WO2024253016A1 - Capteur d'ions chlorure et dispositif de détection de cellules cancéreuses et/ou d'évaluation du degré de malignité de cellules cancéreuses l'utilisant - Google Patents
Capteur d'ions chlorure et dispositif de détection de cellules cancéreuses et/ou d'évaluation du degré de malignité de cellules cancéreuses l'utilisant Download PDFInfo
- Publication number
- WO2024253016A1 WO2024253016A1 PCT/JP2024/019859 JP2024019859W WO2024253016A1 WO 2024253016 A1 WO2024253016 A1 WO 2024253016A1 JP 2024019859 W JP2024019859 W JP 2024019859W WO 2024253016 A1 WO2024253016 A1 WO 2024253016A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- chloride ion
- external force
- electrode
- sensor
- cell
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12M—APPARATUS FOR ENZYMOLOGY OR MICROBIOLOGY; APPARATUS FOR CULTURING MICROORGANISMS FOR PRODUCING BIOMASS, FOR GROWING CELLS OR FOR OBTAINING FERMENTATION OR METABOLIC PRODUCTS, i.e. BIOREACTORS OR FERMENTERS
- C12M1/00—Apparatus for enzymology or microbiology
- C12M1/34—Measuring or testing with condition measuring or sensing means, e.g. colony counters
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/02—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving viable microorganisms
- C12Q1/04—Determining presence or kind of microorganism; Use of selective media for testing antibiotics or bacteriocides; Compositions containing a chemical indicator therefor
- C12Q1/06—Quantitative determination
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N27/00—Investigating or analysing materials by the use of electric, electrochemical, or magnetic means
- G01N27/26—Investigating or analysing materials by the use of electric, electrochemical, or magnetic means by investigating electrochemical variables; by using electrolysis or electrophoresis
- G01N27/28—Electrolytic cell components
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N27/00—Investigating or analysing materials by the use of electric, electrochemical, or magnetic means
- G01N27/26—Investigating or analysing materials by the use of electric, electrochemical, or magnetic means by investigating electrochemical variables; by using electrolysis or electrophoresis
- G01N27/28—Electrolytic cell components
- G01N27/30—Electrodes, e.g. test electrodes; Half-cells
- G01N27/333—Ion-selective electrodes or membranes
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N27/00—Investigating or analysing materials by the use of electric, electrochemical, or magnetic means
- G01N27/26—Investigating or analysing materials by the use of electric, electrochemical, or magnetic means by investigating electrochemical variables; by using electrolysis or electrophoresis
- G01N27/416—Systems
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/483—Physical analysis of biological material
Definitions
- the present invention relates to a chloride ion sensor and a device using the same for detecting cancer cells and/or evaluating the malignancy of cancer cells.
- Non-Patent Document 1 Various molecules that recognize chloride ions have been known.
- the ionophore Bisthiourea-1 can be used as an ion-responsive membrane in an ion-selective electrode (Non-Patent Document 1).
- this molecule needs to be supported on a resin, which poses problems in terms of diffusibility and compatibility.
- the detection limit for chloride ions when this molecule is used is at the micromolar level, which poses problems in terms of detection sensitivity.
- N-Ethoxycarbonylmethyl-6-methoxyquinolinium bromide MQAE
- 6-methoxy-N-(3-sulfopropyl)quinolinium SPQ
- MQAE N-Ethoxycarbonylmethyl-6-methoxyquinolinium bromide
- SPQ 6-methoxy-N-(3-sulfopropyl)quinolinium
- this method examines whether fixed cells are cancer cells or not, so it is not possible to diagnose how cancer cells behave in the body in a short period of time.
- the method described in the above Patent Document 1 can be used to measure the invasiveness of live, unfixed cancer cells, which could not be evaluated by conventional diagnostic methods.
- Chloride ions have also been detected using oligoindole-based foldamers, and it has been shown that due to the accumulation effect of oligoindoles, the binding affinity for chloride ions is greatest in the order octamers > hexamers > tetramers (Non-Patent Document 2).
- the present invention aims to provide a chloride ion sensor and a device using the same for detecting cancer cells and/or evaluating the malignancy of cancer cells.
- chloride ions can be detected with high sensitivity by modifying the surface of an electrode with a compound having a specific structure, and also discovered that such a highly sensitive chloride ion sensor can be used to detect cancer cells and/or evaluate the malignancy of cancer cells, leading to the invention.
- a chloride ion sensor which is an electrode modified by [2]
- a device for detecting cancer cells and/or evaluating the malignancy of cancer cells comprising one or more chloride ion sensors according to any one of [1] to [6] and a pressure-insertion force sensor, characterized in that the pressure-insertion force sensor includes an external force applying means and an external force measuring means, and measures the external force while applying an external force to a test cell in vitro, and the chloride ion sensor measures chloride ions discharged from the cell.
- the pressure-insulation force sensor is a cantilever.
- the cantilever has a cell pressure probe at its tip.
- a method for screening a chloride ion channel inhibitor using a device including one or more chloride ion sensors according to any one of [1] to [6] and a pressure force sensor comprising:
- the device is characterized in that the pressure-insertion force sensor includes an external force applying means and an external force measuring means, and measures the external force while applying an external force to a test cell in vitro, and the chloride ion sensor measures chloride ions discharged from the cell, administering a test substance to the cells;
- the method includes a step of measuring the external force applied to the cell while measuring the external force and measuring the chloride ions excreted from the cell; and a step of screening the test substance as a candidate substance for a chloride ion channel inhibitor using the level of the chloride ions as an index.
- the present invention provides a chloride ion sensor and a device using the same for detecting cancer cells and/or assessing the malignancy of cancer cells.
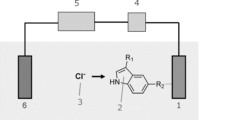
- FIG. 2 is a schematic diagram illustrating the chloride ion detection mechanism of the present invention.
