KR20200044902A - 서방형 마취제 조성물 및 이의 제조 방법 - Google Patents
서방형 마취제 조성물 및 이의 제조 방법 Download PDFInfo
- Publication number
- KR20200044902A KR20200044902A KR1020207008714A KR20207008714A KR20200044902A KR 20200044902 A KR20200044902 A KR 20200044902A KR 1020207008714 A KR1020207008714 A KR 1020207008714A KR 20207008714 A KR20207008714 A KR 20207008714A KR 20200044902 A KR20200044902 A KR 20200044902A
- Authority
- KR
- South Korea
- Prior art keywords
- anesthetic
- amide
- composition
- ropivacaine
- type
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/127—Synthetic bilayered vehicles, e.g. liposomes or liposomes with cholesterol as the only non-phosphatidyl surfactant
- A61K9/1271—Non-conventional liposomes, e.g. PEGylated liposomes or liposomes coated or grafted with polymers
- A61K9/1273—Polymersomes; Liposomes with polymerisable or polymerised bilayer-forming substances
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/16—Amides, e.g. hydroxamic acids
- A61K31/165—Amides, e.g. hydroxamic acids having aromatic rings, e.g. colchicine, atenolol, progabide
- A61K31/167—Amides, e.g. hydroxamic acids having aromatic rings, e.g. colchicine, atenolol, progabide having the nitrogen of a carboxamide group directly attached to the aromatic ring, e.g. lidocaine, paracetamol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/445—Non condensed piperidines, e.g. piperocaine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/22—Heterocyclic compounds, e.g. ascorbic acid, tocopherol or pyrrolidones
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/24—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing atoms other than carbon, hydrogen, oxygen, halogen, nitrogen or sulfur, e.g. cyclomethicone or phospholipids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/28—Steroids, e.g. cholesterol, bile acids or glycyrrhetinic acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/06—Ointments; Bases therefor; Other semi-solid forms, e.g. creams, sticks, gels
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/107—Emulsions ; Emulsion preconcentrates; Micelles
- A61K9/1075—Microemulsions or submicron emulsions; Preconcentrates or solids thereof; Micelles, e.g. made of phospholipids or block copolymers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/127—Synthetic bilayered vehicles, e.g. liposomes or liposomes with cholesterol as the only non-phosphatidyl surfactant
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/127—Synthetic bilayered vehicles, e.g. liposomes or liposomes with cholesterol as the only non-phosphatidyl surfactant
- A61K9/1271—Non-conventional liposomes, e.g. PEGylated liposomes or liposomes coated or grafted with polymers
- A61K9/1272—Non-conventional liposomes, e.g. PEGylated liposomes or liposomes coated or grafted with polymers comprising non-phosphatidyl surfactants as bilayer-forming substances, e.g. cationic lipids or non-phosphatidyl liposomes coated or grafted with polymers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P23/00—Anaesthetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P23/00—Anaesthetics
- A61P23/02—Local anaesthetics
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Molecular Biology (AREA)
- Biophysics (AREA)
- Dispersion Chemistry (AREA)
- Anesthesiology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Dermatology (AREA)
- Pain & Pain Management (AREA)
- Medicinal Preparation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Description
도 1은 로피바카인 조성물(pH 5.5 히스티딘 용액으로 수화됨, 닫힌 사각형; pH 6.0 히스티딘 용액으로 수화됨, 닫힌 삼각형; 및 pH 6.5 히스티딘 용액으로 수화됨, 닫힌 원)의 피하(SC) 주사 후 또는 제형화되지 않은 로피바카인(열린 다이아몬드)의 SC 주사 후 래트에서의 로피바카인의 혈장 농도를 나타내는 그래프이다(모든 데이터는 평균 ± 표준 편차(SD)로 표시됨);
도 2a 및 2b는 기계적 자극에 대한 마우스 발 움츠림 임계값에 대한 로피바카인 조성물(원), 로피바카인(사각형) 및 비히클(삼각형)의 SC 투여 후 효과를 도시한 일련의 그래프이다(모든 데이터는 평균 ± 평균의 표준 오차(SEM)로 표시됨); 도 2a는 시간 대 움츠림 임계값(g)의 그래픽 플롯이다; 도 2b는 시간 대 기계적 임계값의 변화(%)의 그래픽 플롯이다; 그리고
도 3a 및 3b는 동일한 투여량의 로피바카인과 비교하여 로피바카인 조성물의 단일 피내(IC) 주사 후 시간에 따른 마취 효과를 나타내는 일련의 그래프이다(모든 데이터는 평균 ± SEM로 표시됨); 도 3a는 로피바카인 조성물(닫힌 사각형) 또는 로피바카인(열린 사각형)의 IC 팽진당 3.0 mg으로 투여된 기니피그 코호트에 대한 마취 효과를 도시한다; 도 3b는 로피바카인 조성물(닫힌 삼각형) 또는 로피바카인(열린 삼각형)의 IC 팽진당 1.5 mg으로 투여된 기니피그 코호트에 대한 마취 효과를 도시한다.
