242457B1 (en) | 1999-12-28 | 2004-08-11 | Esbatech AG | Intrabodies with defined framework that is stable in a reducing environment and applications thereof |
| GB0013810D0 (en) | 2000-06-06 | 2000-07-26 | Celltech Chiroscience Ltd | Biological products |
| US7667004B2 (en) | 2001-04-17 | 2010-02-23 | Abmaxis, Inc. | Humanized antibodies against vascular endothelial growth factor |
| DE10201450B4 (de) | 2002-01-16 | 2004-09-02 | Infineon Technologies Ag | Carry-Skip-Addierer für verschlüsselte Daten |
| US6576941B1 (en) | 2002-02-20 | 2003-06-10 | Samsung Electronics Co., Ltd. | Ferroelectric capacitors on protruding portions of conductive plugs having a smaller cross-sectional size than base portions thereof |
| CN1187373C (zh) * | 2002-03-20 | 2005-02-02 | 上海中信国健药业有限公司 | 人源化抗血管内皮生长因子单克隆抗体及其制法和药物组合物 |
| CN1305905C (zh) | 2002-03-22 | 2007-03-21 | Aprogen株式会社 | 人源化抗体及其制备方法 |
| EP2332989B1 (en) * | 2002-05-22 | 2015-04-01 | ESBATech, an Alcon Biomedical Research Unit LLC | Immunoglobulin frameworks which demonstrate enhanced stability in the intracellular environment and methods of identifying same |
| CA2492524A1 (en) * | 2002-08-15 | 2004-02-26 | Epitomics, Inc. | Humanized rabbit antibodies |
| KR20160014775A (ko) | 2003-05-30 | 2016-02-11 | 제넨테크, 인크. | 항-vegf 항체를 사용한 치료 |
| US20050048578A1 (en) | 2003-06-26 | 2005-03-03 | Epitomics, Inc. | Methods of screening for monoclonal antibodies with desirable activity |
| US20050106667A1 (en) | 2003-08-01 | 2005-05-19 | Genentech, Inc | Binding polypeptides with restricted diversity sequences |
| WO2005044853A2 (en) * | 2003-11-01 | 2005-05-19 | Genentech, Inc. | Anti-vegf antibodies |
| US7758859B2 (en) | 2003-08-01 | 2010-07-20 | Genentech, Inc. | Anti-VEGF antibodies |
| AU2003259718A1 (en) | 2003-08-07 | 2005-03-07 | Epitomics, Inc. | Methods for humanizing rabbit monoclonal antibodies |
| DK1781321T3 (da) | 2004-08-02 | 2014-04-14 | Zenyth Operations Pty Ltd | Fremgangsmåde til behandling af cancer, der omfatter en vegf-b-antagonist |
| RS52539B (sr) | 2004-10-21 | 2013-04-30 | Genentech Inc. | Metod za tretiranje intraokularnih neovaskularnih bolesti |
| US7462697B2 (en) | 2004-11-08 | 2008-12-09 | Epitomics, Inc. | Methods for antibody engineering |
| US7431927B2 (en) | 2005-03-24 | 2008-10-07 | Epitomics, Inc. | TNFα-neutralizing antibodies |
| RU2008110060A (ru) * | 2005-08-15 | 2009-09-27 | Арана Терапьютикс Лимитед (Au) | Сконструированные антитела с каркасными участками приматов нового света |
| AR059066A1 (es) | 2006-01-27 | 2008-03-12 | Amgen Inc | Combinaciones del inhibidor de la angiopoyetina -2 (ang2) y el inhibidor del factor de crecimiento endotelial vascular (vegf) |
| WO2007140534A1 (en) * | 2006-06-08 | 2007-12-13 | Csl Limited | Vegf-a cross-reactive anti- vegf-b antibodies as antagonists of vegf-a and vegf-b signalling |
| DK2046382T3 (en) | 2006-07-10 | 2016-12-12 | Esbatech Alcon Biomed Res Unit | scFv Antibodies which passes through the epithelial and / or endothelial layer |
| WO2008063932A2 (en) | 2006-11-10 | 2008-05-29 | Genentech, Inc. | Method for treating age-related macular degeneration |
| US7553486B2 (en) | 2006-11-13 | 2009-06-30 | Paul Theodore Finger | Anti-VEGF treatment for radiation-induced vasculopathy |
| HUE029445T2 (en) | 2006-12-19 | 2017-02-28 | Genentech Inc | VEGF-specific antagonists for adjuvant and neoadjuvant therapy and treatment of early-stage tumors |
| JP5622397B2 (ja) | 2007-03-12 | 2014-11-12 | エスバテック− ア ノバルティス カンパニー エルエルシー | 一本鎖抗体の配列に基づく操作および最適化 |
| WO2008149147A2 (en) | 2007-06-06 | 2008-12-11 | Domantis Limited | Polypeptides, antibody variable domains and antagonists |