- FIG. 13 shows the device output when an external force is applied to a cell-pressure cantilever using an atomic force microscope (AFM).
- AFM atomic force microscope
- FIG. 13 is a schematic diagram of an experimental system using a press-in force sensor.
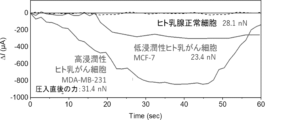
- FIG. 13 shows the measurement results of the press-in force sensor when human breast cancer cells are pressed in.
- This figure shows the measurement results of chloride ions expelled from three types of cells when an external force was applied to them.
- the time immediately after applying an external force to a single cell was set as 0 sec. 1 shows the results of measuring the amount of chloride ions expelled from cells when an external force was applied for 30 seconds to cells to which a chloride ion channel inhibitor had been added. * indicates p ⁇ 0.001.
- One embodiment of the present invention comprises: A chloride ion sensor having a chloride ion detection electrode, The chloride ion detection electrode has at least a part of the electrode surface containing the following compound: The electrode is a chloride ion sensor.
- the pyrrole ring site is a site that recognizes chloride ions
- R2 is a site that transmits the electron density change and dipole moment change accompanying ion capture to an electrode as an electron-conjugated anchor.
- the compound can selectively capture chloride ions due to the proton-donating ability of the pyrrole ring site. With the capture of chloride ions, an electron density change occurs in the pyrrole ring, and is transmitted to the electrode via the conjugated benzene ring and R2 . From the viewpoint of efficiently recognizing chloride ions and transmitting the electron density change and dipole moment change accompanying ion capture to the electrode, the compound can be suitably used in chloride ion detection.
- R1 is H, COOH, C ⁇ N, CHO, NO2 or halogens, preferably H. More preferably, it is an electron-withdrawing substituent that enhances the hydrogen bond donor property of the pyrrole ring moiety.
- R1 is a halogen, it may be any halogen element, for example, fluorine, chlorine, bromine, or iodine.
- R2 is an electron-conjugated anchor that transmits changes in electron density and dipole moment accompanying ion capture to the electrode, and therefore may have a structure having a molecular skeleton with ⁇ -electron conjugation.
- R2 is C ⁇ CH, SH, PO3H2 or COOH , preferably C ⁇ CH, before immobilization on the electrode.
- These functional groups have binding affinity with various electrodes, such as precious metal electrodes and oxide electrodes, as described below, and therefore the compound can be suitably immobilized on the electrode.
- R 2 is preferably C ⁇ C- for immobilizing a compound on a precious metal electrode.
- the R 2 portion of the compound does not need to be directly bonded to the electrode, and the R 2 portion may be bonded to the electrode via a group that serves as a linker, as long as it can transmit the electron density change and dipole moment change associated with chloride ion capture to the electrode.
- R2 when an amino group is added to R2 (e.g., when an amino group is added to C ⁇ C- to form C ⁇ C- NH2 ), a group that reacts with the amino group, such as a succinimide group, can be bonded to the electrode surface.
- the R2 portion is bonded to the electrode using the reactive residues of the amino group and succinimide group as a linker.
- R 2 is C ⁇ C-
- an embodiment can be mentioned in which a group that forms a 1,2,3-triazole ring by [3+2] dipolar cycloaddition reaction with C ⁇ C-, such as an azide group, is bonded to the electrode surface.
- the compound used in the present invention is not particularly limited as long as it can recognize chloride ions, but it is preferable that the compound has a high binding affinity to chloride ions.
- the binding constant Ka( Cl- ) with chloride ions may be 105M -1 or more, 106M -1 or more, or 107M -1 or more.
- the compound used in the present invention is preferably a compound represented by the following chemical formula, namely, 5-Ethynyl-1H-indole (hereinafter, also referred to as 5-EnIND).
- the compounds used in the present invention may be commercially available products or may be synthesized.
- commercially available products for example, 5-ethynyl-1H-indole (5-EnIND, Asta Tech, Inc., Product Code: 94615, CAS: 889108-48-9) can be used.
- the synthesis method is not particularly limited, and the compound can be synthesized by any method known to those skilled in the art (Non-Patent Documents 3-4).
- the material of the chloride ion detection electrode may be, for example, a precious metal such as gold, platinum, silver, or copper, or an oxide such as aluminum oxide (III) (Al 2 O 3 ), titanium oxide (IV) (TiO 2 ), or silicon dioxide (SiO 2 ).
- a precious metal such as gold, platinum, silver, or copper
- an oxide such as aluminum oxide (III) (Al 2 O 3 ), titanium oxide (IV) (TiO 2 ), or silicon dioxide (SiO 2 ).
- the electrode is preferably a precious metal electrode such as gold, platinum, silver, or copper
- the electrode is preferably an oxide electrode such as aluminum oxide (III) (Al 2 O 3 ), titanium oxide (IV) (TiO 2 ), or silicon dioxide (SiO 2 )
- the electrode is preferably an oxide electrode such as aluminum oxide (III) (Al 2 O 3 ), titanium oxide (IV) (TiO 2 ), or silicon dioxide (SiO 2 ).
- the manner in which the compound is immobilized on the electrode is not particularly limited, but it is preferable that the compound is arranged in the form of a monolayer on the electrode, and it is more preferable that the compound is arranged as densely as possible.
- intermolecular interactions between 5-EnIND molecules include ⁇ - ⁇ interactions between phenyl rings, hydrophobic interactions, and hydrogen bonds between NH sites of indole rings.
- solvents that do not easily cause solvation include dimethyl sulfoxide, which is an aprotic solvent.
- the electrode surface is modified with the above compound, but for example, 5% or more, 10% or more, 20% or more, 30% or more, 40% or more, 50% or more, 60% or more, 70% or more, 80% or more, 90% or more, or 100% of the electrode surface area may be modified with the above compound.
- the method for modifying the electrode surface with a compound can be appropriately selected depending on the type of electrode, the type of R2, etc., and can be carried out by methods known to those skilled in the art.