Claims (16)
- 서방형 마취제 조성물을 제조하는 방법으로서,
적어도 하나의 아미드-타입 마취제, 및
적어도 하나의 인지질을 포함하는 지질 혼합물을 포함하는 고도로 포획된 지질 구조체(HELS)를 생성하는 단계, 및
상기 HELS를 pH 5.5 내지 8.0의 완충 수용액으로 수화시키는 단계를 포함하고; 여기서 상기 HELS를 수화시키는 것이 포획된 아미드-타입 마취제를 갖는 다층 소포(MLV)를 형성하고; 포획된 아미드-타입 마취제를 갖는 상기 MLV의 중간 직경이 적어도 1 μm인, 방법. - 제1항에 있어서, HELS가 케이크, 분말, 비-필름 고체 벌크, 또는 이들의 조합의 형태인 방법.
- 제1항에 있어서, 포획된 아미드-타입 마취제를 갖는 MLV 중 아미드-타입 마취제 대 인지질의 몰비가 적어도 0.5:1인 방법.
- 제1항에 있어서, 상기 HELS를 생성하는 방법이
지질 혼합물 및 적어도 하나의 아미드-타입 마취제를 용매 시스템에 용해시켜 액체 구조를 형성하는 단계; 및
상기 액체 구조로부터 상기 용매 시스템을 제거하는 단계를 포함하는 방법. - 제4항에 있어서, 용매 시스템을 제거하는 단계가 액체 구조를 동결건조 또는 분무 건조하는 것을 포함하는 방법.
- 제4항에 있어서, 용매 시스템이 3차-부탄올 또는 3차-부탄올/물 공용매를 포함하는 방법.
- 제1항에 있어서, 지질 혼합물이 콜레스테롤을 포함하는 방법.
- 제7항에 있어서, 지질 혼합물 중 콜레스테롤의 몰 백분율이 50% 이하인 방법.
- 제7항에 있어서, 적어도 하나의 인지질 및 콜레스테롤이 1:0.01 내지 1:1의 몰비로 존재하는 방법.