| EA200901494A1 (ru) | 2007-06-06 | 2010-06-30 | Домантис Лимитед | Способы селекции протеазоустойчивых полипептидов |
| DK2158315T3 (en) | 2007-06-25 | 2016-06-06 | Esbatech Alcon Biomed Res Unit | Methods of modifying antibodies and modified antibodies with improved functional properties |
| AU2008267733B2 (en) | 2007-06-25 | 2013-11-14 | Esbatech, An Alcon Biomedical Research Unit Llc | Sequence based engineering and optimization of single chain antibodies |
| WO2009120178A1 (en) * | 2008-03-26 | 2009-10-01 | Epitomics, Inc. | Anti-vegf antibody |
| US9556265B2 (en) | 2008-06-25 | 2017-01-31 | Esbatech, An Alcon Biomedical Research Unit Llc | Solubility optimization of immunobinders |
| HUE032894T2 (hu) | 2008-06-25 | 2017-11-28 | Esbatech Alcon Biomed Res Unit | VEGF-gátló, stabilis és oldható antitestek |
| CN104004094B (zh) * | 2008-06-25 | 2017-09-22 | 艾斯巴技术-诺华有限责任公司 | 使用通用抗体构架进行的兔抗体的人源化 |
| US8697074B2 (en) * | 2008-07-10 | 2014-04-15 | Esbatech, An Alcon Biomedical Research Unit Llc | Methods and compositions for enhanced delivery of macromolecules |
| KR101758703B1 (ko) * | 2009-12-23 | 2017-07-18 | 에스바테크 - 어 노바티스 컴파니 엘엘씨 | 면역원성의 감소 방법 |
-
2009
- 2009-06-25 HU HUE09768693A patent/HUE032894T2/hu unknown
- 2009-06-25 MX MX2013004871A patent/MX347972B/es unknown
- 2009-06-25 ES ES20158159T patent/ES2998307T3/es active Active
- 2009-06-25 ES ES09768693.5T patent/ES2622470T3/es active Active
- 2009-06-25 KR KR1020187024733A patent/KR102095257B1/ko active Active
- 2009-06-25 KR KR1020187000324A patent/KR20180005753A/ko not_active Abandoned
- 2009-06-25 US US13/000,423 patent/US8349322B2/en active Active
- 2009-06-25 KR KR1020177012884A patent/KR101817284B1/ko active Active
- 2009-06-25 MX MX2017006583A patent/MX392435B/es unknown
- 2009-06-25 CN CN201510205513.0A patent/CN104961828B/zh active Active
- 2009-06-25 DK DK17151756.8T patent/DK3216803T3/da active
- 2009-06-25 PT PT171517568T patent/PT3216803T/pt unknown
- 2009-06-25 PT PT201581592T patent/PT3722310T/pt unknown
- 2009-06-25 PL PL09768693T patent/PL2307454T3/pl unknown
- 2009-06-25 CA CA2727839A patent/CA2727839C/en active Active
- 2009-06-25 DK DK20158159.2T patent/DK3722310T3/da active
- 2009-06-25 CA CA3020290A patent/CA3020290A1/en active Pending
- 2009-06-25 EP EP09768693.5A patent/EP2307454B1/en active Active
- 2009-06-25 CN CN200980124313.5A patent/CN102143976B/zh active Active
- 2009-06-25 HU HUE20158159A patent/HUE068894T2/hu unknown
- 2009-06-25 CN CN201910338494.7A patent/CN110372792A/zh active Pending
- 2009-06-25 SI SI200932061T patent/SI3216803T1/sl unknown
- 2009-06-25 FI FIEP20158159.2T patent/FI3722310T3/fi active
- 2009-06-25 SI SI200932208T patent/SI3722310T1/sl unknown
- 2009-06-25 KR KR1020157032314A patent/KR101671886B1/ko active Active
- 2009-06-25 ES ES17151756T patent/ES2793008T3/es active Active
- 2009-06-25 DK DK09768693.5T patent/DK2307454T3/en active
- 2009-06-25 AU AU2009264565A patent/AU2009264565C1/en active Active
- 2009-06-25 SI SI200931647A patent/SI2307454T1/sl unknown
- 2009-06-25 WO PCT/CH2009/000220 patent/WO2009155724A2/en not_active Ceased
- 2009-06-25 PT PT97686935T patent/PT2307454T/pt unknown
- 2009-06-25 LT LTEP09768693.5T patent/LT2307454T/lt unknown
- 2009-06-25 KR KR1020197022648A patent/KR102157097B1/ko active Active
- 2009-06-25 MX MX2011000011A patent/MX2011000011A/es active IP Right Grant
- 2009-06-25 PL PL20158159.2T patent/PL3722310T3/pl unknown
- 2009-06-25 EP EP17151756.8A patent/EP3216803B1/en active Active
- 2009-06-25 LT LTEP17151756.8T patent/LT3216803T/lt unknown
- 2009-06-25 KR KR1020117001961A patent/KR102013220B1/ko active Active