- Methods for modifying a noble metal electrode or an oxide electrode with a compound include, for example, a solution impregnation method, a vacuum deposition method, and a Langmuir-Blodgett method.
- a solution impregnation method e.g., a solution impregnation method, a vacuum deposition method, and a Langmuir-Blodgett method.
- 5-EnIND is modified on the surface of a gold electrode by the solution impregnation method, it can be carried out by the method shown below.
- the surface of other noble metal electrodes or oxide electrodes can also be modified with compounds by the same modification method.
- the surface of a noble metal electrode or oxide electrode can also be modified by the same modification method. After the gold electrode is exposed to oxygen plasma, the surface is washed with 2-propanol and then with distilled water.
- ⁇ 5-EnIND is immobilized as a covalently bonded monolayer via a gold-alkyne complexation reaction.
- 5-EnIND can be immobilized by immersing a gold thin film substrate in a 5-EnIND solution or dropping the solution onto the substrate and holding the substrate at a desired temperature for a desired time. It is preferable to immobilize the substrate in a light-shielded state.
- the temperature and time are not particularly limited as long as 5-EnIND can be immobilized, but the temperature can be, for example, 40°C to 80°C, 50°C to 70°C, or 55°C to 65°C, and the time can be, for example, 1 hour to 24 hours, 3 hours to 8 hours, or 5 hours to 7 hours.
- the electrode is allowed to stand for a desired period of time until it reaches room temperature. Wash with 2-propanol and then with distilled water to thoroughly wash away the 5-EnIND solution adhering to the surface, and dry with nitrogen gas.
- the chloride ion sensor of this embodiment may further include a reference electrode, which may be, for example, an unmodified gold electrode or a silver/silver chloride electrode, for example, an electrode having an internal solution of 3 M NaCl aqueous solution.
- a reference electrode which may be, for example, an unmodified gold electrode or a silver/silver chloride electrode, for example, an electrode having an internal solution of 3 M NaCl aqueous solution.
- the chloride ion sensor of this embodiment has a field effect transistor (FET)
- the FET may have a control gate terminal
- the reference electrode may be incorporated into the control gate terminal of the FET.
- the chloride ion sensor of this embodiment may further include a field effect transistor (FET).
- FET field effect transistor
- the chloride ion detection mechanism shown in FIG. 1 can be exemplified, and a potentiometric measurement system via a FET can be constructed by incorporating an electrode modified with the compound used in the present invention into the floating gate terminal of the FET.
- the pyrrole ring site in the compound used in the present invention acts as a receptor to capture the target chloride ion, causing a change in potential at the chloride ion detection electrode.
- the chloride ion in the solution can be detected by measuring the change in gate potential when the potential at the reference electrode is set as the reference potential.
- the FET to be used is not particularly limited as long as it can be used to measure chloride ions, and any FET can be used, for example, an n-type FET.
- the method of incorporating the chloride ion detection electrode into the gate terminal of the FET is not particularly limited, and can be performed by a method known to those skilled in the art.
- the potential difference generated between the chloride ion detection electrode and the reference electrode can be obtained as the output voltage or drain current of the FET source follower circuit, and the output voltage or drain current can be used as an index of the chloride ion level.
- an FET that exhibits an operation mode called a depression type i.e., an operation mode in which the drain current exhibits a linear variation range when the gate voltage is 0 V
- a depression type i.e., an operation mode in which the drain current exhibits a linear variation range when the gate voltage is 0 V
- the chloride ion sensor of this embodiment may include other components in addition to the above-mentioned FET, chloride ion detection electrode, and reference electrode, and may include other components such as a voltage measuring device, a power supply, an A/D conversion circuit, and a memory. These other components may be configured, for example, together with the FET as one device that constitutes a FET source follower circuit.
- Another embodiment of the present invention is a method for measuring chloride ions in a solution to be measured, comprising the step of contacting the above-mentioned chloride ion sensor with the solution to be measured.
- the solution to be measured may be any solution containing chloride ions.
- the solvent of the solution to be measured may be an aqueous electrolyte solution such as 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) containing any concentration of NaBr, or may be an isotonic solution. It is preferable that the solvent itself does not contain or substantially does not contain chloride ions, and for example, the chloride ion concentration may be 100 pM or less, 10 pM or less, 1 pM or less, 0.1 pM or less, or 0.01 pM or less.
- an isotonic solution is not particularly limited as long as it has an osmotic pressure approximately the same as that of body fluids, and the osmotic pressure may be, for example, 250 to 320 mOsm/L, 260 to 310 mOsm/L, 270 to 300 mOsm/L, or 280 to 290 mOsm/L.
- the solvent of the solution to be measured is a liquid that does not contain or substantially does not contain electrolytes, and an isotonic solution that does not contain or substantially does not contain chloride ions can be suitably used. Examples of such isotonic solutions include glucose solutions and sucrose solutions.
- the glucose concentration is not particularly limited as long as it allows chloride ions to be detected with high sensitivity, but it can be, for example, a 5.0 to 6.0% (w/v) glucose solution, preferably a 5.2 to 5.8% (w/v) glucose solution, and more preferably a 5.4 to 5.6% (w/v) glucose solution.
- the pH of the solution is not particularly limited and can be adjusted as desired. For example, it may be 7.0 to 8.0.
- Contact with the solution to be measured can be achieved, for example, by immersing the chloride ion detection electrode of the chloride ion sensor in the solution to be measured.
- the measurement method of this embodiment can measure chloride ions, for example, by measuring the potential difference generated between the chloride ion detection electrode and the reference electrode using the chloride ion sensor.
- the potential difference can be obtained as the output voltage or drain current of the FET source follower circuit, and the output voltage or drain current can be used as an index of the chloride ion level.