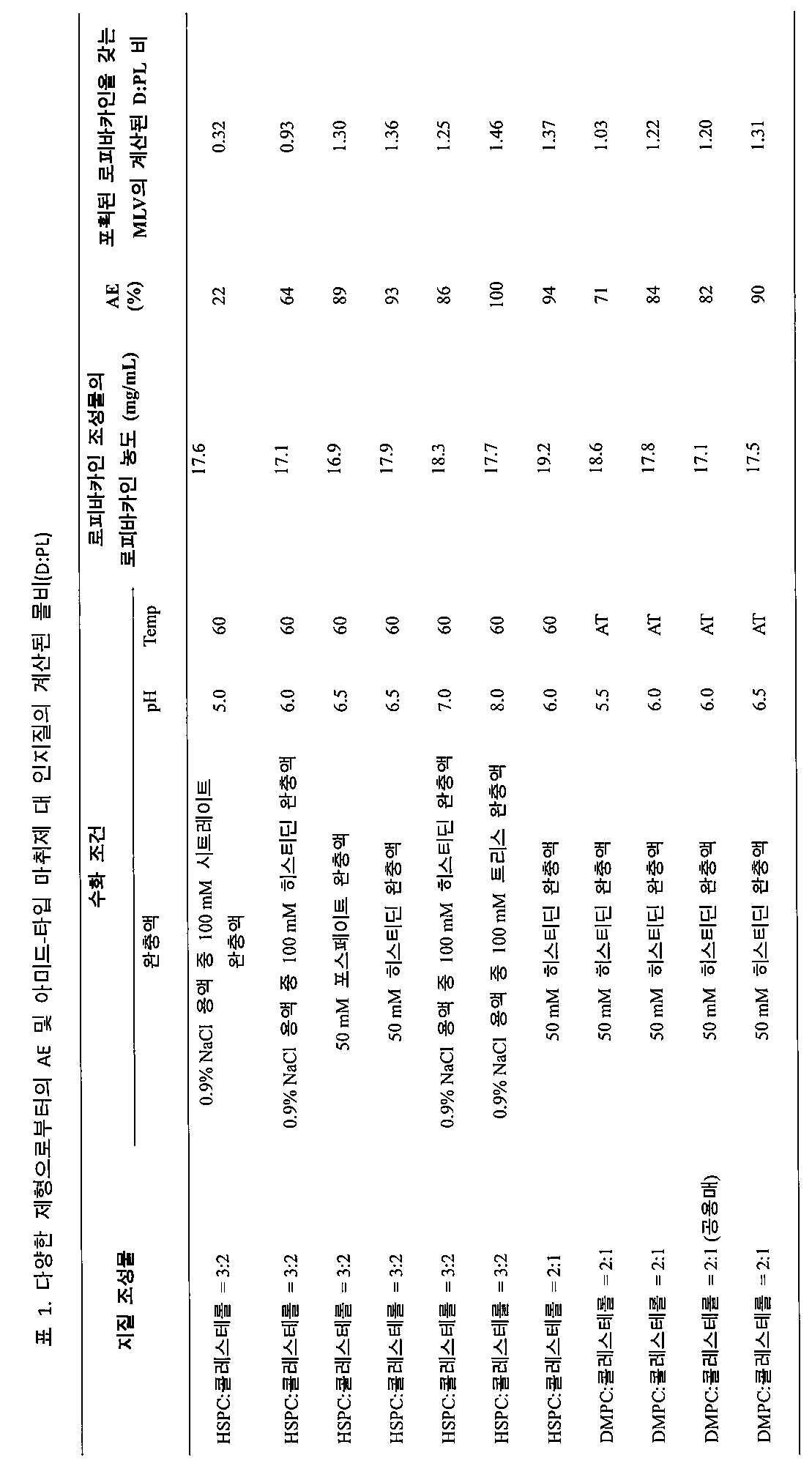
- 제1항 내지 제9항 중 어느 한 항에 있어서, 적어도 하나의 아미드-타입 마취제가 리도카인, 부피바카인, 레보부피바카인, 로피바카인, 메피바카인, 피로카인, 아티카인, 또는 프리로카인인 방법.
- 제1항 내지 제9항 중 어느 한 항에 있어서, 적어도 하나의 아미드-타입 마취제가 로피바카인 염기인 방법.
- 제1항 내지 제9항 중 어느 한 항에 있어서, 완충 수용액이 히스티딘을 1 mM 내지 200 mM 범위의 농도로 포함하는 방법.
- 국소 마취제를 필요로 하는 대상체에게 국소 마취제를 국소 투여하기 위한 서방형 마취제 조성물로서, 상기 조성물이 제1항 내지 제9항 중 어느 한 항에 따른 방법에 의해 제조되고 상기 마취제 조성물 중 포획된 아미드-타입 마취제를 갖는 MLV의 중간 직경이 1 μm 이상인, 서방형 마취제 조성물.
- 제13항에 있어서, 포획된 아미드-타입 마취제를 갖는 MLV 중 아미드-타입 마취제 대 인지질의 몰비가 0.5:1 이상인 서방형 마취제 조성물.
- 제13항에 있어서, 적어도 하나의 아미드-타입 마취제가 로피바카인 염기인 서방형 마취제 조성물.