- 2009-06-25 JP JP2011515049A patent/JP5956752B2/ja active Active
- 2009-06-25 HU HUE17151756A patent/HUE050230T2/hu unknown
- 2009-06-25 LT LTEP20158159.2T patent/LT3722310T/lt unknown
- 2009-06-25 KR KR1020207026266A patent/KR102296443B1/ko active Active
- 2009-06-25 HR HRP20170615TT patent/HRP20170615T1/hr unknown
- 2009-06-25 RU RU2011102583A patent/RU2531523C3/ru active
- 2009-06-25 RU RU2014132888A patent/RU2588467C3/ru active Protection Beyond IP Right Term
- 2009-06-25 PL PL17151756T patent/PL3216803T3/pl unknown
- 2009-06-25 BR BRPI0914251-7A patent/BRPI0914251B1/pt active IP Right Grant
- 2009-06-25 HR HRP20241467TT patent/HRP20241467T1/hr unknown
- 2009-06-25 EP EP20158159.2A patent/EP3722310B1/en active Active
- 2009-06-25 KR KR1020167030125A patent/KR101737466B1/ko active Active
-
2010
- 2010-11-30 ZA ZA2010/08594A patent/ZA201008594B/en unknown
- 2010-12-23 CL CL2010001544A patent/CL2010001544A1/es unknown
-
2012
- 2012-12-07 US US13/708,575 patent/US9090684B2/en active Active
-
2013
- 2013-07-19 RU RU2013133802/10A patent/RU2013133802A/ru not_active Application Discontinuation
-
2014
- 2014-04-21 JP JP2014087194A patent/JP6100199B2/ja active Active
-
2015
- 2015-02-27 JP JP2015037794A patent/JP2015134792A/ja active Pending
- 2015-06-16 US US14/741,430 patent/US9873737B2/en active Active
- 2015-07-20 PH PH12015501593A patent/PH12015501593A1/en unknown
-
2016
- 2016-05-18 RU RU2016119152A patent/RU2648152C2/ru active
- 2016-12-28 JP JP2016255720A patent/JP6527128B2/ja active Active
-
2017
- 2017-04-12 CY CY20171100436T patent/CY1118829T1/el unknown
- 2017-11-16 US US15/814,784 patent/US10590193B2/en active Active
-
2018
- 2018-02-05 JP JP2018018375A patent/JP6821612B2/ja active Active
- 2018-02-16 RU RU2018105866A patent/RU2696972C1/ru active
-
2019
- 2019-05-13 JP JP2019090735A patent/JP6978468B2/ja active Active
- 2019-06-27 RU RU2019120079A patent/RU2747735C3/ru active
-
2020
- 2020-01-31 US US16/779,028 patent/US20200172608A1/en not_active Abandoned
- 2020-05-19 HR HRP20200814TT patent/HRP20200814T1/hr unknown
- 2020-06-05 CY CY20201100507T patent/CY1123028T1/el unknown
- 2020-07-23 LT LTPA2020004C patent/LTC2307454I2/lt unknown
- 2020-07-27 HU HUS2000028C patent/HUS2000028I1/hu unknown
- 2020-07-28 NL NL301058C patent/NL301058I2/nl unknown
- 2020-07-30 CY CY2020026C patent/CY2020026I2/el unknown
- 2020-08-04 NO NO2020021C patent/NO2020021I1/no unknown
-
2021
- 2021-11-11 JP JP2021184262A patent/JP7171877B6/ja active Active
-
2023
- 2023-02-07 US US18/165,845 patent/US20250066462A1/en active Pending
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2000034337A1 (en) * | 1998-12-10 | 2000-06-15 | Tsukuba Research Laboratory, Toagosei Co., Ltd. | Humanized monoclonal antibodies against vascular endothelial cell growth factor |
| WO2000053760A2 (en) * | 1999-03-12 | 2000-09-14 | Genentech, Inc. | Method of preventing the death of retinal neurons and treating ocular diseases |
Non-Patent Citations (2)
| Title |
|---|
| JBC Vol.281(2):951-961(2006. 01. 13.) * |
| Structure. Vol.6(9):1153-1167(1998. 09. 15.)* * |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7171877B6 (ja) | Vegfを阻害する安定かつ可溶性の抗体 | |
| RU2588467C2 (ru) | Стабильные и растворимые антитела, ингибирующие vegf | |
| AU2021202343B2 (en) | Stable and soluble antibodies inhibiting vegf | |
| HK1240240A1 (en) | Stable and soluble antibodies inhibiting vegf | |
| HK40030913A (en) | Stable and soluble antibodies inhibiting vegf |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A107 | Divisional application of patent | ||