- the chloride ion level may, for example, represent the concentration or amount of chloride ions, and may indicate a higher chloride ion level by a greater decrease in the output voltage in the solution being measured compared to an output voltage in a control solution that is free or substantially free of chloride ions, or may indicate a higher chloride ion level by a greater decrease in the drain current in the solution being measured compared to the drain current in a control solution that is free or substantially free of chloride ions.
- a calibration curve between the output voltage or drain current and the chloride ion level may be prepared in advance, and the concentration or amount of chloride ions may be calculated using this calibration curve.
- Another embodiment of the present invention is a device for detecting cancer cells and/or evaluating the malignancy of cancer cells, comprising one or more of the above-mentioned chloride ion sensors and an insertion force sensor, characterized in that the insertion force sensor includes an external force applying means and an external force measuring means, and measures the external force while applying an external force to a test cell in vitro, and the chloride ion sensor measures chloride ions excreted from the cell.
- the external force application means may be any means that applies pressure to the cell membrane from outside the cell, but a mode in which the cell membrane is pressed by mechanical means is preferable. Also, it is preferable that the force applied to the cell membrane from outside the cell is a force that stretches the cell membrane in the lateral direction (parallel to the cell membrane). It may be a mode in which the cell membrane is pressed from the outside, or a mode in which the cell membrane is pulled.
- the external force is preferably applied using a member having a contact surface equivalent to about 1/10 of the surface area of the cell (e.g., a contact area of 30-300 ⁇ m 2 ).
- a member may be, for example, a member having a cell presser that contacts the cell at its tip to apply pressure, or a member having a flat tip.
- the shape of the cell presser is not limited as long as it has the above-mentioned contact surface, and may be pyramidal (conical or pyramidal with an apex angle that does not destroy the cell membrane even when a desired pressure is applied to the cell membrane), cylindrical, prismatic, truncated conical, truncated pyramidal, spherical (including approximately spherical), or hemispherical (including approximately hemispherical).
- the material of the cell presser is not particularly limited as long as it has the above-mentioned contact surface, and examples thereof include organic polymers and metals.
- the size of the cell presser is not limited as long as it has the above-mentioned contact surface.
- the diameter at the base may be 5 to 20 ⁇ m, and if it is spherical or hemispherical, the particle size may be 5 to 20 ⁇ m.
- Examples include cantilevers used in scanning probe microscopes (SPMs) and atomic force microscopes (AFMs), and tipless cantilevers with the above-mentioned cell indenter attached to their tips.
- the material of the cell presser is not particularly limited, but examples include silicon (such as single crystal silicon), silicon nitride, gold, chromium, indium arsenide, silica, polyethylene terephthalate, polyethylene, polypropylene, polystyrene, polycarbonate, and acrylic resin.
- the strength of the applied external force may be such that the excretion of chloride ions through the cell membrane is observed, and it is preferable that the strength is such that the cell membrane can be stretched laterally, for example, 1 to 100 nN, and preferably 5 to 50 nN.
- the duration of application of the external force may be any duration sufficient for observing the discharge of chloride ions due to the application of the external force, and may be, for example, 1 second to 15 minutes, or 10 seconds to 2 minutes.
- the external force measuring means may be any means capable of measuring the external force applied to the cell.
- SPM scanning probe microscope
- AFM atomic force microscope
- an external force can be applied to a cell using a cantilever used in the SPM or AFM, and the external force can be measured by the SPM or AFM.
- the cantilever may have a piezoelectric element for measuring the pressure force.
- SPM and AFM are not used as the means for measuring external force
- a cantilever-type device having a piezoelectric element for measuring the pressure force can be used.
- a device for moving the cantilever up and down e.g.
- Stampo Unit SU100, Olympus can also be used to press the cantilever into the cell and apply an external force.
- the advantage is that it is not necessary to use expensive equipment such as SPM and AFM, since the external force applied to the cell can be measured simply by using a device for moving the cantilever up and down.
- a piezoelectric element for measuring the pressure force may be provided at the base of the cantilever, and the external force applied to the cell can be measured by measuring the resistance change of the piezoelectric element when the cell is pressed in.
- the resistance change of the piezoelectric element may be measured by converting it into a voltage using, for example, a Wheatstone bridge circuit known to those skilled in the art.
- the measurement principle of the press-in force sensor is shown below.
- the cell is pressed into the cell with a cell presser attached to the tip of a cell press-in cantilever located in the center of the press-in force, and the strain generated at the base of the cantilever during this process is measured as a resistance change by a piezoelectric element for measuring the press-in force.
- the piezo-resistance element is also affected by temperature changes in the solution. Therefore, by placing a temperature compensating piezoelectric element near the piezo-element for measuring the press-in force and performing a differential measurement of these two elements, the effects of temperature changes can be eliminated.
- the electrode having the above-mentioned compound modified on its surface which is used to detect chloride ions as a chloride ion sensor, is placed near an external force application means such as a cantilever in the device, but may be on a structure different from the external force application means, or the external force application means such as a cantilever may also function as a chloride ion detection electrode, i.e., the tip of the cantilever may be an electrode having the above-mentioned compound modified on its surface.
- the external force application means functions as a chloride ion detection electrode, it is preferable that the part of the external force application means that comes into contact with the cells and the part modified with the above-mentioned compound are separated. This allows the chloride ion sensor to measure chloride ions without being affected by the negative charge on the cell surface.
- the distance between the chloride ion detection electrode and the external force application portion of the external force application means is not particularly limited as long as it is possible to detect chloride ions discharged by the cell, but is, for example, 100 ⁇ m or less, 90 ⁇ m or less, 80 ⁇ m or less, 70 ⁇ m or less, 60 ⁇ m or less, or 50 ⁇ m or less.
- the lower limit is not particularly limited, but is, for example, 20 ⁇ m or more, 30 ⁇ m or more, or 40 ⁇ m or more. The closer this distance is, the higher the detection sensitivity of chloride ions discharged by the test cell can be.
- Each of the above devices may be provided with one or more chloride ion sensors, and when more than one is provided, the number of chloride ion sensors may be, for example, two, three, four or more.
