EP2103706A1 - Legierung für Beschichtungen durch Pulverspritzen - Google Patents
Legierung für Beschichtungen durch Pulverspritzen Download PDFInfo
- Publication number
- EP2103706A1 EP2103706A1 EP08447018A EP08447018A EP2103706A1 EP 2103706 A1 EP2103706 A1 EP 2103706A1 EP 08447018 A EP08447018 A EP 08447018A EP 08447018 A EP08447018 A EP 08447018A EP 2103706 A1 EP2103706 A1 EP 2103706A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- powder
- substrate
- process according
- coating
- deposition
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- 239000000843 powder Substances 0.000 title claims abstract description 58
- 229910045601 alloy Inorganic materials 0.000 title claims abstract description 11
- 239000000956 alloy Substances 0.000 title claims abstract description 11
- 238000000576 coating method Methods 0.000 claims abstract description 45
- 239000011248 coating agent Substances 0.000 claims abstract description 39
- 239000000758 substrate Substances 0.000 claims abstract description 38
- 229910052751 metal Inorganic materials 0.000 claims abstract description 20
- 239000002184 metal Substances 0.000 claims abstract description 20
- 229910052782 aluminium Inorganic materials 0.000 claims abstract description 19
- 230000008021 deposition Effects 0.000 claims abstract description 18
- 239000011701 zinc Substances 0.000 claims abstract description 18
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 claims abstract description 16
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 claims abstract description 15
- 229910052725 zinc Inorganic materials 0.000 claims abstract description 13
- 239000010410 layer Substances 0.000 claims abstract description 8
- 229910001297 Zn alloy Inorganic materials 0.000 claims abstract description 6
- 229910052729 chemical element Inorganic materials 0.000 claims abstract description 6
- 239000011247 coating layer Substances 0.000 claims abstract description 6
- 239000007788 liquid Substances 0.000 claims abstract description 6
- 239000000203 mixture Substances 0.000 claims abstract description 5
- 239000007787 solid Substances 0.000 claims abstract description 5
- 239000000126 substance Substances 0.000 claims abstract description 5
- 238000010438 heat treatment Methods 0.000 claims abstract description 4
- 239000012159 carrier gas Substances 0.000 claims abstract description 3
- 238000004320 controlled atmosphere Methods 0.000 claims abstract description 3
- 230000003647 oxidation Effects 0.000 claims abstract description 3
- 238000007254 oxidation reaction Methods 0.000 claims abstract description 3
- 230000004927 fusion Effects 0.000 claims abstract 3
- 238000005551 mechanical alloying Methods 0.000 claims abstract 3
- 238000000034 method Methods 0.000 claims description 26
- 229910052749 magnesium Inorganic materials 0.000 claims description 20
- 239000011777 magnesium Substances 0.000 claims description 20
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 claims description 15
- 230000008018 melting Effects 0.000 claims description 7
- 238000002844 melting Methods 0.000 claims description 7
- 229910000831 Steel Inorganic materials 0.000 claims description 5
- 239000010959 steel Substances 0.000 claims description 5
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 claims description 4
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 claims description 4
- 229910052802 copper Inorganic materials 0.000 claims description 4
- 239000010949 copper Substances 0.000 claims description 4
- 239000002245 particle Substances 0.000 claims description 4
- 229910052710 silicon Inorganic materials 0.000 claims description 4
- 239000010703 silicon Substances 0.000 claims description 3
- 229910000838 Al alloy Inorganic materials 0.000 claims description 2
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 claims description 2
- 229910001369 Brass Inorganic materials 0.000 claims description 2
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 claims description 2
- 229910052684 Cerium Inorganic materials 0.000 claims description 2
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 claims description 2
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 claims description 2
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 claims description 2
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 claims description 2
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 claims description 2
- QCWXUUIWCKQGHC-UHFFFAOYSA-N Zirconium Chemical compound [Zr] QCWXUUIWCKQGHC-UHFFFAOYSA-N 0.000 claims description 2
- 238000005275 alloying Methods 0.000 claims description 2
- 229910052797 bismuth Inorganic materials 0.000 claims description 2