| PA0104 | Divisional application for international application |
St.27 status event code: A-0-1-A10-A18-div-PA0104 St.27 status event code: A-0-1-A10-A16-div-PA0104 |
|
| PG1501 | Laying open of application |
St.27 status event code: A-1-1-Q10-Q12-nap-PG1501 |
|
| A201 | Request for examination | ||
| PA0201 | Request for examination |
St.27 status event code: A-1-2-D10-D11-exm-PA0201 |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
St.27 status event code: A-1-2-D10-D21-exm-PE0902 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| AMND | Amendment | ||
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
St.27 status event code: N-2-6-B10-B15-exm-PE0601 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| T13-X000 | Administrative time limit extension granted |
St.27 status event code: U-3-3-T10-T13-oth-X000 |
|
| A107 | Divisional application of patent | ||
| AMND | Amendment | ||
| E13-X000 | Pre-grant limitation requested |
St.27 status event code: A-2-3-E10-E13-lim-X000 |
|
| J201 | Request for trial against refusal decision | ||
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| PA0104 | Divisional application for international application |
St.27 status event code: A-0-1-A10-A18-div-PA0104 St.27 status event code: A-0-1-A10-A16-div-PA0104 |
|
| PJ0201 | Trial against decision of rejection |
St.27 status event code: A-3-3-V10-V11-apl-PJ0201 |
|
| PB0901 | Examination by re-examination before a trial |
St.27 status event code: A-6-3-E10-E12-rex-PB0901 |
|
| E90F | Notification of reason for final refusal | ||
| PE0902 | Notice of grounds for rejection |
St.27 status event code: A-1-2-D10-D21-exm-PE0902 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-3-3-R10-R13-asn-PN2301 St.27 status event code: A-3-3-R10-R11-asn-PN2301 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| E13-X000 | Pre-grant limitation requested |
St.27 status event code: A-2-3-E10-E13-lim-X000 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| B701 | Decision to grant | ||
| PB0701 | Decision of registration after re-examination before a trial |
St.27 status event code: A-3-4-F10-F13-rex-PB0701 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
St.27 status event code: A-2-4-F10-F11-exm-PR0701 |
|
| PR1002 | Payment of registration fee |
St.27 status event code: A-2-2-U10-U12-oth-PR1002 Fee payment year number: 1 |
|
| PG1601 | Publication of registration |
St.27 status event code: A-4-4-Q10-Q13-nap-PG1601 |
|
| S17-X000 | Non-exclusive voluntary license recorded |
St.27 status event code: A-4-4-S10-S17-lic-X000 |
|
| A101 | Application to extend term of patent right by permit | ||
| PA0101 | Application to register extension of term of patent right by permit, etc. |
St.27 status event code: A-4-4-G10-G18-tex-PA0101 Protection beyond ip right term event data comment text: Claim Total Quantity : 14, Claim Number : 1 8, 19 24, Period Limitation Text : 82, Comment Text : 5123 |
|
| PE0702 | Decision to register extension of term of patent right by permit, etc. |
St.27 status event code: A-4-4-G10-G20-tex-PE0702 Protection beyond ip right term event data comment text: Claim Total Quantity : 14, Claim Number : 1 8, 19 24, Period Limitation Text : 82, Comment Text : 5123 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 4 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 5 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 6 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 7 |
|
| U11 | Full renewal or maintenance fee paid |
Free format text: ST27 STATUS EVENT CODE: A-4-4-U10-U11-OTH-PR1001 (AS PROVIDED BY THE NATIONAL OFFICE) Year of fee payment: 7 |