- chloride ions released by cells to which an external force is applied can be efficiently detected.
- the chloride ion detection electrodes in the chloride ion sensors are arranged so as to surround the area where an external force is applied to the cells.
- the chloride ion sensor detects chloride ions released from cells when cells are pressed using a chloride ion detection electrode placed near the cell presser.
- a chloride ion detection electrode placed near the cell presser.
- Another embodiment of the present invention is a method for detecting cancer cells and/or evaluating the malignancy of cancer cells, comprising the steps of applying an external force to test cells and measuring chloride ions by the above-mentioned chloride ion measurement method.
- the method of this embodiment makes it possible to detect cancer cells and evaluate their malignancy. Based on the evaluation results, doctors can determine the course of cancer treatment. For example, if a patient is diagnosed with cancer cells or the cancer cells are determined to be highly malignant, appropriate treatment such as cancer resection surgery can be performed. In this way, the method does not include medical procedures performed by doctors, etc., but can provide data to assist doctors in diagnosing cancer.
- the potential difference between the chloride ion detection electrode and the reference electrode caused by applying an external force to the test cell can be obtained as the output voltage or drain current of the FET source follower circuit, and the output voltage or drain current can be used as an index of the chloride ion level discharged by the test cell into the measurement target solution.
- the chloride ion level for example, represents the concentration or amount of chloride ions.
- a larger decrease in the output voltage in the measurement target solution compared to the output voltage in a control solution that does not contain or substantially does not contain chloride ions may indicate a higher chloride ion level
- a larger decrease in the drain current in the measurement target solution compared to the drain current in a control solution that does not contain or substantially does not contain chloride ions may indicate a higher chloride ion level.
- a calibration curve between the output voltage or drain current and the chloride ion level may be prepared in advance, and the concentration or amount of chloride ions may be calculated using this calibration curve.
- the cancer cells to be analyzed may be mammalian cancer cells, such as human, mouse, or rat cancer cells, but are preferably human cancer cells.
- the cancer cells may be established cultured cells or may be cancer cells isolated from a living body. Cancer tissue may also be used directly for analysis.
- There are no particular limitations on the type of cancer and examples include breast cancer, stomach cancer, lung cancer, pancreatic cancer, liver cancer, colon cancer, prostate cancer, and cervical cancer.
- the test cell when the level of chloride ions discharged by applying an external force to a test cell is higher than the level of chloride ions discharged by applying an external force to a control normal cell, the test cell can be evaluated as being a cancer cell.
- the change in drain current when a drain voltage of 5 V and a gate voltage of 0 V are input as the control voltage for the FET is used as an indicator of chloride ion levels
- the drain current in a control solution that does not contain or substantially does not contain chloride ions e.g., a solution containing normal cells to which an external force has been applied
- the decrease in drain current in a measurement solution containing test cells to which an external force has been applied is, for example, 50 ⁇ A or more, 100 ⁇ A or more, 150 ⁇ A or more, or 200 ⁇ A or more
- the test cells can be evaluated as being cancer cells.
- the test cells can be evaluated as being cancer cells by combining not only the chloride ion level but also other indicators.
- the test cell when the chloride ion level discharged by applying an external force to a test cell in the above-mentioned device is higher than the chloride ion level discharged by applying an external force to a cancer cell with low invasiveness, the test cell can be evaluated as having a high malignancy.
- the malignancy of a cancer is an index that comprehensively indicates invasiveness, metastatic ability, etc., and when invasiveness or metastatic ability is high, the malignancy of the cancer can be evaluated as high.
- the test cells can be evaluated as having high malignancy.
- the test cells can be evaluated as being highly malignant by combining not only the chloride i
- each step of this embodiment may be taken from the above sections ⁇ Chloride ion sensor>, ⁇ Device for detecting cancer cells and/or assessing the malignancy of cancer cells>, or ⁇ Method for measuring chloride ions>.
- Another embodiment of the present invention is a method for screening chloride ion channel inhibitors using a device comprising one or more of the above chloride ion sensors and a pressure force sensor, comprising:
- the device is characterized in that the pressure-insertion force sensor includes an external force applying means and an external force measuring means, applies an external force to a test cell in vitro while measuring the external force, and the chloride ion sensor measures chloride ions discharged from the cell, administering a test substance to the cells;
- the method includes the steps of: applying an external force to the cell while measuring the external force and measuring chloride ions excreted from the cell; and screening the test substance as a candidate for a chloride ion channel inhibitor using the level of the chloride ions as an index.
- the screening step may be a step of screening the test substance as a candidate substance for a chloride ion channel inhibitor in any one of the following cases (1) to (3): (1) the level of chloride ions excreted by cells administered with the test substance due to the load of an external force is lower than the level of chloride ions measured in the same manner using cells not administered with the test substance; (2) The level of chloride ions excreted by the cells administered with the test substance due to the load of an external force is equivalent to or lower than the level of chloride ions measured in the same manner using low-invasive cancer cells or normal cells; (3) The level of chloride ions excreted by the cells administered with the test substance due to the application of an external force is lower than a predetermined cutoff value.
- the screening method is not particularly limited, but may optionally include additional steps. For example, it may include a step of conditioning the cells in advance, or a step of identifying the screened candidate substance as a chloride ion channel inhibitor.
- test substance is not particularly limited, but examples include compounds, antibodies, antigen-binding fragments, peptides, nucleic acids, etc.
- the amount of test substance administered to the cells and the reaction time of the test substance with the cells are not particularly limited and can be adjusted as appropriate depending on the type of test substance.
- the cells are not particularly limited, but are preferably cancer cells, and more preferably highly invasive cancer cells.
- highly invasive human breast cancer cells may be used as the cells, and in this case, the less invasive cancer cells and normal cells in (2) above may be less invasive human breast cancer cells and human mammary epithelial cells, respectively.