- JCXGWMGPZLAOME-UHFFFAOYSA-N bismuth atom Chemical compound [Bi] JCXGWMGPZLAOME-UHFFFAOYSA-N 0.000 claims description 2
- 229910052796 boron Inorganic materials 0.000 claims description 2
- 239000010951 brass Substances 0.000 claims description 2
- 229910052791 calcium Inorganic materials 0.000 claims description 2
- 239000011575 calcium Substances 0.000 claims description 2
- GWXLDORMOJMVQZ-UHFFFAOYSA-N cerium Chemical compound [Ce] GWXLDORMOJMVQZ-UHFFFAOYSA-N 0.000 claims description 2
- 229910052804 chromium Inorganic materials 0.000 claims description 2
- 239000011651 chromium Substances 0.000 claims description 2
- 238000007598 dipping method Methods 0.000 claims description 2
- 229910052746 lanthanum Inorganic materials 0.000 claims description 2
- FZLIPJUXYLNCLC-UHFFFAOYSA-N lanthanum atom Chemical compound [La] FZLIPJUXYLNCLC-UHFFFAOYSA-N 0.000 claims description 2
- 229910052744 lithium Inorganic materials 0.000 claims description 2
- 230000001050 lubricating effect Effects 0.000 claims description 2
- WPBNNNQJVZRUHP-UHFFFAOYSA-L manganese(2+);methyl n-[[2-(methoxycarbonylcarbamothioylamino)phenyl]carbamothioyl]carbamate;n-[2-(sulfidocarbothioylamino)ethyl]carbamodithioate Chemical compound [Mn+2].[S-]C(=S)NCCNC([S-])=S.COC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OC WPBNNNQJVZRUHP-UHFFFAOYSA-L 0.000 claims description 2
- 229910052750 molybdenum Inorganic materials 0.000 claims description 2
- 239000011733 molybdenum Substances 0.000 claims description 2
- 229910052759 nickel Inorganic materials 0.000 claims description 2
- 229910052712 strontium Inorganic materials 0.000 claims description 2
- CIOAGBVUUVVLOB-UHFFFAOYSA-N strontium atom Chemical compound [Sr] CIOAGBVUUVVLOB-UHFFFAOYSA-N 0.000 claims description 2
- 239000002344 surface layer Substances 0.000 claims description 2
- 229910052715 tantalum Inorganic materials 0.000 claims description 2
- GUVRBAGPIYLISA-UHFFFAOYSA-N tantalum atom Chemical compound [Ta] GUVRBAGPIYLISA-UHFFFAOYSA-N 0.000 claims description 2
- 229910052718 tin Inorganic materials 0.000 claims description 2
- 239000011135 tin Substances 0.000 claims description 2
- 229910052719 titanium Inorganic materials 0.000 claims description 2
- 239000010936 titanium Substances 0.000 claims description 2
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 claims description 2
- 229910052721 tungsten Inorganic materials 0.000 claims description 2
- 239000010937 tungsten Substances 0.000 claims description 2
- 229910052726 zirconium Inorganic materials 0.000 claims description 2
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 claims 1
- 238000007493 shaping process Methods 0.000 claims 1
- 229910052720 vanadium Inorganic materials 0.000 claims 1
- LEONUFNNVUYDNQ-UHFFFAOYSA-N vanadium atom Chemical compound [V] LEONUFNNVUYDNQ-UHFFFAOYSA-N 0.000 claims 1
- 238000000151 deposition Methods 0.000 abstract description 11
- 238000005491 wire drawing Methods 0.000 abstract description 2
- 238000000889 atomisation Methods 0.000 abstract 2
- 238000001033 granulometry Methods 0.000 abstract 2
- 238000000133 mechanosynthesis reaction Methods 0.000 abstract 2
- 239000000155 melt Substances 0.000 abstract 2
- 239000000314 lubricant Substances 0.000 abstract 1
- 238000002791 soaking Methods 0.000 abstract 1
- 238000007669 thermal treatment Methods 0.000 abstract 1
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 6
- 238000005260 corrosion Methods 0.000 description 4
- 230000007797 corrosion Effects 0.000 description 4
- 239000000047 product Substances 0.000 description 4
- 229910052742 iron Inorganic materials 0.000 description 3
- 238000009434 installation Methods 0.000 description 2
- 238000005240 physical vapour deposition Methods 0.000 description 2
- 229910018134 Al-Mg Inorganic materials 0.000 description 1
- 229910018467 Al—Mg Inorganic materials 0.000 description 1
- 238000005299 abrasion Methods 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 238000005336 cracking Methods 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 239000012467 final product Substances 0.000 description 1
- 238000007654 immersion Methods 0.000 description 1
- 229910001338 liquidmetal Inorganic materials 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C2/00—Hot-dipping or immersion processes for applying the coating material in the molten state without affecting the shape; Apparatus therefor
- C23C2/26—After-treatment
- C23C2/261—After-treatment in a gas atmosphere, e.g. inert or reducing atmosphere
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C2/00—Hot-dipping or immersion processes for applying the coating material in the molten state without affecting the shape; Apparatus therefor
- C23C2/26—After-treatment
- C23C2/265—After-treatment by applying solid particles to the molten coating
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C24/00—Coating starting from inorganic powder
- C23C24/08—Coating starting from inorganic powder by application of heat or pressure and heat
- C23C24/10—Coating starting from inorganic powder by application of heat or pressure and heat with intermediate formation of a liquid phase in the layer
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C24/00—Coating starting from inorganic powder
- C23C24/08—Coating starting from inorganic powder by application of heat or pressure and heat
- C23C24/10—Coating starting from inorganic powder by application of heat or pressure and heat with intermediate formation of a liquid phase in the layer
- C23C24/103—Coating with metallic material, i.e. metals or metal alloys, optionally comprising hard particles, e.g. oxides, carbides or nitrides
- C23C24/106—Coating with metal alloys or metal elements only
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C26/00—Coating not provided for in groups C23C2/00 - C23C24/00
- C23C26/02—Coating not provided for in groups C23C2/00 - C23C24/00 applying molten material to the substrate
Definitions
- the object of the present invention is to obtain a metal substrate having a zinc-based coating whose corrosion resistance is improved, more particularly by adding magnesium in this coating.
- Magnesium is indeed well known to greatly improve corrosion resistance.
- Today alloys Zn-Al-Mg type are deposited by PVD ( physical vapor deposition ) or immersion in a bath.
- Various coatings have thus been patented and are especially known under the trade names Superdyma ® (Zn, 10% Al, 3% Mg, 0.2% Si), ZAM for ZincAluminium-Magnesium (Zn, 6% Al, 3% Mg) or Magizinc TM (Zn, 1% Al, 1% Mg), etc.
- These alloys containing magnesium are however fragile and crack during deformation of the substrate.
- the object of the invention is to provide a metal substrate coating that makes it possible to overcome the drawbacks of the state of the art.
- the object of the invention is to incorporate, in the alloy constituting this coating, magnesium in order essentially to improve the resistance to corrosion while preserving the resistance to cracking and facilitating the deposition means. More generally, the invention aims to flexibly modify the chemical composition of the coating layer.
- a first object of the present invention relates to a method of coating a metal substrate by deposition of a metal powder composed of a plurality of different chemical elements, characterized in that , at at the time of deposition, said substrate is brought to a temperature higher than the lowest liquidus temperature of said chemical elements and their alloys, so that said powder can melt at least partially during contact with the substrate.
- the melting temperature defined for the pure substances is in fact replaced by the temperature of liquidus, which is the temperature at which the first crystals appear in the liquid.
- a second object of the present invention relates to a metallurgical product or long coated coated obtained by carrying out the method described above, in the case where the powder deposition is performed on a "solid surface Comprising a coating with a thickness of between 1 and 80 ⁇ m.
- a third object of the present invention relates to a coated metallurgical product or long flat obtained by implementing the method described above, in the case where the deposition of powder is carried out on "liquid surface ", Comprising a coating thickness between 5 and 80 ⁇ m for a long product and between 1 and 40 ⁇ m for a flat product.
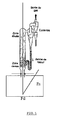
- the figure 1 schematically represents a powder deposition installation per fluidized bed on a wire at the outlet of a zinc bath, according to the present invention.
- the figure 2 represents a micrographic section obtained on a steel wire before (bottom view) and after (top view) the projection of a zinc powder rich in magnesium and aluminum according to the present invention.
- the reference sign 1 represents the steel, the sign 2 the intermetallic layer and the sign 3 the coating.
- the present invention consists in producing a metal powder deposit, containing magnesium, on a metal substrate, coated or not.
- the powder used according to the invention is typically composed of a zinc alloy containing aluminum (0.1 to 15%, by weight) with a large amount of magnesium (0.1 to 50%, typically 2 to 8%). %, in weight). Silicon can also be added to limit the reactivity of the powder with the substrate (max 0.2%, by weight).
- the following alloying elements may also be included: lithium, bismuth, tin, titanium, copper, nickel, chromium, manganese, molybdenum, tungsten, tantalum, zirconium, lanthanum, cerium, strontium, boron, calcium, vanandium, etc.
- this powder is sprayed with a carrier gas at a speed preferably between 1 and 250 m / s, is electrostatically charged or not or is deposited, or by placing (type line "Batch"), or by scrolling ("continuous" type line) the substrate in a fluidized bed.
- a carrier gas at a speed preferably between 1 and 250 m / s, is electrostatically charged or not or is deposited, or by placing (type line "Batch"), or by scrolling ("continuous" type line) the substrate in a fluidized bed.
- FIG. figure 1 An example of a fluidized bed powder deposition installation according to the present invention is shown in FIG. figure 1 .
- the metal substrate intended to receive the powder according to the invention can be in any usual form such as for example a wire, a strip or a profiled piece such as a beam or sheet pile.
- This metal substrate which may be steel, copper, brass, aluminum, etc., may be pre-coated or not with zinc, aluminum or a zinc / aluminum alloy, for example.
- This pre-coating may be liquid or solid depending on the final type of coating targeted.
- the powder projection can produce a second very thin coating (typically ⁇ 10 .mu.m) but very rich in magnesium on a solid coating.
- the advantage of this process is to deposit a magnesium-rich layer, known for its fragility, on a ductile coating based on zinc or zinc / aluminum.
- the final coating can thus have excellent deformation and stamping capabilities, because the microcracks obtained on the final coating will be invisible to the naked eye and therefore not impeding the quality criteria required for parts of visible aspect.
- the figure 2 shows for example a micrographic section obtained on a wire 1 before (bottom view) and after (top view) the projection of a zinc powder rich in magnesium (3% by weight) and aluminum (8% by weight). It is clear that a new type of coating has been obtained with a layer richer in magnesium 3 and an intermetallic iron / zinc 2 containing a large amount of aluminum.
- the powder will preferably be unheated. It may, however, be preheated, preferably in a controlled atmosphere, below its melting point to improve the final quality of the coating. Similarly, the atmosphere around the projection area of the powder may be controlled for example to limit the oxidation of the coating and / or powder.
- Another advantage of the process according to the invention is that it has a great flexibility of application without necessity, as in the state of the technical, to control the composition and temperature of the dipping liquid metal bath, as well as the holding of all submerged equipment.
- Another original application of the process according to the invention consists in creating successive layers in number at least greater than two, in particular lubricating surface layers for forming the substrate.
- the coating obtained after the projection of powder may undergo a heat treatment and possibly a melting to modify the properties (structure, roughness, gloss, etc.). It may also undergo a "skin pass" (in the case of a sheet) or wire drawing to impose a specific roughness, able to receive other coatings of the organic type or to improve the adhesion of the powder.
- the metal powders mainly based on Zn, Al, Mg may be combined with other powders of the carbide or oxide type to increase the hardness of the final coating, to modify its appearance (gloss, etc.) or its adhesion properties. a future organic coating.
- the particle size of the powders may be quite large (between 1 .mu.m and 100 .mu.m) depending on the thickness of the final coating.
- the powder to be deposited will preferably always have the largest particle size possible to reduce its manufacturing cost and improve the quality of the final product. Indeed, since the magnesium-containing powders are always covered with an oxide, it is appropriate to minimize the pollution of the final coating by this oxide by choosing the largest possible particle size.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Mechanical Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Other Surface Treatments For Metallic Materials (AREA)
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP08447018A EP2103706A1 (de) | 2008-03-17 | 2008-03-17 | Legierung für Beschichtungen durch Pulverspritzen |
| PCT/EP2008/067942 WO2009083483A1 (fr) | 2007-12-20 | 2008-12-18 | Alliage de revetement obtenu par projection de poudre |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP08447018A EP2103706A1 (de) | 2008-03-17 | 2008-03-17 | Legierung für Beschichtungen durch Pulverspritzen |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| EP2103706A1 true EP2103706A1 (de) | 2009-09-23 |
Family
ID=39666233
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP08447018A Withdrawn EP2103706A1 (de) | 2007-12-20 | 2008-03-17 | Legierung für Beschichtungen durch Pulverspritzen |
Country Status (2)
| Country | Link |
|---|---|
| EP (1) | EP2103706A1 (de) |
| WO (1) | WO2009083483A1 (de) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| RU2417136C1 (ru) * | 2009-10-13 | 2011-04-27 | Федеральное Государственное Унитарное Предприятие "Центральный Научно-Исследовательский Институт Конструкционных Материалов "Прометей" (Фгуп "Цнии Км "Прометей") | Способ получения агломерированных дисперсных частиц системы "металл-неметалл" износостойкого класса |
Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1343654A (en) | 1971-06-11 | 1974-01-16 | Centre De Recherches Metallurgiques | Coating sheet metal |
| FR2354389A1 (fr) * | 1976-06-07 | 1978-01-06 | Kobe Steel Ltd | Procede de traitement de surface d'un metal par un systeme a lit fluidise applicable notamment a la galvanisation des fils d'acier |
| JPS5858263B2 (ja) | 1978-05-26 | 1983-12-24 | アルフレツド テヴエス ゲ−エムベ−ハ− | オ−トバイブレ−キ装置用ブレ−キ力調整器 |
| JPH0293053A (ja) * | 1988-09-29 | 1990-04-03 | Kobe Steel Ltd | 高耐蝕性溶融Zn−Mg系合金めっき鋼板の製造方法 |
| JPH02118088A (ja) | 1988-10-26 | 1990-05-02 | Nkk Corp | 加工性、塗装性に優れた合金化溶融亜鉛めっき鋼板の製造方法 |
| JPH02267250A (ja) | 1989-04-05 | 1990-11-01 | Kobe Steel Ltd | 溶融金属めっき装置用の粉末噴射装置及びその噴射方法 |
| WO1994029030A1 (en) * | 1993-06-11 | 1994-12-22 | Sms Engineering, Inc. | Method and apparatus to galvanize a ferrous substrate |
| EP0769567A1 (de) * | 1995-10-19 | 1997-04-23 | Sollac S.A. | Verfahren zur Beschichtung eines Blechs |
| JPH101766A (ja) | 1996-06-11 | 1998-01-06 | Nippon Steel Corp | 耐食性及び耐傷付き性に優れた溶射皮膜 |
| KR20010063534A (ko) | 1999-12-22 | 2001-07-09 | 이구택 | 내 파우더링성이 우수한 후도금 합금화 용융아연도금강판의 제조방법 |
| US20070240603A1 (en) * | 2004-02-13 | 2007-10-18 | Ko Kyung-Hyun | Porous Coated Member and Manufacturing Method Thereof Using Cold Spray |
-
2008
- 2008-03-17 EP EP08447018A patent/EP2103706A1/de not_active Withdrawn
- 2008-12-18 WO PCT/EP2008/067942 patent/WO2009083483A1/fr not_active Ceased
Patent Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1343654A (en) | 1971-06-11 | 1974-01-16 | Centre De Recherches Metallurgiques | Coating sheet metal |
| FR2354389A1 (fr) * | 1976-06-07 | 1978-01-06 | Kobe Steel Ltd | Procede de traitement de surface d'un metal par un systeme a lit fluidise applicable notamment a la galvanisation des fils d'acier |
| JPS5858263B2 (ja) | 1978-05-26 | 1983-12-24 | アルフレツド テヴエス ゲ−エムベ−ハ− | オ−トバイブレ−キ装置用ブレ−キ力調整器 |
| JPH0293053A (ja) * | 1988-09-29 | 1990-04-03 | Kobe Steel Ltd | 高耐蝕性溶融Zn−Mg系合金めっき鋼板の製造方法 |