- 제13항에 있어서, 완충 수용액이 히스티딘을 1 mM 내지 200 mM 범위의 농도로 포함하는 서방형 마취제 조성물.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020257001950A KR20250029892A (ko) | 2017-08-28 | 2018-08-28 | 서방형 마취제 조성물 및 이의 제조 방법 |
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201762550983P | 2017-08-28 | 2017-08-28 | |
| US62/550,983 | 2017-08-28 | ||
| US201862621730P | 2018-01-25 | 2018-01-25 | |
| US62/621,730 | 2018-01-25 | ||
| PCT/US2018/048329 WO2019046293A1 (en) | 2017-08-28 | 2018-08-28 | EXTENDED RELEASE ANESTHETIC COMPOSITIONS AND METHODS OF PREPARATION THEREOF |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020257001950A Division KR20250029892A (ko) | 2017-08-28 | 2018-08-28 | 서방형 마취제 조성물 및 이의 제조 방법 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20200044902A true KR20200044902A (ko) | 2020-04-29 |
| KR102800917B1 KR102800917B1 (ko) | 2025-04-24 |
Family
ID=63668016
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020207008714A Active KR102800917B1 (ko) | 2017-08-28 | 2018-08-28 | 서방형 마취제 조성물 및 이의 제조 방법 |
| KR1020257001950A Pending KR20250029892A (ko) | 2017-08-28 | 2018-08-28 | 서방형 마취제 조성물 및 이의 제조 방법 |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020257001950A Pending KR20250029892A (ko) | 2017-08-28 | 2018-08-28 | 서방형 마취제 조성물 및 이의 제조 방법 |
Country Status (22)
| Country | Link |
|---|---|
| US (2) | US11793799B2 (ko) |
| EP (1) | EP3675816B1 (ko) |
| JP (1) | JP7173615B2 (ko) |
| KR (2) | KR102800917B1 (ko) |
| CN (2) | CN111182889A (ko) |
| AU (2) | AU2018323436B2 (ko) |
| CA (1) | CA3073734A1 (ko) |
| CY (1) | CY1126137T1 (ko) |
| DK (1) | DK3675816T3 (ko) |
| ES (1) | ES2946919T3 (ko) |
| FI (1) | FI3675816T3 (ko) |
| HR (1) | HRP20230535T1 (ko) |
| HU (1) | HUE062202T2 (ko) |
| LT (1) | LT3675816T (ko) |
| MX (1) | MX393126B (ko) |
| PL (1) | PL3675816T3 (ko) |
| PT (1) | PT3675816T (ko) |
| RS (1) | RS64297B1 (ko) |
| SI (1) | SI3675816T1 (ko) |
| SM (1) | SMT202300163T1 (ko) |
| TW (1) | TWI806894B (ko) |
| WO (1) | WO2019046293A1 (ko) |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| PT3675816T (pt) * | 2017-08-28 | 2023-05-31 | Taiwan Liposome Co Ltd | Composições anestésicas de libertação prolongada e métodos de preparação das mesmas |
| KR102780006B1 (ko) * | 2018-03-30 | 2025-03-10 | 티엘씨 바이오파머슈티컬즈 인코포레이티드 | 서방형 마취제 조성물 및 이의 제조 방법 |
| CN113473972A (zh) * | 2019-02-26 | 2021-10-01 | 美商Tlc生物医药公司 | 用于治疗疼痛的药学组成物 |
| WO2020259670A1 (zh) | 2019-06-28 | 2020-12-30 | 江苏恒瑞医药股份有限公司 | 一种缓释脂质组合物及其制备方法 |
| CN113116823B (zh) * | 2019-12-30 | 2024-02-20 | 江苏恒瑞医药股份有限公司 | 一种脂质体及其制备方法 |
| WO2021143745A1 (zh) | 2020-01-14 | 2021-07-22 | 中国科学院上海药物研究所 | 一种长效罗哌卡因药物组合物及其制备方法和用途 |
| CN114668723A (zh) * | 2020-12-25 | 2022-06-28 | 江苏恒瑞医药股份有限公司 | 一种含有局部麻醉药的脂质体及其制备方法 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1997034582A1 (en) * | 1996-03-21 | 1997-09-25 | Vrije Universiteit Brussel | Liposome encapsulated amphiphilic drug compositions |
| WO2008040556A1 (en) * | 2006-10-06 | 2008-04-10 | Scil Technology Gmbh | Dried reconstituted vesicle formation for pharmaceutical application |
| US20150250724A1 (en) * | 2012-09-21 | 2015-09-10 | Kansai Medical University Educational Corporation | Long-lasting, controlled-release local anesthetic liposome preparation |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5244678A (en) | 1986-01-14 | 1993-09-14 | Ire-Celltarg S.A. | Pharmaceutical composition containing a local anesthetic and/or centrally acting analgesic encapsulated in liposomes |
| FR2592791A1 (fr) * | 1986-01-14 | 1987-07-17 | Ire Celltarg Sa | Composition pharmaceutique, contenant un anesthesique local et/ou un analgesique central encapsule dans des liposomes |
| US5227165A (en) * | 1989-11-13 | 1993-07-13 | Nova Pharmaceutical Corporation | Liposphere delivery systems for local anesthetics |
| GB9609779D0 (en) | 1996-05-10 | 1996-07-17 | Univ Bruxelles | Freeze dried liposome encapsulated amphiphilic drug compositions and a process for the preparation thereof |
| ATE535232T1 (de) | 1997-09-18 | 2011-12-15 | Pacira Pharmaceuticals Inc | Retardierte freisetzung liposomaler anesthetischer zusammensetzungen |
| IL138672A0 (en) | 1998-03-31 | 2001-10-31 | Yissum Res Dev Co | Liposomal bupivacaine compositions and methods of preparation |
| IL140892A0 (en) | 1998-08-12 | 2002-02-10 | Yissum Res Dev Co | Liposomal bupivacaine compositions prepared using an ammonium sulfate gradient |
| JPWO2013146386A1 (ja) * | 2012-03-27 | 2015-12-10 | 学校法人関西医科大学 | 局所麻酔薬持続性徐放製剤 |
| WO2014043665A2 (en) | 2012-09-17 | 2014-03-20 | Interdigital Patent Holdings, Inc. | Self-optimization of backhaul radio reseources and small cell backhaul delay estimation |
| CN103142458B (zh) * | 2013-01-22 | 2015-09-09 | 莱普德制药有限公司 | 无成瘾性镇痛缓释递药系统的组方与制备方法 |
| US11083730B2 (en) | 2014-04-21 | 2021-08-10 | Heron Therapeutics, Inc. | Long-acting polymeric delivery systems |
| PT3675816T (pt) * | 2017-08-28 | 2023-05-31 | Taiwan Liposome Co Ltd | Composições anestésicas de libertação prolongada e métodos de preparação das mesmas |
-
2018
- 2018-08-28 PT PT187736921T patent/PT3675816T/pt unknown
- 2018-08-28 DK DK18773692.1T patent/DK3675816T3/da active
- 2018-08-28 FI FIEP18773692.1T patent/FI3675816T3/fi active
- 2018-08-28 KR KR1020207008714A patent/KR102800917B1/ko active Active
- 2018-08-28 MX MX2020002231A patent/MX393126B/es unknown
- 2018-08-28 TW TW107130015A patent/TWI806894B/zh active
- 2018-08-28 EP EP18773692.1A patent/EP3675816B1/en active Active
- 2018-08-28 JP JP2020511474A patent/JP7173615B2/ja active Active
- 2018-08-28 CA CA3073734A patent/CA3073734A1/en active Pending
- 2018-08-28 CN CN201880055567.5A patent/CN111182889A/zh active Pending
- 2018-08-28 ES ES18773692T patent/ES2946919T3/es active Active
- 2018-08-28 WO PCT/US2018/048329 patent/WO2019046293A1/en not_active Ceased
- 2018-08-28 PL PL18773692.1T patent/PL3675816T3/pl unknown
- 2018-08-28 SI SI201830921T patent/SI3675816T1/sl unknown
- 2018-08-28 HU HUE18773692A patent/HUE062202T2/hu unknown
- 2018-08-28 RS RS20230409A patent/RS64297B1/sr unknown
- 2018-08-28 AU AU2018323436A patent/AU2018323436B2/en active Active
- 2018-08-28 US US16/642,762 patent/US11793799B2/en active Active
- 2018-08-28 KR KR1020257001950A patent/KR20250029892A/ko active Pending
- 2018-08-28 HR HRP20230535TT patent/HRP20230535T1/hr unknown
- 2018-08-28 CN CN202410245391.7A patent/CN118105349A/zh active Pending
- 2018-08-28 SM SM20230163T patent/SMT202300163T1/it unknown
- 2018-08-28 LT LTEPPCT/US2018/048329T patent/LT3675816T/lt unknown
-
2023
- 2023-06-19 CY CY20231100287T patent/CY1126137T1/el unknown
- 2023-09-13 US US18/466,226 patent/US12280043B2/en active Active
- 2023-11-14 AU AU2023266253A patent/AU2023266253B2/en active Active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1997034582A1 (en) * | 1996-03-21 | 1997-09-25 | Vrije Universiteit Brussel | Liposome encapsulated amphiphilic drug compositions |
| WO2008040556A1 (en) * | 2006-10-06 | 2008-04-10 | Scil Technology Gmbh | Dried reconstituted vesicle formation for pharmaceutical application |
| US20150250724A1 (en) * | 2012-09-21 | 2015-09-10 | Kansai Medical University Educational Corporation | Long-lasting, controlled-release local anesthetic liposome preparation |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR102800917B1 (ko) | 서방형 마취제 조성물 및 이의 제조 방법 | |
| CA1339008C (en) | Amphotericin b liposome preparation | |
| EP2394640A1 (en) | Improved liposomal formulations of lipophilic compounds | |
| CN102223878A (zh) | 包含鞘磷脂的脂质体系统 | |
| CN103370055B (zh) | 缓释性脂质体组合物及其制造方法 | |
| KR102780006B1 (ko) | 서방형 마취제 조성물 및 이의 제조 방법 | |
| JP2009513621A (ja) | リポソームを調製する方法及びその使用 | |
| KR20200065961A (ko) | 서방형 리포좀 제제의 제조방법 | |
| CN112654345A (zh) | 缓释眼用药物组合物及其用途 | |
| US20040175417A1 (en) | Amphotericin B liposome preparation | |
| RU2791481C2 (ru) | Анестетические композиции с замедленным высвобождением и способы их получения | |
| HK40110862A (zh) | 缓释麻醉剂组成物及其制备方法 | |
| HK40029534A (en) | Sustained-release anesthetic compositions and methods of preparation thereof | |
| BR122024008482B1 (pt) | Composições anestésicas de liberação sustentada | |
| BR122024008482A2 (pt) | Composições anestésicas de liberação sustentada | |
| BR112020003743B1 (pt) | Composições anestésicas de liberação sustentada e métodos de preparação dos mesmos | |
| JP2022505378A (ja) | 免疫調節剤を含む徐放性医薬組成物およびその使用 | |
| HK40046575A (en) | Sustained-release ophthalmic pharmaceutical compositions and uses thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20200325 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| A201 | Request for examination | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20210817 Comment text: Request for Examination of Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20240228 Patent event code: PE09021S01D |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
Patent event date: 20241029 Comment text: Decision to Refuse Application Patent event code: PE06012S01D |
|
| AMND | Amendment | ||
| PX0901 | Re-examination |
Patent event code: PX09012R01I Patent event date: 20250120 Comment text: Amendment to Specification, etc. |
|
| PX0701 | Decision of registration after re-examination |
Patent event date: 20250224 Comment text: Decision to Grant Registration Patent event code: PX07013S01D |
|
| X701 | Decision to grant (after re-examination) | ||
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
Comment text: Registration of Establishment Patent event date: 20250422 Patent event code: PR07011E01D |
|
| PR1002 | Payment of registration fee |
Payment date: 20250422 End annual number: 3 Start annual number: 1 |
|
| PG1601 | Publication of registration |