- the chloride ion channel expressed in the cells used in this embodiment is not particularly limited as long as the cells can be used in the screening method of this embodiment, and examples thereof include ClC chloride ion channels (ClC-2, ClC-3, ClC-4, ClC-6, ClC-7, etc.), calcium-dependent chloride ion channels (TMEM16A, TMEM16B, Best1, Best2, etc.), and volume-sensitive outwardly rectifying anion channels (CLIC1, LRRC8A, etc.).
- the screening method of this embodiment measures chloride ions discharged by applying an external force to the cells
- the chloride ion channel expressed in the cells is a channel that discharges chloride ions by applying an external force to the cells
- volume-sensitive outwardly rectifying anion channels such as CLIC1 and LRRC8A, which are channels that detect cell swelling, and CLIC1 is particularly preferable.
- Example 1 Preparation of an ion sensor
- the surface of the detection electrode was modified with 5-ethynyl-1H-indole (5-EnIND, Asta Tech, Inc., Product Code: 94615, CAS: 889108-48-9).
- 5-EnIND was dissolved in dimethyl sulfoxide (DMSO) to prepare a 10 mM 5-EnIND solution.
- DMSO dimethyl sulfoxide
- the gold thin film surface to be the detection electrode was irradiated with oxygen plasma, washed with 2-propanol, washed with distilled water, and dried with nitrogen gas.
- the gold thin film substrate was immersed in the 5-EnIND solution or the solution was dropped onto the substrate, and the substrate was kept on a hot plate at 60°C in a light-shielded state for 6 hours. This resulted in the immobilization of 5-EnIND as a monolayer by covalent bonding via the gold-alkyne complexation reaction. After leaving the substrate at 25°C for 12 hours, the substrate was washed with 2-propanol and then with distilled water to thoroughly wash the 5-EnIND solution attached to the surface, and then dried with nitrogen gas.
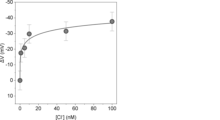
- Example 2 Incorporation of detection electrodes into FET and titration experiment A gold electrode modified with 5-EnIND (hereafter, 5-EnIND-modified electrode) was incorporated into the floating gate terminal of an FET to construct a potentiometric measurement system via an FET ( Figure 1). To examine the chloride ion recognition ability of the 5-EnIND-modified electrode by titration experiments, the electrode was incorporated into the floating gate terminal of an FET source follower circuit (BCT-2, BioCMOS).
- BCT-2 FET source follower circuit
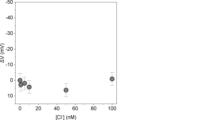
- 4-ethynyl-1H-indole (4-EnIND) is a structural isomer of 5-EnIND.
- R2 at the 4th or 5th position of EnIND is preferred for bonding to an electrode.
- an electrode was prepared using 4-EnIND in the same manner as for 5-EnIND.
- the potential response of the FET source follower circuit to changes in chloride ion concentration was confirmed using the prepared electrode, and when 4-EnIND was used, almost no potential change was observed (Figure 3). Although both have the same pyrrole ring moiety (NH moiety) as 5-EnIND as a hydrogen bond donor responsible for ion capture, the reason for this difference in potential response is thought to be due to the difference in the formation state of each monolayer.
- 5-EnIND which has an alkyne moiety that bonds to a gold electrode added to the 5th position, is thought to have a steric hindrance advantage in forming a monolayer compared to 4-EnIND added to the 4th position. For this reason, it is assumed that the density of EnIND in the monolayer is higher when 5-EnIND is applied compared to when 4-EnIND is applied.
- the factors that contribute to the recognition of chloride ions in aqueous solution include the accumulation effect of EnIND and the formation of a hydrophobic field, so it is presumed that the binding affinity for chloride ions is reduced on the 4-EnIND-modified electrode.
- the reasons for the enhanced response at lower concentrations are thought to be, for example, the absence of contaminating anions that inhibit chloride ion recognition, and the small effect of the Debye shielding effect, which resulted in a stronger change in electron density accompanying ion capture by the 5-EnIND monolayer, which induced a change in the electrode potential.
- Example 3 Structure of a MEMS cantilever type device
- MEMS Micro Electro Mechanical Systems
- This device is a device that combines a pressure-insertion force sensor and a chloride ion (Cl - ) sensor.
- the cantilever-type device used in Example 3 is a device that has two chloride ion sensors in the vicinity of the pressure-insertion force sensor, and specifically, the distance between the chloride ion detection electrode and the center of the cell indenter is set to 60 ⁇ m.
- the chloride ion detection electrode was formed by selectively dropping 5-EnIND solution onto the electrode material using an inkjet device and holding it in a hot chamber at 60°C for 6 hours.
- the cantilever was then washed with 2-propanol and then with distilled water to thoroughly remove unbound 5-EnIND, and used for chloride ion detection.
- An untreated gold electrode was placed near the base of the MEMS, and used as a control gate terminal to set the reference potential for chloride ion measurement.
- the chloride ion detection electrode of the MEMS was connected to the gate terminal of a commercially available n-type FET (Toshiba Semiconductor 2SK241Y) via a flexible wiring board.
- the source and drain terminals of the FET and the control gate terminal of the MEMS were connected to a source meter (Keithley Instruments 2636B), respectively, to control input and output.
- Example 4 Calibration of a MEMS cantilever-type device using an atomic force microscope
- the MEMS cantilever device was fixed on a dish and immersed in a 5.5% glucose solution, and an external force was applied using an AFM.
- the cantilever was lowered using a pyramidal tip until the force applied to the cantilever reached 5, 10, 30, or 50 nN.
- the response of the press-in force sensor of the cantilever device when this external force was applied was measured, and a calibration curve was created (Figure 6). Based on this calibration curve, the press-in force was actually measured when cells were pressed into the device.