| JPH02118088A (ja) | 1988-10-26 | 1990-05-02 | Nkk Corp | 加工性、塗装性に優れた合金化溶融亜鉛めっき鋼板の製造方法 |
| JPH02267250A (ja) | 1989-04-05 | 1990-11-01 | Kobe Steel Ltd | 溶融金属めっき装置用の粉末噴射装置及びその噴射方法 |
| WO1994029030A1 (en) * | 1993-06-11 | 1994-12-22 | Sms Engineering, Inc. | Method and apparatus to galvanize a ferrous substrate |
| EP0769567A1 (de) * | 1995-10-19 | 1997-04-23 | Sollac S.A. | Verfahren zur Beschichtung eines Blechs |
| JPH101766A (ja) | 1996-06-11 | 1998-01-06 | Nippon Steel Corp | 耐食性及び耐傷付き性に優れた溶射皮膜 |
| KR20010063534A (ko) | 1999-12-22 | 2001-07-09 | 이구택 | 내 파우더링성이 우수한 후도금 합금화 용융아연도금강판의 제조방법 |
| US20070240603A1 (en) * | 2004-02-13 | 2007-10-18 | Ko Kyung-Hyun | Porous Coated Member and Manufacturing Method Thereof Using Cold Spray |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| RU2417136C1 (ru) * | 2009-10-13 | 2011-04-27 | Федеральное Государственное Унитарное Предприятие "Центральный Научно-Исследовательский Институт Конструкционных Материалов "Прометей" (Фгуп "Цнии Км "Прометей") | Способ получения агломерированных дисперсных частиц системы "металл-неметалл" износостойкого класса |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2009083483A1 (fr) | 2009-07-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CA2343340C (fr) | Procede de realisation d'une piece a tres hautes caracteristiques mecaniques, mise en forme par emboutissage, a partir d'une bande de tole d'acier laminee et notamment laminee a chaud et revetue | |
| JP7732984B2 (ja) | 曲げ加工性及び耐食性に優れた溶融亜鉛めっき鋼板及びその製造方法 | |
| US3962501A (en) | Method for coating of corrosion-resistant molten alloy | |
| CA2926564C (fr) | Tole a revetement znalmg a flexibilite amelioree et procede de realisation correspondant | |
| EP2954086B1 (de) | Metallblech mit einer znalmg-beschichtung mit einer besonderen mikrostruktur und entsprechendes herstellungsverfahren | |
| CN101558182A (zh) | 热镀Zn-Al系合金钢板及其制造方法 | |
| EP2841613B1 (de) | Herstellung eines znalmg- vorbeschichteten blech und entsprechendes blech | |
| JP2804167B2 (ja) | 加工性に優れた合金化溶融亜鉛めつき鋼板及びその製造方法 | |
| JPH04176878A (ja) | プレス成形性、化成処理性に優れた亜鉛系めっき鋼板 | |
| EP2103706A1 (de) | Legierung für Beschichtungen durch Pulverspritzen | |
| EP0594520B1 (de) | Feuerverzinkte Stahlgegenstände und Verfahren zur Herstellung | |
| JP2000144366A (ja) | アルミニウム・亜鉛めっき浴用部材およびその製造方法 | |
| EP1989339A2 (de) | Stahlblech mit einer beschichtung auf aluminiumbasis | |
| EP0042636A2 (de) | Verfahren zur Oberflächenbehandlung von durch einen Überzug geschützten Oberflächen | |
| JPH0696783B2 (ja) | プレス成形性、化成処理性、溶接性に優れた亜鉛系めっき鋼板 | |
| BE897788A (fr) | Revetement a base de fer - zinc - aluminium et procedes pour le realiser | |
| EP0848076B1 (de) | Verfahren zur Heisstauch-Beschichtung einer Stahlplatte; galvanisierte oder aluminisierte Stahlplatte so hergestellt | |
| JPH0319297B2 (de) | ||
| EP0111039A1 (de) | Verfahren zum kontinuierlichen Tauchverzinken und Anlassen eines Metalldrahtes mit grosser Geschwindigkeit | |
| JPH09241812A (ja) | 合金化溶融亜鉛めっき鋼板 | |
| JPH04235266A (ja) | 加工性及び耐食性に優れた合金化溶融亜鉛めっき鋼板の製造方法 | |
| JP3275686B2 (ja) | プレス成形性に優れた合金化溶融亜鉛めっき鋼板 | |
| JP3095935B2 (ja) | 溶融Znメッキ方法 | |
| JP2000256825A (ja) | 亜鉛・アルミニウム熔融浴用摺動部材 | |
| LU86738A1 (fr) | Procede pour ameliorer les proprietes d'un substrat pourvu d'un revetement a base de zinc |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MT NL NO PL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL BA MK RS |
|
| AKX | Designation fees paid | ||
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: 8566 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION IS DEEMED TO BE WITHDRAWN |
|
| 18D | Application deemed to be withdrawn |
Effective date: 20100324 |