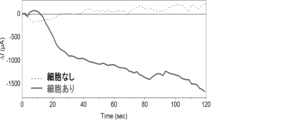
- Example 5 Measurement of ions excreted from cells when hypotonic stimulation is applied
- Highly invasive human breast cancer cells MDA-MB-231 ATCC, Product Code: HTB-26
- the cell medium was replaced with a 5.5% glucose solution, and the FET sensor was brought close to the cells on the dish.
- the solution was then replaced with a 160 mOsm hypotonic phosphate buffer solution to induce chloride ion excretion.
- the FET sensor was used for measurement, the sensor response was confirmed approximately 15 seconds after the phosphate buffer solution was replaced (Figure 7).
- Figure 8A shows a schematic diagram of an experimental system using a pressure force sensor
- Figure 8B shows the results of measuring the pressure force when human breast cancer cells were pressed into the cell.
- the pressure force on the vertical axis is a value calculated based on the calibration curve in Figure 6.
- the pressure force was detected by contacting the cell with the cell presser.
- the cantilever was held in a state where the cell presser was in contact with the cell, and the cell was pressed into the cell for a certain period of time. It was confirmed that the pressure force returned to its initial value by removing the load thereafter. The maximum value immediately after pressing was taken as the pressure force.
- Example 7 Measurement of ions excreted from cells when external force is applied
- Highly invasive human breast cancer cells MDA-MB-231, low-invasive human breast cancer cells MCF-7 (JCRB Cell Bank, Product Code: JCRB0134), and human mammary epithelial cells MCF10A (ATCC, Product Code: CRL-10317) were seeded at 5 ⁇ 104 cells each and cultured overnight in a CO2 incubator.
- the cell medium was replaced with 5.5% glucose solution, and the FET sensor fused cantilever type device was brought close to a single cell on the dish and the cantilever was lowered to press the cell.
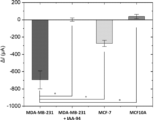
- Example 8 Evaluation of ion efflux ability of cells to which a chloride ion channel inhibitor has been added
- Highly invasive human breast cancer cells MDA-MB-231, low-invasive human breast cancer cells MCF-7, and human mammary epithelial cells MCF10A were seeded at 5 ⁇ 104 cells each and cultured overnight in a CO2 incubator.
- chloride ion channel inhibitor IAA-94 final concentration 100 ⁇ M; Sigma Aldrich, Product Code: I117, CAS: 54197-31-8) was added to the medium and reacted for 1 hour in a CO2 incubator.
- the cell medium was replaced with 5.5% glucose solution, and the FET sensor fused cantilever type device was brought close to a single cell on the dish and the cantilever was lowered to press the cell.
- the drain current was measured 30 seconds after pressing, and the FET sensor responded to untreated MDA-MB-231 cells, and chloride ions were excreted, whereas no sensor response was observed in IAA-94-added cells ( Figure 10).
- this device can be used to evaluate the effects of chloride ion channel inhibitors that receive mechanical stimuli. This device may be useful for screening chloride ion channel inhibitors that contribute to the invasion of cancer cells.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Analytical Chemistry (AREA)
- Physics & Mathematics (AREA)
- General Health & Medical Sciences (AREA)
- Biochemistry (AREA)
- Immunology (AREA)
- Molecular Biology (AREA)
- Organic Chemistry (AREA)
- General Physics & Mathematics (AREA)
- Pathology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Biomedical Technology (AREA)
- Biotechnology (AREA)
- Medicinal Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- Biophysics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Microbiology (AREA)
- Hematology (AREA)
- Urology & Nephrology (AREA)
- Food Science & Technology (AREA)
- General Engineering & Computer Science (AREA)
- Genetics & Genomics (AREA)
- Toxicology (AREA)
- Sustainable Development (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
Abstract
Il a été découvert qu'il était désormais possible de détecter des ions chlorure avec une sensibilité élevée en modifiant au moins une partie de la surface d'une électrode avec un composé représenté par la formule chimique (dans laquelle R1 représente H, COOH, C≡N, CHO, NO2 ou un atome d'halogène ; et R2 représente C≡C-, S-, P(=O)(O-)2 ou COO-). Sur la base de cette découverte, il est possible de fournir : un capteur d'ions chlorure comprenant l'électrode ; et un dispositif équipé du capteur d'ions chlorure et utilisé pour la détection de cellules cancéreuses et/ou l'évaluation du degré de malignité de cellules cancéreuses.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2025526079A JPWO2024253016A1 (fr) | 2023-06-06 | 2024-05-30 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2023-093435 | 2023-06-06 | ||
| JP2023093435 | 2023-06-06 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024253016A1 true WO2024253016A1 (fr) | 2024-12-12 |
Family
ID=93795991
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2024/019859 Ceased WO2024253016A1 (fr) | 2023-06-06 | 2024-05-30 | Capteur d'ions chlorure et dispositif de détection de cellules cancéreuses et/ou d'évaluation du degré de malignité de cellules cancéreuses l'utilisant |
Country Status (2)
| Country | Link |
|---|---|
| JP (1) | JPWO2024253016A1 (fr) |
| WO (1) | WO2024253016A1 (fr) |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH041239A (ja) * | 1990-04-18 | 1992-01-06 | Tokuyama Soda Co Ltd | イオン感応膜の製造方法 |
| JPH04124162A (ja) * | 1990-09-14 | 1992-04-24 | Joko:Kk | 塩化物イオン選択性電極および電極用感応物質 |
| JPH05142191A (ja) * | 1991-11-18 | 1993-06-08 | Hitachi Ltd | イオン選択性電極 |
| JPH1010078A (ja) * | 1996-06-24 | 1998-01-16 | Hitachi Ltd | 塩素イオンセンサ |
| JPH10318973A (ja) * | 1997-05-15 | 1998-12-04 | Hitachi Ltd | 塩素イオン感応膜及びその製造方法 |
| JPH11337515A (ja) * | 1998-05-29 | 1999-12-10 | Hitachi Ltd | 陰イオン選択性電極 |
| JP2017106823A (ja) * | 2015-12-10 | 2017-06-15 | 東亜ディーケーケー株式会社 | 塩素イオン選択性電極 |
| JP2021191238A (ja) * | 2020-06-05 | 2021-12-16 | 国立研究開発法人産業技術総合研究所 | がん細胞の評価方法および評価デバイス |
-
2024
- 2024-05-30 WO PCT/JP2024/019859 patent/WO2024253016A1/fr not_active Ceased
- 2024-05-30 JP JP2025526079A patent/JPWO2024253016A1/ja active Pending
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH041239A (ja) * | 1990-04-18 | 1992-01-06 | Tokuyama Soda Co Ltd | イオン感応膜の製造方法 |
| JPH04124162A (ja) * | 1990-09-14 | 1992-04-24 | Joko:Kk | 塩化物イオン選択性電極および電極用感応物質 |
| JPH05142191A (ja) * | 1991-11-18 | 1993-06-08 | Hitachi Ltd | イオン選択性電極 |
| JPH1010078A (ja) * | 1996-06-24 | 1998-01-16 | Hitachi Ltd | 塩素イオンセンサ |
| JPH10318973A (ja) * | 1997-05-15 | 1998-12-04 | Hitachi Ltd | 塩素イオン感応膜及びその製造方法 |
| JPH11337515A (ja) * | 1998-05-29 | 1999-12-10 | Hitachi Ltd | 陰イオン選択性電極 |
| JP2017106823A (ja) * | 2015-12-10 | 2017-06-15 | 東亜ディーケーケー株式会社 | 塩素イオン選択性電極 |
| JP2021191238A (ja) * | 2020-06-05 | 2021-12-16 | 国立研究開発法人産業技術総合研究所 | がん細胞の評価方法および評価デバイス |
Non-Patent Citations (1)
| Title |
|---|
| YAMAGISHI AYANA, ITO FUMIE, NAKAMURA CHIKASHI: "Study on Cancer Cell Invasiveness via Application of Mechanical Force to Induce Chloride Ion Efflux", ANALYTICAL CHEMISTRY, AMERICAN CHEMICAL SOCIETY, vol. 93, no. 26, 6 July 2021 (2021-07-06), pages 9032 - 9035, XP093247276, ISSN: 0003-2700, DOI: 10.1021/acs.analchem.1c01589 * |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2024253016A1 (fr) | 2024-12-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US9018019B2 (en) | Engineered conductive polymer films to mediate biochemical interactions | |
| Ahmad et al. | An electrochemical sandwich immunosensor for the detection of HER2 using antibody-conjugated PbS quantum dot as a label | |
| Zagitova et al. | Voltammetric chiral recognition of naproxen enantiomers by N-tosylproline functionalized chitosan and reduced graphene oxide based sensor | |
| KR102657461B1 (ko) | 단일-세포 세포내 나노-ph 프로브 | |
| Lee et al. | Peptide-imprinted poly (hydroxymethyl 3, 4-ethylenedioxythiophene) nanotubes for detection of α synuclein in human brain organoids | |
| Hu et al. | The inhibition of fluorescence resonance energy transfer between quantum dots for glucose assay | |
| JPS59225343A (ja) | 生化学リガンドを有する診断装置 | |
| US20140231274A1 (en) | Single molecule detection method and single molecule detection apparatus for biological molecule, and disease marker testing apparatus | |
| KR20050100367A (ko) | 소용적 샘플 내의 분자 분석용 방법 및 장치 | |
| Chocholova et al. | Advanced antifouling zwitterionic layer based impedimetric HER2 biosensing in human serum: Glycoprofiling as a novel approach for breast cancer diagnostics | |
| Yadav et al. | Fabrication of alkoxysilane substituted polymer-modified disposable biosensing platform: Towards sperm protein 17 sensing as a new cancer biomarker | |
| BR112020014894A2 (pt) | Eletrodo para uso na detecção eletroquímica de uma espécie alvo, espectrômetro eletroquímico, uso de um eletrodo e método eletroquímico de detecção de uma espécie alvo | |
| Nishitani et al. | Polymeric nanofilter biointerface for potentiometric small-biomolecule recognition | |
| Saha et al. | Selective pathological and intracellular detection of human serum albumin by photophysical and electrochemical techniques using a FRET-based molecular probe | |
| Lu et al. | Ultrasensitive electrochemical detection of CYFRA 21-1 via in-situ initiated ROP signal amplification strategy | |
| ES2769288T3 (es) | Un electrodo y uso del mismo | |
| Ahmadi et al. | Affinity-based electrochemical biosensor with antifouling properties for detection of lysophosphatidic acid, a promising early-stage ovarian cancer biomarker | |
| Altunkök et al. | Development of potentıometrıc bıosensor for dıagnosıs of prostate cancer | |
| Adu et al. | Rapid and label-free A2 peptide epitope decorated CoFe2O4-C60 nanocomposite-based electrochemical immunosensor for detecting visceral leishmaniasis | |
| Prats‐Alfonso et al. | Cancer Prognostics by Direct Detection of p53‐Antibodies on Gold Surfaces by Impedance Measurements | |
| Wang et al. | A pendulum-type electrochemical aptamer-based sensor for continuous, real-time and stable detection of proteins | |
| Nhu et al. | An evaluation of a gold surface functionalization procedure for antibody binding and protein detection using 11-mercaptoundecanoic acid (11-MUA) | |
| WO2024253016A1 (fr) | Capteur d'ions chlorure et dispositif de détection de cellules cancéreuses et/ou d'évaluation du degré de malignité de cellules cancéreuses l'utilisant | |
| US20150219579A1 (en) | Method for detecting autoantibodies | |
| Yaman et al. | Fabrication of trastuzumab conjugated curcumin nanoparticles based impedimetric cytosensor for the cancer cell detection |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24819247 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2025526079 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2025526079 Country of ref document: JP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |