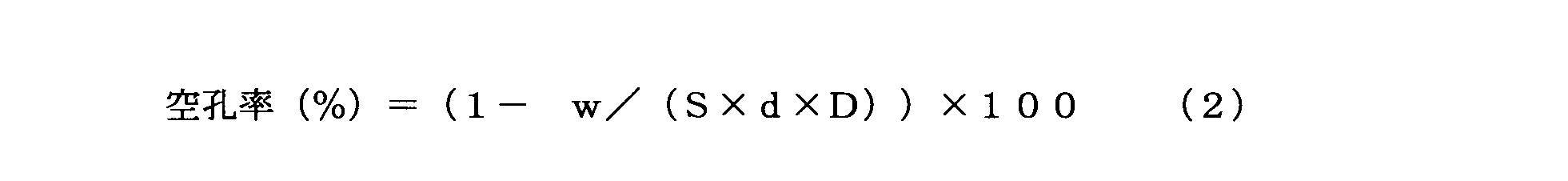WO2016121767A1 - ポリイミド多孔質膜を用いる細胞の長期培養、及びポリイミド多孔質膜を用いる細胞の凍結保存方法 - Google Patents
ポリイミド多孔質膜を用いる細胞の長期培養、及びポリイミド多孔質膜を用いる細胞の凍結保存方法 Download PDFInfo
- Publication number
- WO2016121767A1 WO2016121767A1 PCT/JP2016/052206 JP2016052206W WO2016121767A1 WO 2016121767 A1 WO2016121767 A1 WO 2016121767A1 JP 2016052206 W JP2016052206 W JP 2016052206W WO 2016121767 A1 WO2016121767 A1 WO 2016121767A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cells
- polyimide porous
- porous membrane
- cell
- culture
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/0068—General culture methods using substrates
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N1/00—Preservation of bodies of humans or animals, or parts thereof
- A01N1/10—Preservation of living parts
- A01N1/12—Chemical aspects of preservation
- A01N1/128—Chemically defined matrices for immobilising, holding or storing living parts, e.g. alginate gels; Chemically altering living parts, e.g. by cross-linking
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N1/00—Preservation of bodies of humans or animals, or parts thereof
- A01N1/10—Preservation of living parts
- A01N1/14—Mechanical aspects of preservation; Apparatus or containers therefor
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N1/00—Preservation of bodies of humans or animals, or parts thereof
- A01N1/10—Preservation of living parts
- A01N1/14—Mechanical aspects of preservation; Apparatus or containers therefor
- A01N1/146—Non-refrigerated containers specially adapted for transporting or storing living parts whilst preserving
- A01N1/147—Carriers for immersion in cryogenic fluid for slow freezing or vitrification
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N1/00—Preservation of bodies of humans or animals, or parts thereof
- A01N1/10—Preservation of living parts
- A01N1/16—Physical preservation processes
- A01N1/162—Temperature processes, e.g. following predefined temperature changes over time
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/18—Manufacture of films or sheets
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J9/00—Working-up of macromolecular substances to porous or cellular articles or materials; After-treatment thereof
- C08J9/28—Working-up of macromolecular substances to porous or cellular articles or materials; After-treatment thereof by elimination of a liquid phase from a macromolecular composition or article, e.g. drying of coagulum
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J9/00—Working-up of macromolecular substances to porous or cellular articles or materials; After-treatment thereof
- C08J9/36—After-treatment
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12M—APPARATUS FOR ENZYMOLOGY OR MICROBIOLOGY; APPARATUS FOR CULTURING MICROORGANISMS FOR PRODUCING BIOMASS, FOR GROWING CELLS OR FOR OBTAINING FERMENTATION OR METABOLIC PRODUCTS, i.e. BIOREACTORS OR FERMENTERS
- C12M25/00—Means for supporting, enclosing or fixing the microorganisms, e.g. immunocoatings
- C12M25/02—Membranes; Filters
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12M—APPARATUS FOR ENZYMOLOGY OR MICROBIOLOGY; APPARATUS FOR CULTURING MICROORGANISMS FOR PRODUCING BIOMASS, FOR GROWING CELLS OR FOR OBTAINING FERMENTATION OR METABOLIC PRODUCTS, i.e. BIOREACTORS OR FERMENTERS
- C12M29/00—Means for introduction, extraction or recirculation of materials, e.g. pumps
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N1/00—Microorganisms; Compositions thereof; Processes of propagating, maintaining or preserving microorganisms or compositions thereof; Processes of preparing or isolating a composition containing a microorganism; Culture media therefor
- C12N1/04—Preserving or maintaining viable microorganisms
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N1/00—Microorganisms; Compositions thereof; Processes of propagating, maintaining or preserving microorganisms or compositions thereof; Processes of preparing or isolating a composition containing a microorganism; Culture media therefor
- C12N1/14—Fungi; Culture media therefor
- C12N1/16—Yeasts; Culture media therefor
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N1/00—Microorganisms; Compositions thereof; Processes of propagating, maintaining or preserving microorganisms or compositions thereof; Processes of preparing or isolating a composition containing a microorganism; Culture media therefor
- C12N1/20—Bacteria; Culture media therefor
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0652—Cells of skeletal and connective tissues; Mesenchyme
- C12N5/0656—Adult fibroblasts
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0684—Cells of the urinary tract or kidneys
- C12N5/0686—Kidney cells
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2379/00—Characterised by the use of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen with or without oxygen, or carbon only, not provided for in groups C08J2361/00 - C08J2377/00
- C08J2379/04—Polycondensates having nitrogen-containing heterocyclic rings in the main chain; Polyhydrazides; Polyamide acids or similar polyimide precursors
- C08J2379/08—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2533/00—Supports or coatings for cell culture, characterised by material
- C12N2533/30—Synthetic polymers
Definitions
- the present invention relates to a method for long-term cell culture, a cell culture device, and a kit. In detail, it is related with the long-term culture
- the present invention also relates to a cell cryopreservation method and kit. In detail, it is related with the cryopreservation method and kit of the cell using a polyimide porous membrane.
- Cell culture cells generally exist as a three-dimensional population in vivo, but in classic planar culture, cells are cultivated in a single layer so that the cells stick to the container.
- adherent cells various culture methods have been developed by Petri et al. When culturing with this petri dish or the like, the cultured cells continue to grow and become unable to grow any more, called “confluent”, and the growth stops. Although it depends on the cell type, many cells have also found that when this confluent state is continued without passage, detachment starts spontaneously after a certain period of time, and the cells cannot be passaged. Yes.
- the feature of these methodologies is the expansion of the breeding place by giving a three-dimensional environment to the growth space using the carrier, and the carrier itself is protected in a strong environment by equipment and method ingenuity at this large growth place.
- the purpose is to extend the culture period.
- the system used for these methods is a closed system that requires a complicated device or a large-volume device, or that does not participate in the culture environment itself once the culture starts. There are many cases. For this reason, a methodology capable of culturing cells for a long period of time and easily handling the system is desired, but an appropriate methodology has not been developed.
- Non-Patent Documents 1 and 2 there have been reported research examples using a three-dimensional carrier for a long-term cell culture method simulating an in vivo organ. These are examples of the results of research aiming at site-specific reconstruction, such as in vivo experiments on the reconstruction of pancreatic islets of Langerhans and long-term culture of bone marrow cells in vitro. This is an important achievement that shows the significance and value of cell culture in a three-dimensional environment for the long-term culture, but each carrier has high specificity for each application, and the materials used are biocompatible materials. Because of the high-order structure constituted by the fibrous structure and plotting, the versatility is poor and the handling is also lacking in generality. There is a demand for the development of methodologies that can be used in a more convenient and adaptable manner.
- the cells are roughly classified into two types, floating cells and adherent cells, depending on the characteristics of their survival form.
- any cell When any cell is subjected to artificial culture, it undergoes a cycle of cell seeding, culture, growth, passage, and preservation by freezing.
- Patent Document 4 A technique that tries to freeze cells on the culture plate itself more easily than the classical freezing method (Patent Document 4), or a method that aims to improve cell viability in freezing using a three-dimensional carrier (Patent Document 5)
- Patent Document 6 a technique that tries to freeze cells on the culture plate itself more easily than the classical freezing method
- Patent Document 5 a method that aims to improve cell viability in freezing using a three-dimensional carrier
- Patent Document 6 there are reports such as an example of improving the cryopreservation characteristics of cultured cells using a fibrous carrier (Patent Document 6) and an example of verifying the survival efficiency of stem cells (Non-Patent Document 3).
- cryopreservation medium is limited to a fibrous material having a specific structure. Further, although this material has a function as a cryopreservation medium, it is not a material that can be used as a cell culture carrier as it is, and is temporarily used. It remains to be used as a preservation body. There is a demand for the construction of a new methodology that can carry out consistently from cryopreservation of cells to cell thawing (cell wake-up) and use to refreezing in a simple and efficient manner.
- the polyimide porous membrane polyimide is a general term for polymers containing an imide bond in a repeating unit.
- the aromatic polyimide means a polymer in which aromatic compounds are directly linked by an imide bond.
- Aromatic polyimide has a conjugated structure through the imide bond between aromatic and aromatic, so it has a rigid and strong molecular structure, and the imide bond has a strong intermolecular force, so it has a very high level of heat. Has mechanical, mechanical and chemical properties.
- Patent Documents 7 to 9 are particularly excellent in permeability of substances such as gas, high porosity, excellent smoothness of both surfaces, relatively high in strength, and in the film thickness direction despite high porosity.
- a polyimide porous membrane having a large number of macrovoids having excellent proof stress against compressive stress is described.
- the present invention is capable of easily and stably culturing cells for a long period of time according to the required situation and form, and can easily cope with changes during the culture, and It aims at providing the cell culture apparatus and kit for using for the said culture
- Another object of the present invention is to provide a method for freezing cells that can be carried out simply and efficiently from cell cryopreservation to cell thawing (cell wake-up) and from use to refreezing. .
- this invention preferably includes the following aspects.
- a long-term culture method for cells (1) applying cells to a polyimide porous membrane; (2) A method comprising applying a polyimide porous membrane to which cells are applied to a cell culture medium and culturing the cells for 30 days or more.
- Aspect 2 The method according to aspect 1, wherein the cells are cultured for 60 days or longer in step (2).
- Aspect 3 The method according to aspect 1, wherein the cells are cultured for 120 days or more in step (2).
- the culture in the step (2) is performed in a system in which the cell culture medium is continuously or intermittently supplied into the cell culture container from the cell culture medium supply means installed outside the cell culture container.
- [Aspect 10] The method according to embodiment 9, wherein the cell culture medium circulates between the cell culture medium supply means and the cell culture container.
- [Aspect 11] The method according to embodiment 9 or 10, wherein the system includes a culture unit that is a cell culture container and a culture medium supply unit that is a cell culture medium supply means, wherein the culture unit is a cell.
- the culture medium supply unit includes a culture medium storage container, a culture medium supply line, and a liquid feed pump that continuously or intermittently feeds the culture medium via the culture medium supply line, where the first end of the culture medium supply line is The medium supply unit is in contact with the medium in the medium container, and the second end of the medium supply line communicates with the culture unit via the medium supply port of the culture unit.
- the culture unit further comprises a medium discharge line, wherein the first end of the medium discharge line is connected to the medium storage container, and the second end of the medium discharge line is connected to the culture unit via the medium discharge port of the culture unit.
- the method according to the aspect 11 or 12, wherein the medium can be circulated between the medium supply unit and the culture unit.
- the cells are selected from the group consisting of animal cells, insect cells, plant cells, yeasts and bacteria.
- the animal cell is a cell derived from an animal belonging to the vertebrate phylum.
- the cells are selected from the group consisting of CHO cells, CHO-K1 cell lines, CHO DP-12 cell lines, CHO cell related lines, Vero cells, and MDCK cells.
- the polyimide porous membrane is a polyimide porous membrane containing a polyimide obtained from tetracarboxylic dianhydride and diamine.
- the polyimide porous membrane was colored by forming a polyamic acid solution composition containing a polyamic acid solution obtained from tetracarboxylic dianhydride and diamine and a colored precursor, and then heat-treating at 250 ° C. or higher.
- a cell cryopreservation method comprising: (1) a step of supporting cells on a polyimide porous membrane; (2) a step of freezing cells carried on the polyimide porous membrane by placing the polyimide porous membrane carrying cells on the condition that the cells are frozen; and (3) a polyimide porous membrane carrying cells.
- a method comprising the step of storing the membrane under conditions that keep it frozen.
- step (1) the cells are seeded on the polyimide porous membrane and cultured so that the cells are supported on the polyimide porous membrane.
- step (3) the method according to any one of embodiments 23 to 25, further comprising a step of thawing the cells supported on the polyimide porous membrane by placing the polyimide porous membrane supporting the cells under conditions for thawing the cells.
- step 27 Following step (4), (5) The method according to aspect 26, further comprising a step of applying the polyimide porous membrane after thawing the cells to the cell culture medium and culturing the cells.
- step (5) the cells are cultured until the cultured cells proliferate outside the polyimide porous membrane.
- step (5) another one or more polyimide porous membranes not carrying cells are applied to the cell culture medium together with the polyimide porous membrane from which the cells have been thawed, and another one or more are obtained by culturing. 29.
- step 30 Following step (5), (6) A step of freezing the cells supported on the polyimide porous membrane by placing a part or all of the polyimide porous membrane supporting the cells under conditions in which the cells are frozen, and (7) 30.
- steps (1) to (7) are repeated a plurality of times.
- the cell is selected from the group consisting of animal cells, insect cells, plant cells, yeasts and bacteria.
- the animal cell is a cell derived from an animal belonging to the vertebrate phylum.
- the cell is an adherent cell.
- polyimide porous membrane is a multilayer porous polyimide membrane having two different surface layers and a macrovoid layer.
- Polyimide porous membrane for cryopreservation of cells are a polyimide porous membrane for cryopreservation of cells.
- the present invention makes it possible to culture cells easily and stably for a long period of time by using a polyimide porous membrane.
- the polyimide porous membrane serving as a culture carrier has a large-diameter connecting hole through which cells can pass, even if a very large amount of cells inhabit the space, the three-dimensional space is finally obtained. Is secured. Therefore, the limit of the culture period is less likely to occur when a confluent state occurs in classical planar culture.
- the polyimide porous film is a flexible thin film material, it can be easily bent, folded or cut into a free shape.
- the polyimide porous membrane containing cells it is possible to take out the polyimide porous membrane containing cells at any time through culture and use them for treatment, measurement and the like. It can also be applied to automation of cell culture. Furthermore, since the polyimide porous membrane is a material having very excellent heat resistance, operations such as sterilization can be carried out very simply.
- the polyimide porous membrane By freezing the cells using the polyimide porous membrane, it is possible to efficiently cryopreserve a large number of cells and operate the cells efficiently. After thawing from the frozen state, cell culture can be continued on the polyimide porous membrane as it is. Furthermore, the cell is moved / proliferated by contacting the polyimide porous membrane in which cell culture is continued with another carrier such as a polyimide porous membrane in which cells are not grown or in the vicinity thereof. Can do. After the transfer of cells, the original polyimide porous membrane can be taken out and re-frozen, and stored until the next use. In this way, a series of operations from cell raising to use / freezing can be performed with the same material.
- a cell detachment operation such as trypsin and a complicated operation such as collagen coating are not particularly necessary. Therefore, the operation is simple and efficient, and a rapid operation is possible.
- the method according to the invention is also suitable for applications involving automated processes. Also in terms of efficiency, since a large amount of cells can be grown on a very thin film of at least 25 micrometers, the efficiency per volume is unprecedentedly high. For example, when a substance is produced using a promising cell line or the like, in addition to long-term use, it is possible to freeze the cells as they are and store them again, and use them again when necessary. A library of promising cell lines can be easily implemented.
- FIG. 1 shows a model diagram of cell culture using a polyimide porous membrane.
- FIG. 2 shows an example of a cell culture device.
- FIG. 3 shows the results of long-term culture using a polyimide porous membrane of human skin fibroblasts.
- FIG. 4 shows the results of long-term culture using a polyimide porous membrane of human dermal fibroblasts.
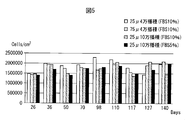
- FIG. 5 shows the results of long-term culture using a Vero cell polyimide porous membrane.
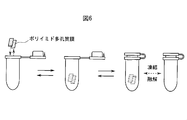
- FIG. 6 shows a conceptual diagram of cell freezing and thawing processes using a polyimide porous membrane. Unlike the cell suspension freezing / thawing process, it is possible to directly handle the cell aggregate as a polyimide porous membrane, so that steps such as centrifugation are drastically omitted.
- FIG. 1 shows a model diagram of cell culture using a polyimide porous membrane.
- FIG. 2 shows an example of a cell culture device.
- FIG. 3 shows the results of long-term culture using a
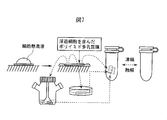
- FIG. 7 shows a conceptual diagram regarding freezing / thawing and operation of suspension cells using a polyimide porous membrane.
- Suspension cells have shown the advantage of repeated use as a sample, taking advantage of the property of overflowing from the polyimide porous membrane in the process of growth.
- FIG. 8 shows the results of long-term culture using a polyimide porous membrane of human dermal fibroblasts.
- FIG. 9 shows the results of long-term culture using a polyimide porous membrane of human skin fibroblasts.
- FIG. 10 shows the results of freezing, thawing and culturing CHO DP-12 cells using a polyimide porous membrane.
- FIG. 11 shows the results of freezing, thawing and culturing human dermal fibroblasts using a polyimide porous membrane.
- the present invention relates to a long-term culture method for cells.
- the entire contents of International Application No. PCT / JP2014 / 070407 are incorporated herein by reference.
- the cell culture method of the present invention includes applying the cells to a polyimide porous membrane and culturing.
- the present inventors have found that a polyimide porous membrane is suitable for cell adhesion and culture, and have arrived at the present invention.
- the method of the present invention includes applying cells to a polyimide porous membrane and culturing the cells on or inside the polyimide membrane.
- A an embodiment comprising a step of seeding cells on the surface of the polyimide porous membrane;
- B Place a cell suspension on the dried surface of the polyimide porous membrane, Leave or move the polyimide porous membrane to promote fluid outflow, or stimulate a portion of the surface to draw cell suspension into the membrane; and The cells in the cell suspension are retained in the membrane, and the water flows out.
- An embodiment comprising steps; and
- C Wet one side or both sides of the polyimide porous membrane with a cell culture medium or a sterilized liquid, Loading the wet polyimide porous membrane with a cell suspension; and The cells in the cell suspension are retained in the membrane, and the water flows out.
- a mode comprising the steps.
- the mode includes directly seeding cells and cell clusters on the surface of the polyimide porous membrane. Or the aspect which puts a polyimide porous membrane in a cell suspension and infiltrate a cell culture solution from the surface of a membrane is also included.
- the cells seeded on the surface of the polyimide porous membrane adhere to the polyimide porous membrane and enter the inside of the porous body.
- the cells spontaneously adhere to the polyimide porous membrane without any physical or chemical force applied from the outside.
- Cells seeded on the surface of the polyimide porous membrane can grow and proliferate stably on the surface and / or inside of the membrane. Cells can take a variety of different forms depending on the location of the membrane in which they grow and multiply.
- a cell suspension is placed on the dried surface of the polyimide porous membrane.
- the cell suspension is sucked into the membrane, Cell suspension penetrates into the membrane. Without being bound by theory, it is considered that this is due to the properties derived from the surface shape of the polyimide porous membrane.
- the cells are sucked and seeded at the portion of the membrane where the cell suspension is loaded.
- one or both sides or the whole of the polyimide porous membrane is wetted with a cell culture medium or a sterilized liquid, and then the suspended polyimide porous membrane is subjected to cell suspension.
- the liquid may be loaded. In this case, the passage speed of the cell suspension is greatly improved.
- a method of wetting a part of the membrane electrode for the main purpose of preventing the scattering of the membrane (hereinafter referred to as “one-point wet method”) can be used.
- the one-point wet method is substantially similar to the dry method (the embodiment (B)) that does not substantially wet the film.
- a method in which a cell suspension is loaded into a fully porous one or both surfaces of a polyimide porous membrane hereinafter referred to as “wet membrane”).
- this Is described as “wet film method”.
- the passage speed of the cell suspension is greatly improved in the entire polyimide porous membrane.
- the cells in the cell suspension are retained in the membrane and the water is allowed to flow out.
- processing such as concentrating the concentration of cells in the cell suspension or allowing unnecessary components other than cells to flow out together with moisture.
- the mode of (A) may be referred to as “natural sowing” (B) and the mode of (C) as “suction sowing”.
- living cells remain selectively in the polyimide porous membrane. Therefore, in a preferred embodiment of the present invention, living cells remain in the polyimide porous membrane, and dead cells preferentially flow out with moisture.
- the sterilized liquid used in the embodiment (C) is not particularly limited, but is a sterilized buffer or sterilized water.
- the buffer include (+) and ( ⁇ ) Dulbecco ’s PBS, (+) and ( ⁇ ) Hank's Balanced Salt Solution. Examples of buffer solutions are shown in Table 1 below.
- the application of the cells to the polyimide porous membrane is an embodiment in which cells are attached to the membrane by allowing the adhesive cells in a suspended state to coexist with the polyimide porous membrane (entanglement). )
- a cell culture medium, cells, and one or more of the polyimide porous membranes may be placed in a cell culture container in order to apply the cells to the polyimide porous membrane.
- the cell culture medium is liquid
- the polyimide porous membrane is suspended in the cell culture medium. Due to the nature of the polyimide porous membrane, cells can adhere to the polyimide porous membrane.
- the polyimide porous membrane can be cultured in a suspended state in the cell culture medium.
- the cells spontaneously adhere to the polyimide porous membrane. “Spontaneously adheres” means that the cells remain on or inside the polyimide porous membrane without any physical or chemical force applied from the outside.
- cultured cells can be classified into adhesion culture cells and suspension culture cells depending on the form of cell culture.
- Adherent culture cells are cultured cells that adhere to a culture vessel and proliferate, and the medium is changed during passage.
- Floating culture cells are cultured cells that proliferate in a floating state in a medium. In general, dilution culture is performed without replacing the medium during passage.
- Suspension culture can be cultured in a floating state, that is, in a liquid state, so that it can be cultured in large quantities. Compared with adherent cells that grow only on the surface of the culture vessel, it is a three-dimensional culture. There is an advantage that the number of cells that can be cultured is large.
- the polyimide porous membrane when used in a suspended state in the cell culture medium, two or more pieces of the polyimide porous membrane may be used. Since the polyimide porous membrane is a flexible thin film, for example, it is possible to bring a polyimide porous membrane having a large surface area into a certain volume of cell culture medium by using small pieces suspended in the culture medium. Become. In the case of normal culture, the bottom area of the container is the upper limit of the cell culture area, but in the cell culture using the polyimide porous membrane of the present invention, all of the large surface area of the previously introduced polyimide porous membrane is the cell. It becomes the area that can be cultured. Since the polyimide porous membrane allows the cell culture solution to pass therethrough, for example, nutrients, oxygen and the like can be supplied into the folded membrane.
- the size and shape of the polyimide porous membrane pieces are not particularly limited.
- the shape can take any shape such as a circle, an ellipse, a square, a triangle, a polygon, and a string.
- the polyimide porous membrane of the present invention has flexibility, it can be used by changing its shape.
- the polyimide porous membrane may be processed into a three-dimensional shape instead of a flat shape.
- a polyimide porous membrane is i) folded and ii) rolled into a roll, iii) a sheet or piece is connected with a thread-like structure, or iv) tied in a rope shape in a cell culture vessel May be suspended or fixed in the cell culture medium.
- many polyimide porous membranes can be placed in a fixed volume of cell culture medium, as in the case of using small pieces.
- each piece can be handled as an aggregate, cell bodies can be aggregated and moved, and the overall applicability is high.
- two or more polyimide porous membranes may be used by being laminated in a cell culture medium vertically or horizontally.
- Lamination also includes a mode in which the polyimide porous membranes partially overlap. Stacked culture enables cells to be cultured at high density in a narrow space. It is also possible to form a multilayer system with different types of cells by further stacking the membranes on the membrane where the cells are already grown.
- the number of polyimide porous membranes to be laminated is not particularly limited.
- the above-described cell culture method of the present invention may be used in combination of two or more methods.
- the cells may be first applied to the polyimide porous membrane using any one of the methods (A) to (C), and then the polyimide porous membrane to which the cells are adhered may be subjected to suspension culture.
- any one of the methods (A) to (C) may be used in combination of two or more.
- the cells preferably grow and proliferate on and inside the polyimide porous membrane.
- the cells can be used for a long period of 30 days or more, 60 days or more, 120 days or more, 200 days or more, or 300 days or more without performing subculture operations such as conventional trypsin treatment. It can be cultured.
- the period can be cultivated by the conventional flat culture for longer than, for example, 1.5 times, 2 times, 2.5 times, 3 times, 3 times Cells can be cultured for a period of 5 times or more, 4 times or more, or 4.5 times or more.
- the present invention it is possible to maintain a dynamic life, not a resting state, for a long period of time without causing detachment or death of cells generated by long-term cell culture in a petri dish or the like.
- cell viability or cell properties for example, differentiation induction efficiency, expression level of cell surface marker, etc.
- cells grow three-dimensionally in the polyimide porous membrane contact inhibition caused by the limitation of the culture area and the planar environment as seen in conventional planar culture is unlikely to occur.
- the culture can be grown for a long period of time.
- the present invention it is possible to arbitrarily increase the space in which cell culture is possible by bringing another polyimide porous membrane into contact with the polyimide porous membrane to which cells are adhered. Without performing a subculture operation with treatment, it is possible to perform culture that grows for a long period of time while avoiding a confluent state that causes contact inhibition.
- a new storage method in which cells are stored for a long period of time without being frozen.
- Cells The types of cells that can be used in the method of the present invention are not particularly limited, and can be used for the growth of arbitrary cells.
- the cells are selected from the group consisting of animal cells, insect cells, plant cells, yeasts and bacteria.
- Animal cells are roughly classified into cells derived from animals belonging to the vertebrate phylum and cells derived from invertebrates (animals other than animals belonging to the vertebrate phylum).
- the origin of the animal cell is not particularly limited.
- Vertebrates include the maxilla and maxilla, and the maxilla includes mammals, birds, amphibians, reptiles, and the like.
- it is a cell derived from an animal belonging to the mammal class generally called a mammal. Mammals are not particularly limited, but preferably include mice, rats, humans, monkeys, pigs, dogs, sheep, goats and the like.
- Plant cells including moss plants, fern plants, and seed plants are targeted.
- Plants from which seed plant cells are derived include monocotyledonous plants and dicotyledonous plants.
- monocotyledonous plants include orchids, gramineous plants (rice, corn, barley, wheat, sorghum, etc.), cyperaceae plants, and the like.
- Dicotyledonous plants include plants belonging to many subclasses such as Chrysanthemum, Magnolia, and Rose.
- Algae can also be regarded as cell-derived organisms. Different from eubacteria, cyanobacteria (Cyanobacteria), eukaryotes that are unicellular (diatoms, yellow green algae, dinoflagellates, etc.) and multicellular organisms, seaweeds (red algae, brown algae, green algae) Includes groups.
- the archaea and the types of bacteria in this specification are not particularly limited.
- the archaea is composed of a group consisting of methane bacteria, highly halophilic bacteria, thermophilic acidophiles, hyperthermophilic bacteria, and the like.
- the bacterium is selected from the group consisting of lactic acid bacteria, Escherichia coli, Bacillus subtilis, cyanobacteria and the like.
- animal cells or plant cells that can be used in the method of the present invention are not limited, but are preferably selected from the group consisting of pluripotent stem cells, tissue stem cells, somatic cells, and germ cells.
- pluripotent stem cell is intended to be a generic term for stem cells having the ability to differentiate into cells of any tissue (differentiation pluripotency).
- the pluripotent stem cells include, but are not limited to, embryonic stem cells (ES cells), induced pluripotent stem cells (iPS cells), embryonic germ stem cells (EG cells), germ stem cells (GS cells), and the like. .
- ES cells embryonic stem cells
- iPS cells induced pluripotent stem cells
- EG cells embryonic germ stem cells
- GS cells germ stem cells
- Any known pluripotent stem cell can be used.
- the pluripotent stem cell described in International Publication WO2009 / 123349 PCT / JP2009 / 057041
- PCT / JP2009 / 057041 can be used.
- tissue stem cell means a stem cell that has the ability to differentiate into various cell types (differentiated pluripotency) although the cell line that can be differentiated is limited to a specific tissue.
- hematopoietic stem cells in the bone marrow become blood cells, and neural stem cells differentiate into nerve cells.
- the tissue stem cells are selected from mesenchymal stem cells, hepatic stem cells, pancreatic stem cells, neural stem cells, skin stem cells, or hematopoietic stem cells.
- somatic cells refers to cells other than germ cells among the cells constituting multicellular organisms. In sexual reproduction, it is not passed on to the next generation.
- the somatic cells are hepatocytes, pancreatic cells, muscle cells, bone cells, osteoblasts, osteoclasts, chondrocytes, adipocytes, skin cells, fibroblasts, pancreatic cells, kidney cells, lung cells, or , Lymphocytes, erythrocytes, leukocytes, monocytes, macrophages or megakaryocyte blood cells.
- Reproductive cells means cells that have a role in transmitting genetic information to the next generation in reproduction. For example, gametes for sexual reproduction, ie eggs, egg cells, sperm, sperm cells, spores for asexual reproduction, and the like.
- the cells may be selected from the group consisting of sarcoma cells, established cells and transformed cells.
- “Sarcoma” is a cancer that develops in connective tissue cells derived from non-epithelial cells such as bone, cartilage, fat, muscle, blood, etc., and includes soft tissue sarcoma, malignant bone tumor and the like.
- Sarcoma cells are cells derived from sarcomas.
- the “established cell” means a cultured cell that has been maintained outside the body for a long period of time, has a certain stable property, and is capable of semi-permanent subculture.
- PC12 cells derived from rat adrenal medulla
- CHO cells derived from Chinese hamster ovary
- HEK293 cells derived from human fetal kidney
- HL-60 cells derived from human leukocyte cells
- HeLa cells derived from human cervical cancer
- Vero cells There are cell lines derived from various tissues of various biological species including humans (derived from African green monkey kidney epithelial cells), MDCK cells (derived from canine kidney tubular epithelial cells), HepG2 cells (derived from human liver cancer).
- a “transformed cell” means a cell in which a nucleic acid (DNA or the like) has been introduced from the outside of the cell to change its genetic properties. Appropriate methods are known for transformation of animal cells, plant cells, and bacteria.
- Polyimide Porous Membrane Polyimide is a general term for polymers containing imide bonds in repeating units, and usually means an aromatic polyimide in which aromatic compounds are directly linked by imide bonds.
- Aromatic polyimide has a conjugated structure through the imide bond between aromatic and aromatic, so it has a rigid and strong molecular structure, and the imide bond has a strong intermolecular force, so it has a very high level of heat. Has mechanical, mechanical and chemical properties.
- the polyimide porous membrane used in the present invention is a polyimide porous membrane containing a polyimide obtained from tetracarboxylic dianhydride and diamine (as a main component), more preferably from tetracarboxylic dianhydride and diamine.
- This is a polyimide porous membrane made of the resulting polyimide.
- “Containing as a main component” means that a component other than polyimide obtained from tetracarboxylic dianhydride and diamine may be essentially not included or included as a component of the polyimide porous membrane. It means that it is an additional component that does not affect the properties of the polyimide obtained from tetracarboxylic dianhydride and diamine.
- a colored polyimide porous film obtained by molding a polyamic acid solution composition containing a polyamic acid solution obtained from a tetracarboxylic acid component and a diamine component and a colored precursor and then heat-treating at 250 ° C. or higher. .
- Polyamic acid A polyamic acid is obtained by polymerizing a tetracarboxylic acid component and a diamine component.
- Polyamic acid is a polyimide precursor that can be ring-closed to form polyimide by thermal imidization or chemical imidization.
- the polyamic acid even if a part of the amic acid is imidized, it can be used as long as it does not affect the present invention. That is, the polyamic acid may be partially thermally imidized or chemically imidized.
- fine particles such as an imidization catalyst, an organic phosphorus-containing compound, inorganic fine particles, and organic fine particles can be added to the polyamic acid solution as necessary.
- fine particles such as a chemical imidating agent, a dehydrating agent, inorganic fine particles, and organic fine particles, etc. can be added to a polyamic acid solution as needed. Even when the above components are added to the polyamic acid solution, it is preferable that the coloring precursor is not precipitated.
- the colored precursor means a precursor that is partially or wholly carbonized by heat treatment at 250 ° C. or higher to produce a colored product.
- the colored precursor used in the present invention is uniformly dissolved or dispersed in a polyamic acid solution or a polyimide solution, and heat treatment at 250 ° C. or higher, preferably 260 ° C. or higher, more preferably 280 ° C. or higher, more preferably 300 ° C. or higher.
- heat treatment at 250 ° C. or higher, preferably 260 ° C. or higher, more preferably 280 ° C. or higher, more preferably 300 ° C. or higher in the presence of oxygen such as air to produce a colored product by carbonization
- oxygen such as air
- the carbon-based coloring precursor is not particularly limited.
- polymers such as petroleum tar, petroleum pitch, coal tar, coal pitch, or polymers obtained from monomers including pitch, coke, and acrylonitrile, ferrocene compounds (ferrocene and ferrocene derivatives). Etc.
- the polymer and / or ferrocene compound obtained from the monomer containing acrylonitrile are preferable, and polyacrylonitrile is preferable as a polymer obtained from the monomer containing acrylonitrile.
- tetracarboxylic dianhydride any tetracarboxylic dianhydride can be used, and can be appropriately selected according to desired characteristics.
- tetracarboxylic dianhydride include pyromellitic dianhydride, 3,3 ′, 4,4′-biphenyltetracarboxylic dianhydride (s-BPDA), 2,3,3 ′, 4 ′.
- -Biphenyltetracarboxylic dianhydride such as biphenyltetracarboxylic dianhydride (a-BPDA), oxydiphthalic dianhydride, diphenylsulfone-3,4,3 ', 4'-tetracarboxylic dianhydride, bis (3,4-dicarboxyphenyl) sulfide dianhydride, 2,2-bis (3,4-dicarboxyphenyl) -1,1,1,3,3,3-hexafluoropropane dianhydride, 2, 3,3 ′, 4′-benzophenone tetracarboxylic dianhydride, 3,3 ′, 4,4′-benzophenone tetracarboxylic dianhydride, bis (3,4-dicarboxyphenyl) methane dianhydride 2,2-bis (3,4-dicarboxyphenyl) propane dianhydride, p-phenylenebis (trimellitic acid monoester acid an
- At least one aromatic tetracarboxylic dianhydride selected from the group consisting of biphenyltetracarboxylic dianhydride and pyromellitic dianhydride is particularly preferable.
- the biphenyltetracarboxylic dianhydride 3,3 ′, 4,4′-biphenyltetracarboxylic dianhydride can be suitably used.
- diamines include the following. 1) One benzene nucleus such as 1,4-diaminobenzene (paraphenylenediamine), 1,3-diaminobenzene, 2,4-diaminotoluene, 2,6-diaminotoluene, etc .; 2) 4,4'-diaminodiphenyl ether, diaminodiphenyl ether such as 3,4'-diaminodiphenyl ether, 4,4'-diaminodiphenylmethane, 3,3'-dimethyl-4,4'-diaminobiphenyl, 2,2'- Dimethyl-4,4′-diaminobiphenyl, 2,2′-bis (trifluoromethyl) -4,4′-diaminobiphenyl, 3,3′-dimethyl-4,4′-diaminodiphenylmethane, 3,3′- Dicar
- the diamine to be used can be appropriately selected according to desired characteristics.
- aromatic diamine compounds are preferable, and 3,3′-diaminodiphenyl ether, 3,4′-diaminodiphenyl ether, 4,4′-diaminodiphenyl ether and paraphenylenediamine, 1,3-bis (3-aminophenyl) Benzene, 1,3-bis (4-aminophenyl) benzene, 1,4-bis (3-aminophenyl) benzene, 1,4-bis (4-aminophenyl) benzene, 1,3-bis (4-amino) Phenoxy) benzene and 1,4-bis (3-aminophenoxy) benzene can be preferably used.
- at least one diamine selected from the group consisting of benzenediamine, diaminodiphenyl ether and bis (aminophenoxy) phenyl is
- the polyimide porous membrane has a glass transition temperature of 240 ° C. or higher, or a tetracarboxylic dianhydride and a diamine having a clear transition point of 300 ° C. or higher. It is preferable that it is formed from the polyimide obtained combining these.
- the polyimide porous membrane of the present invention is preferably a polyimide porous membrane made of the following aromatic polyimide from the viewpoints of heat resistance and dimensional stability at high temperatures.
- an aromatic polyimide comprising at least one tetracarboxylic acid unit selected from the group consisting of a biphenyltetracarboxylic acid unit and a pyromellitic acid unit, and an aromatic diamine unit
- an aromatic polyimide comprising a tetracarboxylic acid unit and at least one aromatic diamine unit selected from the group consisting of a benzenediamine unit, a diaminodiphenyl ether unit and a bis (aminophenoxy) phenyl unit
- a polyimide porous film a polyimide having a multilayer structure having at least two surface layers (A surface and B surface) and a macrovoid layer sandwiched between the two surface layers Porous membranes can be used in the method of the present invention.
- the polyimide porous film is a film in which the macrovoid layer is surrounded by a partition wall bonded to the surface layer (A surface and B surface), and the partition wall and the surface layer (A surface and B surface).
- a plurality of macrovoids having an average pore diameter in the plane direction of 10 to 500 ⁇ m, and the partition walls of the macrovoid layer and the surface layers (A surface and B surface) each have a thickness of 0.01 to 20 ⁇ m.
- the polyimide porous film has a total film thickness of 5 to 500 ⁇ m and a porosity of 40% or more and less than 95%.
- the total film thickness of the polyimide porous film used in the present invention is not limited, but may be 20 to 75 ⁇ m as one aspect. Due to the difference in film thickness, differences in cell growth rate, cell morphology, in-plane cell saturation, etc. can be observed.
- the average hole diameter of the holes may be different from the average hole diameter of the holes existing on the B surface.
- the average pore diameter of the holes present on the A plane is smaller than the average pore diameter of the holes present on the B plane.
- the average pore diameter of the holes existing on the A plane is smaller than the average pore diameter of the holes existing on the B plane, and the average pore diameter of the holes existing on the A plane is 0.01 to 50 ⁇ m, 0.01 ⁇ m to 40 ⁇ m, 0 0.01 ⁇ m to 30 ⁇ m, 0.01 ⁇ m to 20 ⁇ m, or 0.01 ⁇ m to 15 ⁇ m, and the average pore diameter of the holes present on the B surface is 20 ⁇ m to 100 ⁇ m, 30 ⁇ m to 100 ⁇ m, 40 ⁇ m to 100 ⁇ m, 50 ⁇ m to 100 ⁇ m, or 60 ⁇ m to 100 ⁇ m It is.
- the A surface of the polyimide porous membrane has a mesh structure having small holes with an average pore diameter of 15 ⁇ m or less, for example, 0.01 ⁇ m to 15 ⁇ m, and the B surface has a large hole structure with an average pore diameter of 20 ⁇ m or more, for example, 20 ⁇ m to 100 ⁇ m. .
- the total film thickness of the polyimide porous membrane used in the present invention can be measured with a contact-type thickness meter.
- the average pore diameter on the surface of the polyimide porous membrane was determined by measuring the pore area of 200 or more apertures from the scanning electron micrograph on the surface of the porous membrane, and calculating the pore size according to the following formula (1) from the average value of the pore area.
- the average diameter when the shape is assumed to be a perfect circle can be obtained by calculation. (In the formula, Sa means the average value of the pore area.)
- the porosity of the polyimide porous membrane used in the present invention can be determined from the mass per unit area according to the following formula (2) by measuring the thickness and mass of the porous film cut into a predetermined size.
- S is the area of the porous film
- d is the total film thickness
- w is the measured mass
- D is the polyimide density.
- the polyimide density is 1.34 g / cm 3 ).
- polyimide porous membrane described in International Publication WO2010 / 038873, JP2011-219585, or JP2011-219586 can also be used in the method of the present invention.
- Cells seeded on the surface of the polyimide porous membrane can stably grow and proliferate on the surface and / or inside of the membrane.
- Cells can take a variety of different forms depending on where they grow and proliferate in the membrane.
- the cells may proliferate while changing the shape while moving on and inside the polyimide porous membrane.
- the polyimide porous membrane to which the cells are applied in the method of the present invention does not contain cells other than the cells to be applied, that is, is sterilized.
- the method of the present invention preferably includes a step of pre-sterilizing the polyimide porous membrane.
- the polyimide porous membrane is extremely excellent in heat resistance, is lightweight, can be freely selected in shape and size, and is easy to sterilize. Arbitrary sterilization treatments such as dry heat sterilization, steam sterilization, sterilization with a disinfectant such as ethanol, and electromagnetic wave sterilization such as ultraviolet rays and gamma rays are possible.
- FIG. 4 A model diagram of cell culture using a polyimide porous membrane is shown in FIG.
- a large amount of cells can be cultured while the amount of the medium used for cell culture is greatly reduced as compared with the conventional method.
- the total volume of the cell culture medium contained in the cell culture container can be significantly reduced with respect to the total volume of the polyimide porous membrane including the cell survival area.
- the volume occupied by the porous porous membrane not containing cells in the space including the volume of the internal gap is referred to as “apparent polyimide porous membrane volume” (state on the left in FIG. 1). Then, when the cells are applied to the polyimide porous membrane, and the cells are supported on the surface and inside of the polyimide porous membrane, the polyimide porous membrane, the cells, and the medium infiltrated inside the polyimide porous membrane are entirely space.
- the volume occupying the inside is referred to as “polyimide porous membrane volume including cell survival region” (right state in FIG. 1).
- the polyimide porous membrane volume including the cell survival area is apparently about 50% larger than the polyimide porous membrane volume at the maximum.
- a plurality of polyimide porous membranes can be accommodated and cultured in one cell culture container. In that case, the cell survival area for each of the plurality of polyimide porous membranes carrying cells.
- the total volume of the polyimide porous membrane containing s is sometimes simply referred to as “the total volume of the polyimide porous membrane including the cell survival region”.
- the cells can be cultured well over a long period of time even under conditions where the total volume of the cell culture medium contained in the cell culture vessel is 1000 times or less than the total volume of the polyimide porous membrane including the cell survival area. . Furthermore, even when the total volume of the cell culture medium contained in the cell culture container is 100 times or less than the total volume of the polyimide porous membrane including the cell survival area, the cells can be cultured well over a long period of time. . And even if the total volume of the cell culture medium contained in the cell culture container is 10 times or less than the total volume of the polyimide porous membrane including the cell survival area, the cells can be cultured well over a long period of time. .
- the space (container) for cell culture can be miniaturized to the limit as compared with the conventional cell culture apparatus for performing two-dimensional culture. Moreover, when it is desired to increase the number of cells to be cultured, it is possible to increase the volume of cell culture flexibly by a simple operation such as increasing the number of polyimide porous membranes to be laminated. If it is a cell culture apparatus provided with the polyimide porous membrane used for this invention, it becomes possible to isolate
- the space (container) in which the cell culture medium is stored may be enlarged or reduced according to the purpose, or may be a replaceable container, and is not particularly limited. This concept of integration is also important in freezing, and a large amount of cells can be frozen and stored in a very small space.
- a method for measuring the number of cells during or after the culture various known methods can be used.
- a method of measuring the number of cells contained in a cell culture vessel after culturing using a polyimide porous membrane as if all the cells are uniformly dispersed in the cell culture medium contained in the cell culture vessel Any known method can be used as appropriate.
- a cell number counting method using CCK8 can be suitably used.
- Cell Counting Kit 8; a solution reagent manufactured by Dojindo Laboratories (hereinafter referred to as “CCK8”) was used to measure the number of cells in a normal culture without using a polyimide porous membrane. The correlation coefficient with the actual cell number is obtained.
- the applied polyimide porous membrane was transferred to a medium containing CCK8, stored in an incubator for 1 to 3 hours, the supernatant was extracted, and the absorbance was measured at a wavelength of 480 nm. The number of cells is calculated from the obtained correlation coefficient.
- the cell culture system and culture conditions can be appropriately determined according to the cell type and the like. Culture methods suitable for animal cells, plant cells, and bacterial cells are known, and those skilled in the art can cultivate cells applied to the polyimide porous membrane using any known method. A cell culture medium can also be suitably prepared according to the kind of cell.
- Animal cell culture methods and cell culture media are described, for example, in the cell culture media catalog of Lonza.
- Plant cell culture methods and cell culture media are described in, for example, the plant tissue culture series from WAKO.
- Bacterial cell culture methods and cell culture media are described in, for example, the general bacterial culture catalog of BD.
- the cell culture medium used in the method of the present invention may be in any form such as a liquid medium, a semi-solid medium, a solid medium and the like.
- the medium may be brought into contact with the porous polyimide membrane supporting the cells by spraying a liquid medium in the form of droplets into the cell culture container.
- cell culture using a polyimide porous membrane it can be coexisted with other floating culture carriers such as microcarriers and cellulose sponges.
- the shape, scale, etc. of the system used for culture are not particularly limited, and it can be suitably used from petri dishes for cell culture, flasks, plastic bags, test tubes to large tanks.
- a cell culture dish manufactured by BD Falcon, a Nunc cell factory manufactured by Thermo Scientific, and the like are included.
- a polyimide porous membrane in the present invention it has become possible to culture cells in a state similar to suspension culture using a suspension culture apparatus even for cells that were not inherently capable of suspension culture.
- a spinner flask manufactured by Corning, rotary culture, or the like can be used as an apparatus for suspension culture.
- hollow fiber culture such as FiberCell (registered trademark) System manufactured by VERITAS can be used as an environment in which similar functions can be realized.
- the culture in the method of the present invention is a type in which the polyimide porous membrane sheet is exposed to the air using a continuous circulation or open type device that continuously adds and recovers the medium on the polyimide porous membrane. It is also possible to execute with.
- cells may be cultured in a system in which the cell culture medium is supplied into the cell culture container continuously or intermittently from a cell culture medium supply means installed outside the cell culture container.
- the cell culture medium can be a system in which the cell culture medium is circulated between the cell culture medium supply means and the cell culture container.
- the system When cell culture is performed in a system in which the cell culture medium is continuously or intermittently supplied from the cell culture medium supply means installed outside the cell culture container, the system is a cell culture container. It may be a cell culture apparatus including a culture unit and a culture medium supply unit which is a cell culture medium supply means, wherein the culture unit is a culture unit containing one or more polyimide porous membranes for supporting cells.
- the culture medium supply unit includes a culture medium storage container, a culture medium supply line, and a liquid feed pump that continuously or intermittently feeds the culture medium via the culture medium supply line, where the first end of the culture medium supply line is
- the cell culture device may be a culture medium supply unit, which is in contact with the culture medium in the culture medium storage container and the second end of the culture medium supply line communicates with the culture unit via the culture medium supply port of the culture unit.
- the culture unit may be a culture unit that does not include an air supply port, an air discharge port, and an oxygen exchange membrane, and further includes an air supply port and an air discharge port, or an oxygen exchange membrane. It may be a culture unit. Even if the culture unit does not include an air supply port, an air discharge port, and an oxygen exchange membrane, oxygen and the like necessary for cell culture are sufficiently supplied to the cells through the medium. Furthermore, in the cell culture apparatus, the culture unit further includes a medium discharge line, wherein the first end of the medium discharge line is connected to the medium storage container, and the second end of the medium discharge line is the culture unit. The culture medium may be circulated between the culture medium supply unit and the culture unit by communicating with the culture unit via the culture medium outlet.
- FIG. 2 shows an example of a cell culture apparatus which is an example of the cell culture system, but the cell culture system that can be used for the purpose of the present invention is not limited to this.
- the present invention also relates to a cell culture device for use in the culture method of the present invention, including a polyimide porous membrane.
- the polyimide porous membrane may be used in a fixed state, or may be used suspended in the cell culture medium, or may be placed in the medium or exposed from the medium. Also good.
- two or more polyimide porous membranes may be laminated vertically or horizontally. Laminated aggregates and aggregates may be placed in the medium or exposed from the medium.
- the cell culture device of the present invention may take any form as long as it includes a polyimide porous membrane.
- any of the cell culture systems used in the above-described long-term culture method of the present invention can be used as the cell culture apparatus of the present invention.
- Kit for use in cell culture method The present invention further relates to a kit for use in a cell culture method, comprising a polyimide porous membrane.
- the kit of the present invention may appropriately contain components necessary for cell culture in addition to the polyimide porous membrane.
- Examples include cells to be applied to polyimide porous membranes, cell culture media, continuous media supply devices, continuous media circulation devices, scaffolds or modules that support polyimide porous membranes, cell culture devices, instruction manuals for kits, etc. It is.
- a sterilized polyimide porous membrane is stored alone or in a plurality of sheets in a transparent pouch, and a package containing a form that can be used for cell culture as it is, or the same
- a sterilizing liquid is enclosed in a pouch together with a polyimide porous membrane, and includes an integrated membrane / liquid kit that enables efficient suction seeding.
- the present invention also relates to the use of a polyimide porous membrane for a long-term culture method of cells.
- the present invention also relates to the use of the above-described cell culture apparatus for a long-term cell culture method.
- the present invention is a method for cryopreserving cells, (1) a step of supporting cells on a polyimide porous membrane; (2) a step of freezing cells carried on the polyimide porous membrane by placing the polyimide porous membrane carrying cells on the condition that the cells are frozen; and (3) a polyimide porous membrane carrying cells.
- the present invention relates to a method comprising a step of storing a membrane under conditions that keep it frozen. According to the present invention, it is possible to freeze a polyimide porous membrane in a state where cells cultured at a high density are supported, which is overwhelming as compared with a conventional method for freezing cells in a non-adhered state. It is possible to freeze cells in a high density state.
- Cells The types of cells that can be used in the method of the present invention are not particularly limited, and any of the cells described above can be used.
- Step of supporting cells on polyimide porous membrane includes the step of supporting cells on a polyimide porous membrane. Although any method can be used as a method for supporting cells on the polyimide porous membrane, for example, the following method can be used.
- A an embodiment comprising a step of seeding cells on the surface of the polyimide porous membrane;
- B Place a cell suspension on the dried surface of the polyimide porous membrane, Leave or move the polyimide porous membrane to promote fluid outflow, or stimulate a portion of the surface to draw cell suspension into the membrane; and The cells in the cell suspension are retained in the membrane, and the water flows out.
- An embodiment comprising steps; and
- C Wet one side or both sides of the polyimide porous membrane with a cell culture medium or a sterilized liquid, Loading the wet polyimide porous membrane with a cell suspension; and The cells in the cell suspension are retained in the membrane, and the water flows out.
- a mode comprising the steps.
- the cells seeded on the surface of the polyimide porous membrane adhere to the polyimide porous membrane and enter the inside of the pore.
- the cells spontaneously adhere to the polyimide porous membrane without any physical or chemical force applied from the outside.
- Cells seeded on the surface of the polyimide porous membrane can grow and proliferate stably on the surface and / or inside of the membrane. Cells can take a variety of different forms depending on the location of the membrane in which they grow and multiply.
- another polyimide porous membrane not supporting cells By applying and culturing another polyimide porous membrane not supporting cells together with the polyimide porous membrane supporting cells to the same cell culture medium, another one or more polyimides Cells may be supported on the porous membrane. At that time, the polyimide porous membrane supporting the cells and one or more polyimide porous membranes not supporting the cells may be brought into contact with each other by lamination or the like, or simply in the same cell culture medium. You may install in.
- a polyimide porous membrane can be immersed in a cell cryopreservation solution, frozen under a first low temperature condition, and then transferred to a second low temperature condition lower than the first low temperature condition for freezing. it can.
- a first low temperature condition for example, a condition of about minus 20 to 25 ° C. can be used
- a condition of about minus 80 to 90 ° C. can be used.
- the step of placing the first low temperature condition may be omitted.
- the transition to a second low temperature condition that is lower than the first low temperature condition may take any form of linear, stepwise, curvilinear, or immediate.
- the apparatus that provides the first low temperature condition and the apparatus that provides the second low temperature condition may be separate or the same. In the case where the apparatus that provides the first low-temperature condition and the apparatus that provides the second low-temperature condition are separate, for example, the apparatus that provides the first low-temperature condition is a normal freezer, Although the case where the apparatus which provides low temperature conditions of this is a deep freezer is mentioned, it is not limited to this.
- a program freezer that can reduce the temperature at a constant rate for example, a program freezer manufactured by Neppagene Corporation) Etc.
- cell cryopreservation solution known ones can be used as appropriate.
- a cell culture solution added with DMSO to about 5% to 20% a cell culture solution added with glycerol to about 5% to 20%, and a catalog made by ZENOAQ
- commercially available cell cryopreservation solutions such as various cell bankers can be suitably used.
- the step of storing the polyimide porous membrane supporting the cells under the condition of keeping the frozen state is the step of storing the polyimide porous membrane supporting the cells under the condition of maintaining the frozen state as described above.
- the condition for maintaining the frozen state include a condition for storing as it is under the second low temperature condition and a condition for storing under a third low temperature condition that is lower than the second low temperature condition.
- An example of the third low temperature condition is, for example, a case where it is placed in liquid nitrogen, but is not limited thereto.
- the method of the present invention whether it is a floating cell or an adherent cell, it can be cryopreserved well regardless of the type of cell.
- the suspension cells are cryopreserved by the method of the present invention, the cells are stored and stored in the three-dimensional structure of the polyimide porous membrane while maintaining the spherical shape.
- the adherent cells are cryopreserved by the method of the present invention, the three-dimensional structure of the polyimide porous membrane remains in the same non-spherical state as when grown and propagated in the polyimide porous membrane. Cells are stored and stored.
- the polyimide porous membrane supporting cells in the method of the present invention does not contain cells other than supporting cells, that is, is sterilized.
- the method of the present invention preferably includes a step of pre-sterilizing the polyimide porous membrane.
- the polyimide porous membrane is extremely excellent in heat resistance, is lightweight, can be freely selected in shape and size, and is easy to sterilize. Arbitrary sterilization treatments such as dry heat sterilization, steam sterilization, sterilization with a disinfectant such as ethanol, and electromagnetic wave sterilization such as ultraviolet rays and gamma rays are possible.
- the polyimide porous membrane carrying cells can be thawed after cryopreservation.
- a method for thawing the cells carried on the polyimide porous membrane a method can be mentioned in which a cryopreserved polyimide porous membrane is heated from the outside of the storage container and thawed.
- a heating method a method such as immersing the container in constant temperature hot water of about 37 ° C. can be appropriately used.
- Step of culturing cells by applying the polyimide porous membrane after thawing the cells to the cell culture medium by applying the polyimide porous membrane after thawing the cells as it is to the cell culture medium, the thawed cells Can be cultured on the same polyimide porous membrane.
- the cells may be cultured until the cultured cells proliferate outside the polyimide porous membrane.
- suspension cells those transferred from the polyimide porous membrane into the cell culture medium can be grown in the medium as they are.
- adherent cells many remain in or on the polyimide porous membrane, but some cells must be transferred to a culture vessel in contact with the polyimide porous membrane or another culture carrier that does not carry cells. Can do.
- the polyimide porous membrane in which the cells are thawed and another polyimide porous membrane in which the cells are newly supported under the condition that the cells are frozen, the polyimide porous membrane is obtained.
- the supported cells can be frozen and stored.
- the steps of freezing, storing, thawing and culturing as described above can be repeated multiple times.
- it is possible to freeze and store a polyimide porous membrane in a state where a large amount of cells are supported. Etc.).
- the cells can be cultured, frozen, stored, thawed, and re-cultured while the cells are adhered to the polyimide porous membrane without going through a pre-culturing step after thawing that is conventionally required. Therefore, for example, if a large amount of polyimide porous material frozen in a state of supporting cells is prepared, a large amount of cells can be used at a desired time without going through an expansion culture step.
- the present invention also relates to a polyimide porous membrane for cryopreservation of cells.
- the polyimide porous membrane for cryopreservation of cells of the present invention may be a polyimide porous membrane for the above-described cell cryopreservation method.
- Kit This invention also relates to the kit for the cryopreservation of the cell containing a polyimide porous membrane.
- the kit of the present invention can appropriately contain components necessary for cell culture in addition to the polyimide porous membrane.
- a polyimide porous membrane, a cell cryopreservation solution, a freezing tube, a freezing cane, an instruction manual for the kit, and the like are included.
- a sterilized polyimide porous membrane is stored in a transparent pouch alone or in a plurality of sheets, and a package containing a form that can be used for cell freezing as it is, or the same
- a cell cryopreservation solution is enclosed in a pouch together with a polyimide porous membrane, and includes a membrane / liquid integrated kit that can be used quickly.
- polyimide porous membrane refers to a polyimide porous membrane having a total film thickness of 25 ⁇ m and a porosity of 73%.
- the polyimide porous membrane had two different surface layers (A surface and B surface) and a macrovoid layer sandwiched between the two surface layers.
- the average hole diameter of the holes existing on the A surface was 6 ⁇ m
- the average hole diameter of the holes existing on the B surface was 46 ⁇ m.
- the polyimide porous membrane used in the following examples is composed of 3,3 ′, 4,4′-biphenyltetracarboxylic dianhydride (s-BPDA), which is a tetracarboxylic acid component, and a diamine component.
- s-BPDA 4,4′-biphenyltetracarboxylic dianhydride
- IMDM, -06465) ⁇ 3.5cm petri dish (Falcon cat. 353001) ⁇ Cell Counting Kit 8 (Dojindo Laboratories Co., Ltd. CCK8 CK04) -Cryotube (Thermo Fisher Scientific 1.8ml cat. 377267) 2 cm ⁇ 2 cm sterilized square container (Thermo Fisher Scientific cat. 103) ⁇ Cell banker (Nippon Zenyaku Kogyo Co., Ltd. CEllBANKER 1 Plus cat. CB021) -Microscope name, used image software name (Carl Zeiss LSM 700 software ZEN
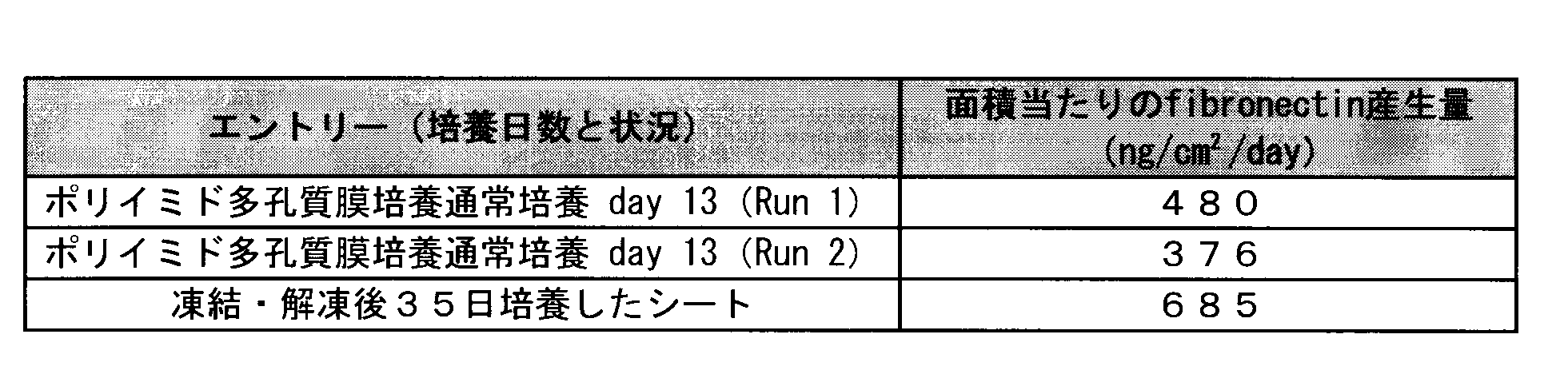
- the cell-grown sheet is cut into three vertically long sheets, stored in a medium for 20 hours at 5 ° C., the polyimide porous membrane is taken out and placed in a cryotube to which 1 ml of a cell banker is added. After storage at 24 ° C. for 24 hours, it was further stored at ⁇ 80 ° C. for 24 hours and transferred to liquid nitrogen. After 20 days, the tube was heated to 37 ° C. to melt the contents and left in the incubator for 16 hours. When the number of cells was measured using CCK8, the number of cells on the sheet was 4.6 ⁇ 10 6 .
- the sheet in which the cells are grown is cut into three vertically long sheets, stored in a medium for 8 hours at 5 ° C., and then the polyimide porous membrane is taken out and placed in a cryotube to which 1 ml of a cell banker is added. Stored at 24 ° C. for 24 hours and transferred into liquid nitrogen. Two days later, the tube was heated to 37 ° C to thaw the contents and transferred to the culture conditions in the incubator. When the specific activity by absorbance was measured using CCK8 after 3 days and 5 days, the specific activities on the 3rd and 5th days were 36% and 64%.
- Human skin fibroblast long-term culture Long-term culture using human dermal fibroblast polyimide porous membrane 0.5 cm of 2% FBS-containing cell culture medium is added to a sterilized square container of 2 cm ⁇ 2 cm, and sterilized 1.4 cm square polyimide porous membrane Were immersed with the A-side of the mesh structure facing up. 4 ⁇ 10 4 human skin fibroblast suspensions were added per sheet, the medium was changed twice a week, and cell culture was continuously performed. After 231 days, 261 days, 294 days, 324 days, 365 days, 401 days, and 471 days, the number of cells was measured using CCK8, and the growth behavior was observed. The results are shown in FIG. A stable number of viable cells was confirmed throughout the culture period.
- the cells were moved into a CO 2 incubator and the cells were continuously cultured by changing the medium twice a week. After 7 days of culture, each sheet was divided into one sheet and the culture was continued as a single sheet. After 7 days, 10 days, 16 days, 21 days, 28 days, 42 days and 56 days, the number of cells was measured using CCK8, and the original sheet and the empty polyimide porous membrane grounded later were The growth behavior was observed using the staining method with CCK8. From the polyimide porous membrane in which human skin fibroblasts were cultured for a long period of time, it was observed that the cells efficiently migrated to the empty polyimide porous membrane and continuously proliferated. The results are shown in FIG.
- Cell culture using CHO DP-12 cells freezing and substance production 0.5 ml of cell culture medium (2% FBS, IMDM, Wako Pure Chemical Industries, Ltd.) was added to a 2 cm ⁇ 2 cm sterilized square container and sterilized 1 .4 cm square square polyimide porous membranes were each immersed with the A side of the mesh structure facing upward. 4 ⁇ 10 4 human anti-IL-8 producing CHO DP-12 cell suspensions per sheet were each added to the sheet in the medium, and the medium was changed twice a week. Conducted continuously. After culturing the cells for 78 days, the number of cells was measured using CCK8.
- the four sheets were transferred to four frozen storage bags so as to form one sheet per bag under sterile conditions, and 3 ml of cell bunker was added to each bag.
- the sample was frozen at ⁇ 80 ° C. under two conditions (1 ° C. every minute or 1 ° C. every 10 minutes) in a program freezer, stored at ⁇ 80 ° C. for 24 hours, and transferred to liquid nitrogen.
- the bag was heated to 37 ° C. to melt the contents, and each sheet was moved to four 10 cm 2 petri dishes filled with 2 ml of the previously prepared medium and left in the incubator for 24 hours. Thereafter, the sheet was transferred to a sterilized square container of 2 cm ⁇ 2 cm, and 1 ml of cell culture medium was added and further cultured for 3 days. After 24 hours, 2 days, 3 days, and 4 days, the number of cells was measured using CCK8. The results are shown in FIG.
- the non-frozen sheet 2 sheets was continued for sowing and cultivation simultaneously also the freeze sheet 4
- the freeze sheet 4 Like the cell culture placed in a 10 cm 2 dish one by one, in addition 2ml cell culture medium, respectively, 5% CO 2, 37 Incubation was performed at 0 ° C. for 24 hours, and the culture supernatant was collected. The amount of anti-human IL-8 antibody contained in the collected supernatant was quantified by ELISA. As shown in Table 2, no change in anti-IL-8 production due to freezing was observed. The results are shown in Table 2.
- Freezing human skin fibroblasts and production of substances Add 0.5 ml of cell culture medium to a sterilized square container of 2 cm x 2 cm, and place a sterilized 1.4 cm square square polyimide porous membrane on the A side of the mesh structure. Each was immersed. 4 ⁇ 10 4 human skin fibroblast suspensions per sheet were added onto the medium sheet, and culture was started in a CO 2 incubator. Cell culture was continuously carried out by changing the medium twice a week. After 49 days of cell culture, the number of cells was measured using CCK8 and found to be 9.1 ⁇ 10 4 cells.
- the sheet on which the cells are grown is taken out of the medium, transferred to a pre-prepared cryopreservation bag containing 3 ml of a cell banker, lowered by 1 ° C. every 10 minutes with a program freezer, frozen at ⁇ 80 ° C., and then ⁇ It preserve
- the bag was heated to 37 ° C. to melt the contents, and only the sheet was transferred to a 10 cm 2 petri dish filled with 2 ml of the medium prepared in advance, and the culture was continued in the CO 2 incubator.
- the medium was exchanged twice a week.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical & Material Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Organic Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Genetics & Genomics (AREA)
- Biotechnology (AREA)
- Biomedical Technology (AREA)
- General Health & Medical Sciences (AREA)
- Microbiology (AREA)
- Biochemistry (AREA)
- General Engineering & Computer Science (AREA)
- Medicinal Chemistry (AREA)
- Dentistry (AREA)
- Environmental Sciences (AREA)
- Cell Biology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Polymers & Plastics (AREA)
- Materials Engineering (AREA)
- Virology (AREA)
- Tropical Medicine & Parasitology (AREA)
- Sustainable Development (AREA)
- Mycology (AREA)
- Immunology (AREA)
- Urology & Nephrology (AREA)
- Manufacturing & Machinery (AREA)
- Botany (AREA)
- Rheumatology (AREA)
- Apparatus Associated With Microorganisms And Enzymes (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Mechanical Engineering (AREA)
- Manufacture Of Porous Articles, And Recovery And Treatment Of Waste Products (AREA)
- Dispersion Chemistry (AREA)
Abstract
Description
細胞は生体内では一般に三次元的な集団として存在するが、古典的な平面培養では、細胞が容器に張り付く形で単層状に培養される。付着細胞の培養においては、古くより、様々な培養法がシャーレ等により開発されている。このシャーレ等による培養を行った場合、培養細胞は増殖を続け、“コンフルエント”と呼ばれるこれ以上増殖出来ない状況になり、増殖が停止する。細胞種にもよるが、継代せずにこのコンフルエントな状態を継続した場合、ある程度の期間の後に自発的に剥離が始まり、細胞が継代出来なくなることも、多くの細胞で見出されている。
ポリイミドとは、繰り返し単位にイミド結合を含む高分子の総称である。芳香族ポリイミドは、芳香族化合物が直接イミド結合で連結された高分子を意味する。芳香族ポリイミドは芳香族と芳香族とがイミド結合を介して共役構造を持つため、剛直で強固な分子構造を持ち、かつ、イミド結合が強い分子間力を持つために非常に高いレベルの熱的、機械的、化学的性質を有する。
限定されるわけではないが、本発明は好ましくは以下の態様を含む。
[態様1]
細胞の長期培養方法であって、
(1)細胞をポリイミド多孔質膜に適用することと、
(2)細胞を適用したポリイミド多孔質膜を細胞培養培地に適用し、30日以上細胞を培養することと
を含む、方法。
[態様2]
工程(2)において、60日以上細胞を培養する、態様1に記載の方法。
[態様3]
工程(2)において、120日以上細胞を培養する、態様1に記載の方法。
[態様4]
2以上のポリイミド多孔質膜を、上下又は左右に細胞培養培地中に積層して用いる、態様1~3のいずれか一項に記載の方法。
[態様5]
ポリイミド多孔質膜を
i)折り畳んで、
ii)ロール状に巻き込んで、
iii)シートもしくは小片を糸状の構造体で連結させて、あるいは、
iv)縄状に結んで
細胞培養容器中の細胞培養培地中で浮遊もしくは固定させて用いる、態様1~4のいずれか一項に記載の方法。
[態様6]
工程(2)における培養において、ポリイミド多孔質膜の一部分又は全体が、細胞培養培地の液相と接触していない、態様1~5のいずれか一項に記載の方法。
[態様7]
工程(2)における培養において、細胞培養容器中に含まれる細胞培養培地の総体積が、細胞生存域を含むポリイミド多孔質膜体積の総和の10000倍又はそれより少ない、態様1~6のいずれか一項に記載の方法。
[態様8]
工程(2)における培養において、細胞培養容器中に含まれる細胞培養培地の総体積が、細胞生存域を含むポリイミド多孔質膜体積の総和の100倍又はそれより少ない、態様1~7のいずれか一項に記載の方法。
[態様9]
工程(2)における培養が、細胞培養容器外に設置された細胞培養培地供給手段から連続的又は間歇的に細胞培養培地が細胞培養容器中に供給される系で行われる、態様1~8のいずれか一項に記載の方法。
[態様10]
細胞培養培地が細胞培養培地供給手段と細胞培養容器との間を循環する、態様9に記載の方法。
[態様11]
態様9又は10に記載の方法であって、前記系が、細胞培養容器である培養ユニットと細胞培養培地供給手段である培地供給ユニットとを含む細胞培養装置であり、ここで
培養ユニットは細胞を担持するための1又は複数のポリイミド多孔質膜を収容する培養ユニットであって、培地供給口および培地排出口を備えた培養ユニットであり、
培地供給ユニットは培地収納容器と、培地供給ラインと、培地供給ラインを介して連続的又は間歇的に培地を送液する送液ポンプとを備え、ここで培地供給ラインの第一の端部は培地収納容器内の培地に接触し、培地供給ラインの第二の端部は培養ユニットの培地供給口を介して培養ユニット内に連通している、培地供給ユニットである、
方法。
[態様12]
培養ユニットが空気供給口、空気排出口、及び酸素交換膜を備えない培養ユニットである、態様13に記載の方法。
[態様13]
培養ユニットが培地排出ラインをさらに備え、ここで培地排出ラインの第一の端部は培地収納容器に接続し、培地排出ラインの第二の端部は培養ユニットの培地排出口を介して培養ユニット内に連通し、培地が培地供給ユニットと培養ユニットとを循環可能である、態様11又は12に記載の方法。
[態様14]
細胞が、動物細胞、昆虫細胞、植物細胞、酵母菌及び細菌からなる群から選択される、態様1~13のいずれかに記載の方法。
[態様15]
動物細胞が、脊椎動物門に属する動物由来の細胞である、態様14に記載の方法。
[態様16]
細胞が、CHO細胞、CHO-K1細胞株、CHO DP-12細胞株、CHO細胞関連株、Vero細胞、及びMDCK細胞からなる群より選択される、態様15に記載の方法。
[態様17]
ポリイミド多孔質膜が、テトラカルボン酸二無水物とジアミンとから得られるポリイミドを含む、ポリイミド多孔質膜である、請求項1~16のいずれかに記載の方法。
[態様18]
ポリイミド多孔質膜が、テトラカルボン酸二無水物とジアミンとから得られるポリアミック酸溶液と着色前駆体とを含むポリアミック酸溶液組成物を成形した後、250℃以上で熱処理する事により得られる着色したポリイミド多孔質膜である、請求項17に記載の方法。
[態様19]
ポリイミド多孔質膜を含む、請求項1~18のいずれかに記載の方法に使用するための細胞培養装置。
[態様20]
2以上のポリイミド多孔質膜が、上下又は左右に積層している、請求項19に記載の細胞培養装置。
[態様21]
ポリイミド多孔質膜を含む、請求項1~13のいずれかに記載の方法に使用するためのキット。
[態様22]
細胞の長期培養方法のための、ポリイミド多孔質膜の使用。
[態様23]
細胞の凍結保存方法であって、
(1)細胞をポリイミド多孔質膜に担持させる工程、
(2)細胞を担持させたポリイミド多孔質膜を、細胞が凍結する条件下に置くことで、ポリイミド多孔質膜に担持された細胞を凍結する工程、及び
(3)細胞を担持させたポリイミド多孔質膜を、凍結状態を保つ条件で保存する工程
を含む、方法。
[態様24]
工程(1)において、細胞をポリイミド多孔質膜に播種することにより、細胞をポリイミド多孔質膜に担持させる、態様23に記載の方法。
[態様25]
工程(1)において、細胞をポリイミド多孔質膜に播種して培養することにより、細胞をポリイミド多孔質膜に担持させる、態様23に記載の方法。
[態様26]
工程(3)に続いて、
(4)細胞を担持させたポリイミド多孔質膜を、細胞が解凍する条件下に置くことで、ポリイミド多孔質膜に担持された細胞を解凍する工程
をさらに含む、態様23~25のいずれか一項に記載の方法。
[態様27]
工程(4)に続いて、
(5)細胞を解凍したポリイミド多孔質膜を細胞培養培地に適用し、細胞を培養する工程
をさらに含む、態様26に記載の方法。
[態様28]
工程(5)において、培養した細胞がポリイミド多孔質膜外においても増殖するまで細胞を培養する、態様27に記載の方法。
[態様29]
工程(5)において、細胞を解凍したポリイミド多孔質膜と共に、細胞を担持させていない別の1又は複数のポリイミド多孔質膜を細胞培養培地に適用し、培養することにより、別の1又は複数のポリイミド多孔質膜に細胞を担持させる、態様27又は28に記載の方法。
[態様30]
工程(5)に続いて、
(6)細胞を担持させたポリイミド多孔質膜の一部又はすべてを、細胞が凍結する条件下に置くことで、ポリイミド多孔質膜に担持された細胞を凍結する工程、及び
(7)細胞を担持させたポリイミド多孔質膜を、凍結状態を保つ条件で保存する工程
をさらに含む、態様27~29のいずれか一項に記載の方法。
[態様31]
工程(1)~(7)を複数回繰り返す、態様30に記載の方法。
[態様32]
細胞が、動物細胞、昆虫細胞、植物細胞、酵母菌及び細菌からなる群から選択される、態様23~31のいずれか一項に記載の方法。
[態様33]
動物細胞が、脊椎動物門に属する動物由来の細胞である、態様32に記載の方法。
[態様34]
細胞が付着性細胞である、態様32又は33に記載の方法。
[態様35]
細胞が浮遊性細胞である、態様32又は33に記載の方法。
[態様36]
ポリイミド多孔質膜が、テトラカルボン酸二無水物とジアミンとから得られるポリイミドを含む、ポリイミド多孔質膜である、態様23~35のいずれか一項に記載の方法。
[態様37]
ポリイミド多孔質膜が、テトラカルボン酸二無水物とジアミンとから得られるポリアミック酸溶液と着色前駆体とを含むポリアミック酸溶液組成物を成形した後、250℃以上で熱処理する事により得られる着色したポリイミド多孔質膜である、態様36に記載の方法。
[態様38]
ポリイミド多孔質膜が、2つの異なる表層面とマクロボイド層を有する多層構造のポリイミド多孔質膜である、態様36又は37に記載の方法。
[態様39]
細胞の凍結保存のためのポリイミド多孔質膜。
[態様40]
態様23~28のいずれか一項に記載の方法に用いるためのポリイミド多孔質膜。
[態様41]
ポリイミド多孔質膜を含む、態様23~38のいずれか一項に記載の方法に用いるためのキット。
[態様42]
態様23~38のいずれか一項に記載の方法のためのポリイミド多孔質膜の使用。
本発明は、細胞の長期培養方法に関する。なお、国際出願番号PCT/JP2014/070407の全内容を、参照により本明細書に援用する。
細胞のポリイミド多孔質膜への適用の具体的な工程は特に限定されない。本明細書に記載の工程、あるいは、細胞を膜状の担体に適用するのに適した任意の手法を採用することが可能である。限定されるわけではないが、本発明の方法において、細胞のポリイミド多孔質膜への適用は、例えば、以下のような態様を含む。
(B)前記ポリイミド多孔質膜の乾燥した表面に細胞縣濁液を載せ、
放置するか、あるいは前記ポリイミド多孔質膜を移動して液の流出を促進するか、あるいは表面の一部を刺激して、細胞縣濁液を前記膜に吸い込ませ、そして、
細胞縣濁液中の細胞を前記膜内に留め、水分は流出させる、
工程を含む、態様;並びに、
(C)前記ポリイミド多孔質膜の片面又は両面を、細胞培養液又は滅菌された液体で湿潤し、
前記湿潤したポリイミド多孔質膜に細胞縣濁液を装填し、そして、
細胞縣濁液中の細胞を前記膜内に留め、水分は流出させる、
工程を含む、態様。
本発明の長期培養方法により、細胞は従来のようにトリプシン処理等を行う継代操作を行う事なく、30日以上、60日以上、120日以上、200日以上、または300日以上の長期にわたって培養することができる。また、本発明の長期培養方法により、従来の平面培養で培養することができる期間以上、例えば、平面培養期間の1.5倍以上、2倍以上、2.5倍以上、3倍以上、3.5倍以上、4倍以上、4.5倍以上の期間、細胞を培養することができる。本発明によれば、シャーレ等の平面培養での長期間の細胞培養で発生する細胞の剥離や死滅等を生じる事なく、休止状態ではなく動的な生命を長期間維持する事ができる。また、本発明によれば、長期培養した細胞であってもセルバイアビリティ又は細胞の性質(例えば、分化誘導効率、細胞表面マーカーの発現量等)が、長期培養前の細胞と比較してほとんど変化しない。また、本発明によれば、細胞がポリイミド多孔質膜内において三次元的に増殖するため、従来の平面培養で見られるような培養領域の制限及び平面環境によりおこるコンタクトインヒビションが起こりにくいため、長期間、生育させる培養が可能となる。また、本発明によれば、細胞が接着したポリイミド多孔質膜に別のポリイミド多孔質膜を接触させることによって、細胞培養可能な空間を任意に増加することが可能であり、従来のようなトリプシン処理を伴った継代操作を行うことなく、コンタクトインヒビションが引き起こされるコンフルエント状態を回避しながら長期間、増殖させる培養が可能となる。また、本発明によれば、細胞を凍結等行うことなく、生きたまま長期間保存するという新たな保存方法までも提供するものである。
本発明の方法に利用し得る細胞の種類は特に限定されず、任意の細胞の増殖に利用可能である。
種子植物細胞が由来する植物は、単子葉植物、双子葉植物のいずれも含まれる。限定されるわけではないが、単子葉植物には、ラン科植物、イネ科植物(イネ、トウモロコシ、オオムギ、コムギ、ソルガム等)、カヤツリグサ科植物などが含まれる。双子葉植物には、キク亜綱、モクレン亜綱、バラ亜綱など多くの亜綱に属する植物が含まれる。
ポリイミドとは、繰り返し単位にイミド結合を含む高分子の総称であり、通常は、芳香族化合物が直接イミド結合で連結された芳香族ポリイミドを意味する。芳香族ポリイミドは芳香族と芳香族とがイミド結合を介して共役構造を持つため、剛直で強固な分子構造を持ち、かつ、イミド結合が強い分子間力を持つために非常に高いレベルの熱的、機械的、化学的性質を有する。
ポリアミック酸は、テトラカルボン酸成分とジアミン成分とを重合して得られる。ポリアミック酸は、熱イミド化又は化学イミド化することにより閉環してポリイミドとすることができるポリイミド前駆体である。
本発明において着色前駆体とは、250℃以上の熱処理により一部または全部が炭化して着色化物を生成する前駆体を意味する。
1)1,4-ジアミノベンゼン(パラフェニレンジアミン)、1,3-ジアミノベンゼン、2,4-ジアミノトルエン、2,6-ジアミノトルエンなどのベンゼン核1つのべンゼンジアミン;
2)4,4’-ジアミノジフェニルエーテル、3,4’-ジアミノジフェニルエーテルなどのジアミノジフェニルエーテル、4,4’-ジアミノジフェニルメタン、3,3’-ジメチル-4,4’-ジアミノビフェニル、2,2’-ジメチル-4,4’-ジアミノビフェニル、2,2’-ビス(トリフルオロメチル)-4,4’-ジアミノビフェニル、3,3’-ジメチル-4,4’-ジアミノジフェニルメタン、3,3’-ジカルボキシ-4,4’-ジアミノジフェニルメタン、3,3’,5,5’-テトラメチル-4,4’-ジアミノジフェニルメタン、ビス(4-アミノフェニル)スルフィド、4,4’-ジアミノベンズアニリド、3,3’-ジクロロベンジジン、3,3’-ジメチルベンジジン、2,2’-ジメチルベンジジン、3,3’-ジメトキシベンジジン、2,2’-ジメトキシベンジジン、3,3’-ジアミノジフェニルエーテル、3,4’-ジアミノジフェニルエーテル、4,4’-ジアミノジフェニルエーテル、3,3’-ジアミノジフェニルスルフィド、3,4’-ジアミノジフェニルスルフィド、4,4’-ジアミノジフェニルスルフィド、3,3’-ジアミノジフェニルスルホン、3,4’-ジアミノジフェニルスルホン、4,4’-ジアミノジフェニルスルホン、3,3’-ジアミノベンゾフェノン、3,3’-ジアミノ-4,4’-ジクロロベンゾフェノン、3,3’-ジアミノ-4,4’-ジメトキシベンゾフェノン、3,3’-ジアミノジフェニルメタン、3,4’-ジアミノジフェニルメタン、4,4’-ジアミノジフェニルメタン、2,2-ビス(3-アミノフェニル)プロパン、2,2-ビス(4-アミノフェニル)プロパン、2,2-ビス(3-アミノフェニル)-1,1,1,3,3,3-ヘキサフルオロプロパン、2,2-ビス(4-アミノフェニル)-1,1,1,3,3,3-ヘキサフルオロプロパン、3,3’-ジアミノジフェニルスルホキシド、3,4’-ジアミノジフェニルスルホキシド、4,4’-ジアミノジフェニルスルホキシドなどのベンゼン核2つのジアミン;
3)1,3-ビス(3-アミノフェニル)ベンゼン、1,3-ビス(4-アミノフェニル)ベンゼン、1,4-ビス(3-アミノフェニル)ベンゼン、1,4-ビス(4-アミノフェニル)ベンゼン、1,3-ビス(4-アミノフェノキシ)ベンゼン、1,4-ビス(3-アミノフェノキシ)ベンゼン、1,4-ビス(4-アミノフェノキシ)ベンゼン、1,3-ビス(3-アミノフェノキシ)-4-トリフルオロメチルベンゼン、3,3’-ジアミノ-4-(4-フェニル)フェノキシベンゾフェノン、3,3’-ジアミノ-4,4’-ジ(4-フェニルフェノキシ)ベンゾフェノン、1,3-ビス(3-アミノフェニルスルフィド)ベンゼン、1,3-ビス(4-アミノフェニルスルフィド)ベンゼン、1,4-ビス(4-アミノフェニルスルフィド)ベンゼン、1,3-ビス(3-アミノフェニルスルホン)ベンゼン、1,3-ビス(4-アミノフェニルスルホン)ベンゼン、1,4-ビス(4-アミノフェニルスルホン)ベンゼン、1,3-ビス〔2-(4-アミノフェニル)イソプロピル〕ベンゼン、1,4-ビス〔2-(3-アミノフェニル)イソプロピル〕ベンゼン、1,4-ビス〔2-(4-アミノフェニル)イソプロピル〕ベンゼンなどのベンゼン核3つのジアミン;
4)3,3’-ビス(3-アミノフェノキシ)ビフェニル、3,3’-ビス(4-アミノフェノキシ)ビフェニル、4,4’-ビス(3-アミノフェノキシ)ビフェニル、4,4’-ビス(4-アミノフェノキシ)ビフェニル、ビス〔3-(3-アミノフェノキシ)フェニル〕エーテル、ビス〔3-(4-アミノフェノキシ)フェニル〕エーテル、ビス〔4-(3-アミノフェノキシ)フェニル〕エーテル、ビス〔4-(4-アミノフェノキシ)フェニル〕エーテル、ビス〔3-(3-アミノフェノキシ)フェニル〕ケトン、ビス〔3-(4-アミノフェノキシ)フェニル〕ケトン、ビス〔4-(3-アミノフェノキシ)フェニル〕ケトン、ビス〔4-(4-アミノフェノキシ)フェニル〕ケトン、ビス〔3-(3-アミノフェノキシ)フェニル〕スルフィド、ビス〔3-(4-アミノフェノキシ)フェニル〕スルフィド、ビス〔4-(3-アミノフェノキシ)フェニル〕スルフィド、ビス〔4-(4-アミノフェノキシ)フェニル〕スルフィド、ビス〔3-(3-アミノフェノキシ)フェニル〕スルホン、ビス〔3-(4-アミノフェノキシ)フェニル〕スルホン、ビス〔4-(3-アミノフェノキシ)フェニル〕スルホン、ビス〔4-(4-アミノフェノキシ)フェニル〕スルホン、ビス〔3-(3-アミノフェノキシ)フェニル〕メタン、ビス〔3-(4-アミノフェノキシ)フェニル〕メタン、ビス〔4-(3-アミノフェノキシ)フェニル〕メタン、ビス〔4-(4-アミノフェノキシ)フェニル〕メタン、2,2-ビス〔3-(3-アミノフェノキシ)フェニル〕プロパン、2,2-ビス〔3-(4-アミノフェノキシ)フェニル〕プロパン、2,2-ビス〔4-(3-アミノフェノキシ)フェニル〕プロパン、2,2-ビス〔4-(4-アミノフェノキシ)フェニル〕プロパン、2,2-ビス〔3-(3-アミノフェノキシ)フェニル〕-1,1,1,3,3,3-ヘキサフルオロプロパン、2,2-ビス〔3-(4-アミノフェノキシ)フェニル〕-1,1,1,3,3,3-ヘキサフルオロプロパン、2,2-ビス〔4-(3-アミノフェノキシ)フェニル〕-1,1,1,3,3,3-ヘキサフルオロプロパン、2,2-ビス〔4-(4-アミノフェノキシ)フェニル〕-1,1,1,3,3,3-ヘキサフルオロプロパンなどのベンゼン核4つのジアミン。
これらの中でも、芳香族ジアミン化合物が好ましく、3,3’-ジアミノジフェニルエーテル、3,4’-ジアミノジフェニルエーテル、4,4’-ジアミノジフェニルエーテル及びパラフェニレンジアミン、1,3-ビス(3-アミノフェニル)ベンゼン、1,3-ビス(4-アミノフェニル)ベンゼン、1,4-ビス(3-アミノフェニル)ベンゼン、1,4-ビス(4-アミノフェニル)ベンゼン、1,3-ビス(4-アミノフェノキシ)ベンゼン、1,4-ビス(3-アミノフェノキシ)ベンゼンを好適に用いることができる。特に、ベンゼンジアミン、ジアミノジフェニルエーテル及びビス(アミノフェノキシ)フェニルからなる群から選ばれる少なくとも一種のジアミンが好ましい。
(i)ビフェニルテトラカルボン酸単位及びピロメリット酸単位からなる群から選ばれる少なくとも一種のテトラカルボン酸単位と、芳香族ジアミン単位とからなる芳香族ポリイミド、
(ii)テトラカルボン酸単位と、ベンゼンジアミン単位、ジアミノジフェニルエーテル単位及びビス(アミノフェノキシ)フェニル単位からなる群から選ばれる少なくとも一種の芳香族ジアミン単位とからなる芳香族ポリイミド、
及び/又は、
(iii)ビフェニルテトラカルボン酸単位及びピロメリット酸単位からなる群から選ばれる少なくとも一種のテトラカルボン酸単位と、ベンゼンジアミン単位、ジアミノジフェニルエーテル単位及びビス(アミノフェノキシ)フェニル単位からなる群から選ばれる少なくとも一種の芳香族ジアミン単位とからなる芳香族ポリイミド。
ポリイミド多孔質膜表面の平均孔径は、多孔質膜表面の走査型電子顕微鏡写真より、200点以上の開孔部について孔面積を測定し、該孔面積の平均値から下式(1)に従って孔の形状が真円であるとした際の平均直径を計算より求めることができる。
ポリイミド多孔質膜を用いた細胞培養のモデル図を図1に示す。本発明の方法では、細胞培養に用いる培地の量を従来の方法よりも大幅に減らしつつ、大量の細胞を培養することが可能となる。たとえば、ポリイミド多孔質膜の一部分又は全体が、細胞培養培地の液相と接触していない状態であっても、大量の細胞を長期にわたって培養することができる。また、細胞生存域を含むポリイミド多孔質膜体積の総和に対して、細胞培養容器中に含まれる細胞培養培地の総体積を著しく減らすことも可能となる。
本発明の方法において、細胞の培養システム及び培養条件は、細胞の種類等に応じて適宜決定することができる。動物細胞、植物細胞、及び細菌の各細胞に適した培養方法が公知であり、当業者は任意の公知の方法を用いてポリイミド多孔質膜に適用した細胞を培養することができる。細胞培養培地も細胞の種類に応じて適宜調製することができる。
本発明の方法において、培養に用いるシステムの形状、規模などは特に限定されず、細胞培養用のシャーレ、フラスコ、プラスチックバッグ、試験管から大型のタンクまで適宜利用可能である。例えば、BD Falcon社製のセルカルチャーディッシュやサーモサイエンティフィック社製のNunc セルファクトリー等が含まれる。なお、本発明においてポリイミド多孔質膜を用いることにより、生来浮遊培養が可能でなかった細胞についても浮遊培養向け装置にて、浮遊培養類似状態での培養を行うことが可能になった。浮遊培養用の装置としては、例えば、コーニング社製のスピナーフラスコや回転培養等が使用可能である。また、同様の機能を実現出来る環境として、VERITAS社のFiberCell(登録商標)Systemの様な中空糸培養も使用することが可能である。
培養ユニットは細胞を担持するための1又は複数のポリイミド多孔質膜を収容する培養ユニットであって、培地供給口および培地排出口を備えた培養ユニットであり、
培地供給ユニットは培地収納容器と、培地供給ラインと、培地供給ラインを介して連続的又は間歇的に培地を送液する送液ポンプとを備え、ここで培地供給ラインの第一の端部は培地収納容器内の培地に接触し、培地供給ラインの第二の端部は培養ユニットの培地供給口を介して培養ユニット内に連通している、培地供給ユニットである
細胞培養装置であってよい。
本発明はまた、ポリイミド多孔質膜を含む、本発明の培養方法に使用するための細胞培養装置に関する。本発明の細胞培養装置において、ポリイミド多孔質膜は固定されて用いられてもよく、あるいは細胞培養培地中に浮遊して用いられてもよく、培地中に置かれても、培地から露出しても良い。細胞培養装置において、2以上のポリイミド多孔質膜が、上下又は左右に積層してもよい。積層された集合体や集積体は、培地中に置かれても培地から露出していてもかまわない。
本発明はさらに、ポリイミド多孔質膜を含む、細胞の培養方法に使用するためのキットに関する。
本発明は、細胞の長期培養方法のための、ポリイミド多孔質膜の使用にも関する。また、細胞の長期培養方法のための、上述した細胞培養装置の使用にも関する。
本発明は、細胞の凍結保存方法であって、
(1)細胞をポリイミド多孔質膜に担持させる工程、
(2)細胞を担持させたポリイミド多孔質膜を、細胞が凍結する条件下に置くことで、ポリイミド多孔質膜に担持された細胞を凍結する工程、及び
(3)細胞を担持させたポリイミド多孔質膜を、凍結状態を保つ条件で保存する工程
を含む、方法に関する。本発明によれば、高密度に培養された細胞を担持した状態でポリイミド多孔質膜を凍結することが可能であり、従来の非接着状態で凍結させる細胞の凍結方法と比較して、圧倒的に高密度の状態で細胞を凍結することが可能となる。
本発明の方法に利用し得る細胞の種類は特に限定されず、上述の任意の細胞が利用可能である。
本発明の方法は、細胞をポリイミド多孔質膜に担持させる工程を含む。
ポリイミド多孔質膜に細胞を担持させる方法としては任意の方法を用いることができるが、たとえば以下のような方法を用いることができる。
(B)前記ポリイミド多孔質膜の乾燥した表面に細胞縣濁液を載せ、
放置するか、あるいは前記ポリイミド多孔質膜を移動して液の流出を促進するか、あるいは表面の一部を刺激して、細胞縣濁液を前記膜に吸い込ませ、そして、
細胞縣濁液中の細胞を前記膜内に留め、水分は流出させる、
工程を含む、態様;並びに、
(C)前記ポリイミド多孔質膜の片面又は両面を、細胞培養液又は滅菌された液体で湿潤し、
前記湿潤したポリイミド多孔質膜に細胞縣濁液を装填し、そして、
細胞縣濁液中の細胞を前記膜内に留め、水分は流出させる、
工程を含む、態様。
ポリイミド多孔質膜の表面に播種された細胞は、ポリイミド多孔質膜に接着し、多孔の内部に入り込んでいく。好ましくは、特に外部から物理的又は化学的な力を加えなくても、細胞はポリイミド多孔質膜に自発的に接着する。ポリイミド多孔質膜の表面に播種された細胞は、膜の表面及び/又は内部において安定して生育・増殖することが可能である。細胞は生育・増殖する膜の位置に応じて、種々の異なる形態をとりうる。
細胞が凍結する条件としては、ポリイミド多孔質膜に担持された細胞の一部又はすべてが、凍結後に解凍された際に生命機能を保持しているような条件であれば、適宜設定することができる。また、ポリイミド多孔質膜を入れるための凍結用チューブや凍結用ケーン等も、市販されている製品を使用することができる。
第一の低温条件を提供する装置と、第二の低温条件を提供する装置は別個のものでもよいし、同じでもよい。第一の低温条件を提供する装置と、第二の低温条件を提供する装置とが別個のものである場合としては、たとえば第一の低温条件を提供する装置が通常のフリーザーであり、第二の低温条件を提供する装置がディープフリーザーである場合が挙げられるが、これに限定されない。また、第一の低温条件を提供する装置と、第二の低温条件を提供する装置とが同じ場合としては、一定の速度で温度を低下させることができるプログラムフリーザー(たとえばネッパジーン株式会社製プログラムフリーザー等)を用いる場合が挙げられるが、これに限定されない。
本発明の方法は、上述したように細胞を担持させたポリイミド多孔質膜を、凍結状態を保つ条件で保存する工程を含む。凍結状態を保つ条件としては、上記第二の低温条件下でそのまま保存する条件や、第二の低温条件よりさらに低温である第三の低温条件下で保存する条件が挙げられる。第三の低温条件の例としては、たとえば液体窒素中におく場合が挙げられるが、これに限定されない。
本発明の方法では、細胞を担持したポリイミド多孔質膜を凍結保存後に解凍することができる。ポリイミド多孔質膜に担持された細胞を解凍する方法としては、凍結保存されたポリイミド多孔質膜を保存容器の外部から加温し、解凍する方法が挙げられる。加温の方法としては、37℃程度の恒温の温湯に容器を浸す等の方法を適宜用いることができる。
本発明の方法では、細胞を解凍したポリイミド多孔質膜をそのまま細胞培養培地に適用することで、解凍された細胞を同じポリイミド多孔質膜上で培養することができる。
本発明は、細胞の凍結保存のためのポリイミド多孔質膜にも関する。本発明の、細胞の凍結保存のためのポリイミド多孔質膜は、上述した細胞の凍結保存方法のためのポリイミド多孔質膜であってよい。
本発明は、ポリイミド多孔質膜を含む、細胞の凍結保存のためのキットにも関する。
本発明のキットは、ポリイミド多孔質膜の他に、細胞培養に必要な構成要素を適宜含みうる。例えば、ポリイミド多孔質膜、細胞凍結保存液、凍結用チューブ、凍結用ケーン及びキットの取り扱い説明書などが含まれる。
・ヒト線維芽細胞(LONZA社 product code CC-2511)
・Vero細胞(DSファーマバイオメディカル社、cat. DSIU002)
・CHO-K1(パブリックヘルスイングランド cat. 85051005)
・CHO DP-12(Summit Pharmaceuticals Intl CRL-12445)
・ヒト線維芽細胞用培地(LONZA社 product code CC-3132)
・Vero細胞用培地(和光純薬工業株式会社 E-MEM 051-07615)
・CHO-K1用培地(和光純薬工業株式会社 Ham’s F-12 087-08335)
・CHO DP-12用培地(和光純薬工業株式会社 IMDM,-06465)
・3.5cmシャーレ(Falcon社 cat. 353001)
・Cell Counting Kit 8(株式会社同仁化学研究所 CCK8 CK04)
・クライオチューブ(Thermo Fisher Scientific 社 1.8ml cat. 377267)
・2cm×2cmの滅菌された正方形容器(Thermo Fisher Scientific 社 cat. 103)
・セルバンカー(日本全薬工業株式会社 CEllBANKER 1 Plus cat. CB021)
・顕微鏡名、使用した画像ソフト名(Carl Zeiss 社製 LSM 700 使用ソフト ZEN
2cm×2cmの滅菌された正方形容器に細胞培養培地0.5mlを加え、滅菌した1.4cm角の正方形のポリイミド多孔質膜をメッシュ構造のA面もしくは大穴構造のB面を上にしてそれぞれ浸漬させた。1枚のシートあたり5×104個のヒト皮膚線維芽細胞及び、1枚のシートあたり3×105個のヒト皮膚線維芽細胞懸濁液をそれぞれ添加し、1週間に2回の割合で培地交換し、細胞培養を継続的に実施した。これらシートを各5枚ずつ用意し、20cm2シャーレに移動し、4mlの培地を加えて培養を継続した。4日間、7日間、13日間、31日間、56日間、84日間後にCCK8を用いて細胞数を計測し、増殖挙動を観察した。結果を図3に示す。参照例として、平面培養(播種密度:3.5×103個/cm2)で14日までの経過も併記する。
2cm×2cmの滅菌された正方形容器に細胞培養培地0.5mlを加え、滅菌した1.4cm角の正方形のポリイミド多孔質膜をメッシュ構造のA面もしくは大穴構造のB面を上にして浸漬させた。1枚のシートあたり4×104個の細胞を播種し、CO2インキュベータ内で培養を継続的に行った。週2回培地(1ml)を交換した。培養開始後、89日間、118日間、127日間、139日間後にCCK8を用いて細胞数を計測し、細胞生育挙動を観察した。観測期間を通じて、1cm2当り1.5×105個以上の細胞数が観測された。結果を図4に示す。
2cm×2cmの滅菌された正方形容器に細胞培養培地1mlを加え、滅菌した1.4cm角の正方形のポリイミド多孔質膜をメッシュ構造のA面もしくは大穴構造のB面を上にして浸漬させた。1枚のシートあたり4×104個及び1×105個の細胞を播種し、CO2インキュベータ内で培養を継続的に行った。この際、通常使用の厚み25μmのポリイミド多孔質膜に加え、75μmのポリイミド多孔質膜を使用した。また、FBS量も、培地に対して10%もしくは5%の量を使用した。週2回培地(1ml)を交換して、培養を継続した。140日の期間で、CCK8を用いて間歇的に細胞数を計測し、細胞生育挙動を観察した。安定した細胞の生育が観察された。図5に結果を示す。
ヒト皮膚線維芽細胞のポリイミド多孔質膜を用いる長期培養
2cm×2cmの滅菌された正方形容器に2%FBS入細胞培養培地0.5mlを加え、滅菌した1.4cm角の正方形のポリイミド多孔質膜をメッシュ構造のA面を上にしてそれぞれ浸漬させた。1枚のシートあたり4×104個のヒト皮膚線維芽細胞懸濁液をそれぞれ添加し、1週間に2回の割合で培地交換し、細胞培養を継続的に実施した。231日間、261日間、294日間、324日間、365日間、401日間、471日間後にCCK8を用いて細胞数を計測し、増殖挙動を観察した。結果を図8に示す。培養期間を通じて、安定した生細胞数が確認された。
直径6cmのシャーレに2mlの2%FBS入培地を加え、滅菌した1.4cm角の正方形のポリイミド多孔質膜のメッシュ構造のA面に1枚のシートあたり4×104個のヒト皮膚線維芽細胞を播種し、1ヶ月培養した。その後、シートを4分の1に切断し、更に培養を継続して合計230日培養した。その後、3.5cmディッシュ中央に、1.4cm角のステンレスメッシュを3枚重ねて設置し、その上に、前記ポリイミド多孔質膜を置き、滅菌した1.4cm角の空のポリイミド多孔質膜2枚で挟んだ。その状態で培地1mlを加えると、培地は、シートと同様の高さとなった。この状態でCO2インキュベータ内に移動させ、1週間に2回の割合で培地交換して細胞培養を継続的に実施した。培養7日後、各シートを1枚ずつに分け、単一のシートとして培養を継続した。7日、10日、16日、21日、28日、42日、56日後にCCK8を用いて細胞数を計測し、元のシートと後から接地させた空のポリイミド多孔質膜について、細胞の増殖挙動をCCK8での染色法を用いて観察した。ヒト皮膚線維芽細胞を長期間培養したポリイミド多孔質膜から、効率的に細胞が空のポリイミド多孔質膜に移動し、継続的に増殖してゆく挙動が観察された。結果を図9に示す。
2cm×2cmの滅菌された正方形容器に細胞培養培地(2%FBS、IMDM、和光純薬工業株式会社)0.5mlを加え、滅菌した1.4cm角の正方形のポリイミド多孔質膜をメッシュ構造のA面を上にしてそれぞれ浸漬させた。1枚のシートあたり4×104個のヒト抗IL-8産生CHO DP-12細胞懸濁液をそれぞれ培地内シート上に添加し、1週間に2回の割合で培地交換し、細胞培養を継続的に実施した。78日間細胞培養した後、CCK8を用いて細胞数を測定した。
2cm×2cmの滅菌された正方形容器に細胞培養培地0.5mlを加え、滅菌した1.4cm角の正方形のポリイミド多孔質膜をメッシュ構造のA面を上にしてそれぞれ浸漬させた。1枚のシートあたり4×104個のヒト皮膚線維芽細胞懸濁液を培地内シート上にそれぞれ添加し、CO2インキュベータ内で培養を開始した。1週間に2回の割合で培地交換して細胞培養を継続的に実施し、49日間細胞培養した後に、CCK8を用いて細胞数を測定したところ、9.1×104個であった。
Claims (42)
- 細胞の長期培養方法であって、
(1)細胞をポリイミド多孔質膜に適用することと、
(2)細胞を適用したポリイミド多孔質膜を細胞培養培地に適用し、30日以上細胞を培養することと
を含む、方法。 - 工程(2)において、60日以上細胞を培養する、請求項1に記載の方法。
- 工程(2)において、120日以上細胞を培養する、請求項1に記載の方法。
- 2以上のポリイミド多孔質膜を、上下又は左右に細胞培養培地中に積層して用いる、請求項1~3のいずれか一項に記載の方法。
- ポリイミド多孔質膜を
i)折り畳んで、
ii)ロール状に巻き込んで、
iii)シートもしくは小片を糸状の構造体で連結させて、あるいは、
iv)縄状に結んで
細胞培養容器中の細胞培養培地中で浮遊もしくは固定させて用いる、請求項1~4のいずれか一項に記載の方法。 - 工程(2)における培養において、ポリイミド多孔質膜の一部分又は全体が、細胞培養培地の液相と接触していない、請求項1~5のいずれか一項に記載の方法。
- 工程(2)における培養において、細胞培養容器中に含まれる細胞培養培地の総体積が、細胞生存域を含むポリイミド多孔質膜体積の総和の10000倍又はそれより少ない、請求項1~6のいずれか一項に記載の方法。
- 工程(2)における培養において、細胞培養容器中に含まれる細胞培養培地の総体積が、細胞生存域を含むポリイミド多孔質膜体積の総和の100倍又はそれより少ない、請求項1~7のいずれか一項に記載の方法。
- 工程(2)における培養が、細胞培養容器外に設置された細胞培養培地供給手段から連続的又は間歇的に細胞培養培地が細胞培養容器中に供給される系で行われる、請求項1~8のいずれか一項に記載の方法。
- 細胞培養培地が細胞培養培地供給手段と細胞培養容器との間を循環する、請求項9に記載の方法。
- 請求項9又は10に記載の方法であって、前記系が、細胞培養容器である培養ユニットと細胞培養培地供給手段である培地供給ユニットとを含む細胞培養装置であり、ここで
培養ユニットは細胞を担持するための1又は複数のポリイミド多孔質膜を収容する培養ユニットであって、培地供給口および培地排出口を備えた培養ユニットであり、
培地供給ユニットは培地収納容器と、培地供給ラインと、培地供給ラインを介して連続的又は間歇的に培地を送液する送液ポンプとを備え、ここで培地供給ラインの第一の端部は培地収納容器内の培地に接触し、培地供給ラインの第二の端部は培養ユニットの培地供給口を介して培養ユニット内に連通している、培地供給ユニットである、
方法。 - 培養ユニットが空気供給口、空気排出口、及び酸素交換膜を備えない培養ユニットである、請求項13に記載の方法。
- 培養ユニットが培地排出ラインをさらに備え、ここで培地排出ラインの第一の端部は培地収納容器に接続し、培地排出ラインの第二の端部は培養ユニットの培地排出口を介して培養ユニット内に連通し、培地が培地供給ユニットと培養ユニットとを循環可能である、請求項11又は12に記載の方法。
- 細胞が、動物細胞、昆虫細胞、植物細胞、酵母菌及び細菌からなる群から選択される、請求項1~13のいずれかに記載の方法。
- 動物細胞が、脊椎動物門に属する動物由来の細胞である、請求項14に記載の方法。
- 細胞が、CHO細胞、CHO-K1細胞株、CHO DP-12細胞株、CHO細胞関連株、Vero細胞、及びMDCK細胞からなる群より選択される、請求項15に記載の方法。
- ポリイミド多孔質膜が、テトラカルボン酸二無水物とジアミンとから得られるポリイミドを含む、ポリイミド多孔質膜である、請求項1~16のいずれかに記載の方法。
- ポリイミド多孔質膜が、テトラカルボン酸二無水物とジアミンとから得られるポリアミック酸溶液と着色前駆体とを含むポリアミック酸溶液組成物を成形した後、250℃以上で熱処理する事により得られる着色したポリイミド多孔質膜である、請求項17に記載の方法。
- ポリイミド多孔質膜を含む、請求項1~18のいずれかに記載の方法に使用するための細胞培養装置。
- 2以上のポリイミド多孔質膜が、上下又は左右に積層している、請求項19に記載の細胞培養装置。
- ポリイミド多孔質膜を含む、請求項1~13のいずれかに記載の方法に使用するためのキット。
- 細胞の長期培養方法のための、ポリイミド多孔質膜の使用。
- 細胞の凍結保存方法であって、
(1)細胞をポリイミド多孔質膜に担持させる工程、
(2)細胞を担持させたポリイミド多孔質膜を、細胞が凍結する条件下に置くことで、ポリイミド多孔質膜に担持された細胞を凍結する工程、及び
(3)細胞を担持させたポリイミド多孔質膜を、凍結状態を保つ条件で保存する工程
を含む、方法。 - 工程(1)において、細胞をポリイミド多孔質膜に播種することにより、細胞をポリイミド多孔質膜に担持させる、請求項23に記載の方法。
- 工程(1)において、細胞をポリイミド多孔質膜に播種して培養することにより、細胞をポリイミド多孔質膜に担持させる、請求項23に記載の方法。
- 工程(3)に続いて、
(4)細胞を担持させたポリイミド多孔質膜を、細胞が解凍する条件下に置くことで、ポリイミド多孔質膜に担持された細胞を解凍する工程
をさらに含む、請求項23~25のいずれか一項に記載の方法。 - 工程(4)に続いて、
(5)細胞を解凍したポリイミド多孔質膜を細胞培養培地に適用し、細胞を培養する工程
をさらに含む、請求項26に記載の方法。 - 工程(5)において、培養した細胞がポリイミド多孔質膜外においても増殖するまで細胞を培養する、請求項27に記載の方法。
- 工程(5)において、細胞を解凍したポリイミド多孔質膜と共に、細胞を担持させていない別の1又は複数のポリイミド多孔質膜を細胞培養培地に適用し、培養することにより、別の1又は複数のポリイミド多孔質膜に細胞を担持させる、請求項27又は28に記載の方法。
- 工程(5)に続いて、
(6)細胞を担持させたポリイミド多孔質膜の一部又はすべてを、細胞が凍結する条件下に置くことで、ポリイミド多孔質膜に担持された細胞を凍結する工程、及び
(7)細胞を担持させたポリイミド多孔質膜を、凍結状態を保つ条件で保存する工程
をさらに含む、請求項27~29のいずれか一項に記載の方法。 - 工程(1)~(7)を複数回繰り返す、請求項30に記載の方法。
- 細胞が、動物細胞、昆虫細胞、植物細胞、酵母菌及び細菌からなる群から選択される、請求項23~31のいずれか一項に記載の方法。
- 動物細胞が、脊椎動物門に属する動物由来の細胞である、請求項32に記載の方法。
- 細胞が付着性細胞である、請求項32又は33に記載の方法。
- 細胞が浮遊性細胞である、請求項32又は33に記載の方法。
- ポリイミド多孔質膜が、テトラカルボン酸二無水物とジアミンとから得られるポリイミドを含む、ポリイミド多孔質膜である、請求項23~25のいずれか一項に記載の方法。
- ポリイミド多孔質膜が、テトラカルボン酸二無水物とジアミンとから得られるポリアミック酸溶液と着色前駆体とを含むポリアミック酸溶液組成物を成形した後、250℃以上で熱処理する事により得られる着色したポリイミド多孔質膜である、請求項36に記載の方法。
- ポリイミド多孔質膜が、2つの異なる表層面とマクロボイド層を有する多層構造のポリイミド多孔質膜である、請求項36又は37に記載の方法。
- 細胞の凍結保存のためのポリイミド多孔質膜。
- 請求項23~38のいずれか一項に記載の方法に用いるためのポリイミド多孔質膜。
- ポリイミド多孔質膜を含む、請求項23~38のいずれか一項に記載の方法に用いるためのキット。
- 請求項23~38のいずれか一項に記載の方法のためのポリイミド多孔質膜の使用。
Priority Applications (11)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP16743370.5A EP3252145A4 (en) | 2015-01-26 | 2016-01-26 | Long-term cell-cultivation using polyimide porous membrane and cell-cryopreservation method using polyimide porous membrane |
| SG11201706009PA SG11201706009PA (en) | 2015-01-26 | 2016-01-26 | Long-term cell-cultivation using polyimide porous membrane and cell-cryopreservation method using polyimide porous membrane |
| BR112017015130-8A BR112017015130A2 (ja) | 2015-01-26 | 2016-01-26 | Long-term culture in a cell using a polyimide porous membrane, and a freeze preserving method of a cell using a polyimide porous membrane |
| JP2016572068A JP6292323B2 (ja) | 2015-01-26 | 2016-01-26 | ポリイミド多孔質膜を用いる細胞の長期培養、及びポリイミド多孔質膜を用いる細胞の凍結保存方法 |
| CA2974276A CA2974276C (en) | 2015-01-26 | 2016-01-26 | Long-term cell-cultivation using polyimide porous membrane and cell-cryopreservation method using polyimide porous membrane |
| CN201680007125.4A CN107208046B (zh) | 2015-01-26 | 2016-01-26 | 使用聚酰亚胺多孔膜的细胞的长期培养、与使用聚酰亚胺多孔膜的细胞的冷冻保存方法 |
| US15/545,252 US10626365B2 (en) | 2015-01-26 | 2016-01-26 | Long-term cell-cultivation using polyimide porous membrane and cell-cryopreservation method using polyimide porous membrane |
| EP18155384.3A EP3406707A1 (en) | 2015-01-26 | 2016-01-26 | Long-term cell-cultivation using polyimide porous membrane and cell-cryopreservation method using polyimide porous membrane |
| CN202110140236.5A CN113142187B (zh) | 2015-01-26 | 2016-01-26 | 使用聚酰亚胺多孔膜的细胞的长期培养及冷冻保存方法 |
| KR1020177020771A KR102000381B1 (ko) | 2015-01-26 | 2016-01-26 | 폴리이미드 다공질막을 이용하는 세포의 장기 배양, 및 폴리이미드 다공질막을 이용하는 세포의 동결 보존 방법 |
| US15/880,457 US10443037B2 (en) | 2015-01-26 | 2018-01-25 | Long-term cell-cultivation using polyimide porous membrane and cell-cryopreservation method using polyimide porous membrane |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015-012810 | 2015-01-26 | ||
| JP2015012810 | 2015-01-26 | ||
| JP2015-012537 | 2015-01-26 | ||
| JP2015012537 | 2015-01-26 |
Related Child Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US15/545,252 A-371-Of-International US10626365B2 (en) | 2015-01-26 | 2016-01-26 | Long-term cell-cultivation using polyimide porous membrane and cell-cryopreservation method using polyimide porous membrane |
| EP18155384.3A Previously-Filed-Application EP3406707A1 (en) | 2015-01-26 | 2016-01-26 | Long-term cell-cultivation using polyimide porous membrane and cell-cryopreservation method using polyimide porous membrane |
| US15/880,457 Division US10443037B2 (en) | 2015-01-26 | 2018-01-25 | Long-term cell-cultivation using polyimide porous membrane and cell-cryopreservation method using polyimide porous membrane |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016121767A1 true WO2016121767A1 (ja) | 2016-08-04 |
Family
ID=56543378
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2016/052206 Ceased WO2016121767A1 (ja) | 2015-01-26 | 2016-01-26 | ポリイミド多孔質膜を用いる細胞の長期培養、及びポリイミド多孔質膜を用いる細胞の凍結保存方法 |
Country Status (9)
| Country | Link |
|---|---|
| US (2) | US10626365B2 (ja) |
| EP (2) | EP3406707A1 (ja) |
| JP (1) | JP6292323B2 (ja) |
| KR (1) | KR102000381B1 (ja) |
| CN (2) | CN107208046B (ja) |
| BR (1) | BR112017015130A2 (ja) |
| CA (1) | CA2974276C (ja) |
| SG (1) | SG11201706009PA (ja) |
| WO (1) | WO2016121767A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2020067434A1 (ja) * | 2018-09-27 | 2020-04-02 | テルモ株式会社 | 細胞の凍結保存方法 |
| WO2024257852A1 (ja) | 2023-06-15 | 2024-12-19 | Ube株式会社 | 細胞の凍結保存方法、細胞培養方法および凍結保存基材 |
| US12284955B2 (en) | 2021-08-05 | 2025-04-29 | W. L. Gore & Associates, Inc. | Composite materials promoting the catchment and attachment of seaweed holdfasts |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN112940940B (zh) | 2015-01-26 | 2024-03-15 | Ube株式会社 | 细胞的培养方法及试剂盒 |
| CN107208058B (zh) * | 2015-01-26 | 2021-06-25 | 宇部兴产株式会社 | 利用骨髓类似结构的细胞培养方法、和用于骨损伤部位的治疗的聚酰亚胺多孔膜 |
| WO2016121767A1 (ja) | 2015-01-26 | 2016-08-04 | 宇部興産株式会社 | ポリイミド多孔質膜を用いる細胞の長期培養、及びポリイミド多孔質膜を用いる細胞の凍結保存方法 |
| SG11201900674XA (en) | 2016-07-25 | 2019-02-27 | Ube Industries | Cell culture module |
| WO2018231993A1 (en) * | 2017-06-14 | 2018-12-20 | New York University | System and method for paper-based cryopreservation |
| EP3989716A1 (en) * | 2019-06-27 | 2022-05-04 | W.L. Gore & Associates, Inc. | Biointerfaces for growing seaweed |
| JP7490668B2 (ja) * | 2019-10-25 | 2024-05-27 | 富士フイルム株式会社 | 細胞培養用基材及び細胞付き細胞培養用基材 |
| CN113248748A (zh) * | 2020-02-12 | 2021-08-13 | 精拓生技股份有限公司 | 用于体外扩增循环肿瘤细胞的培养容器的复合材料薄膜的制备方法 |
| CN114081995B (zh) * | 2021-11-30 | 2022-09-27 | 广东工业大学 | 一种以聚酰亚胺为基体的硬组织修复材料及其制备方法和应用 |
Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS63196286A (ja) * | 1987-02-12 | 1988-08-15 | Sumitomo Electric Ind Ltd | 細胞培養用基材 |
| JPS63198975A (ja) * | 1987-02-13 | 1988-08-17 | Sumitomo Electric Ind Ltd | 細胞培養用基材 |
| JPS63198978A (ja) * | 1987-02-13 | 1988-08-17 | Sumitomo Electric Ind Ltd | 細胞培養用基材 |
| JP2003524404A (ja) * | 1999-05-19 | 2003-08-19 | ソニー インターナショナル (ヨーロッパ) ゲゼルシャフト ミット ベシュレンクテル ハフツング | 配向した軸索突起外殖のための基質構造の構成方法、基質構造、及び神経単位の監視装置 |
| JP2004173563A (ja) * | 2002-11-26 | 2004-06-24 | Applied Cell Biotechnologies Inc | ヒト細胞培養に関する方法 |
| JP2005536233A (ja) * | 2002-08-19 | 2005-12-02 | バイオプロセッサーズ コーポレイション | リアクター環境条件の決定および/または制御 |
| WO2010038873A1 (ja) * | 2008-10-02 | 2010-04-08 | 宇部興産株式会社 | 多孔質ポリイミド膜及びその製造方法 |
| JP2011172533A (ja) * | 2010-02-25 | 2011-09-08 | Fusao Komada | マイクロ空間構造体を用いた高密度三次元細胞培養法 |
| JP2011219585A (ja) * | 2010-04-07 | 2011-11-04 | Ube Industries Ltd | 多孔質ポリイミド膜及びその製造方法 |
| WO2015012415A1 (ja) * | 2013-07-26 | 2015-01-29 | 宇部興産株式会社 | 細胞の培養方法、細胞培養装置及びキット |
Family Cites Families (26)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AT293313B (de) | 1970-06-01 | 1971-10-11 | Jungbunzlauer Spiritus | Verfahren zur Herstellung von Zitronensäure durch submerse Gärung |
| JPS5831635B2 (ja) | 1974-10-02 | 1983-07-07 | 東京電力株式会社 | ランダムデ−タジユシンホウシキ |
| US4228243A (en) * | 1978-07-13 | 1980-10-14 | Toray Industries, Inc. | Cell culture propagation apparatus |
| JPH0630570B2 (ja) | 1985-09-18 | 1994-04-27 | 株式会社ミドリ十字 | 付着性細胞の連続大量培養法 |
| JPS63196271A (ja) * | 1987-02-10 | 1988-08-15 | Sumitomo Electric Ind Ltd | 細胞培養用基材 |
| JPS63196274A (ja) * | 1987-02-10 | 1988-08-15 | Sumitomo Electric Ind Ltd | 細胞培養用基材 |
| JPS63196272A (ja) * | 1987-02-10 | 1988-08-15 | Sumitomo Electric Ind Ltd | 細胞培養用基材 |
| JP2965391B2 (ja) | 1991-06-07 | 1999-10-18 | アルプス電気株式会社 | 圧電モータ |
| JPH05123168A (ja) | 1991-10-30 | 1993-05-21 | Green Cross Corp:The | 細胞長期培養方法 |
| JPH10108673A (ja) | 1996-10-04 | 1998-04-28 | Asahi Medical Co Ltd | 中空糸型培養器を用いた動物細胞の培養方法 |
| JP2002034551A (ja) | 2000-07-18 | 2002-02-05 | Sumitomo Bakelite Co Ltd | 細胞付細胞培養器の製造方法および使用方法 |
| US20040058437A1 (en) | 2001-04-10 | 2004-03-25 | Rodgers Seth T. | Materials and reactor systems having humidity and gas control |
| JP2003047464A (ja) | 2001-08-02 | 2003-02-18 | Institute Of Tsukuba Liaison Co Ltd | 動物細胞の保存方法 |
| US20050265980A1 (en) | 2004-05-14 | 2005-12-01 | Becton, Dickinson And Company | Cell culture environments for the serum-free expansion of mesenchymal stem cells |
| TW200708304A (en) | 2005-03-25 | 2007-03-01 | Nipro Corp | Package of freeze storage container and process for producing the same |
| GB0512214D0 (en) * | 2005-06-15 | 2005-07-27 | Capsant Neurotechnologies Ltd | Method |
| JP5464443B2 (ja) | 2008-03-31 | 2014-04-09 | オリエンタル酵母工業株式会社 | 多能性幹細胞を増殖させる方法 |
| JP5140155B2 (ja) | 2008-06-24 | 2013-02-06 | 独立行政法人海洋研究開発機構 | 動物細胞の凍結保存用担体、それを用いた凍結保存用バイオデバイス及び凍結保存方法 |
| JP5720574B2 (ja) * | 2009-10-09 | 2015-05-20 | 宇部興産株式会社 | 着色ポリイミド成形体及びその製造方法 |
| WO2011125988A1 (ja) * | 2010-04-07 | 2011-10-13 | 宇部興産株式会社 | 多孔質ポリイミド膜及びその製造方法 |
| JP5577804B2 (ja) | 2010-04-07 | 2014-08-27 | 宇部興産株式会社 | 多孔質ポリイミド膜及びその製造方法 |
| US8366804B2 (en) * | 2010-05-28 | 2013-02-05 | Uop Llc | High permeance polyimide membranes for air separation |
| CN103710263B (zh) * | 2012-09-28 | 2015-05-27 | 江苏吉锐生物技术有限公司 | 细胞培养装置 |
| JP6343492B2 (ja) * | 2013-07-09 | 2018-06-13 | 豊田合成株式会社 | 細胞培養用および癌細胞増殖抑制用の少なくとも一方の用途に用いるポリウレタン多孔質膜の製造方法 |
| CN204079987U (zh) * | 2014-07-04 | 2015-01-07 | 赫柏慧康生物科技无锡有限公司 | 一种新型细胞片层培养皿 |
| WO2016121767A1 (ja) | 2015-01-26 | 2016-08-04 | 宇部興産株式会社 | ポリイミド多孔質膜を用いる細胞の長期培養、及びポリイミド多孔質膜を用いる細胞の凍結保存方法 |
-
2016
- 2016-01-26 WO PCT/JP2016/052206 patent/WO2016121767A1/ja not_active Ceased
- 2016-01-26 CN CN201680007125.4A patent/CN107208046B/zh active Active
- 2016-01-26 EP EP18155384.3A patent/EP3406707A1/en active Pending
- 2016-01-26 US US15/545,252 patent/US10626365B2/en active Active
- 2016-01-26 BR BR112017015130-8A patent/BR112017015130A2/ja not_active IP Right Cessation
- 2016-01-26 KR KR1020177020771A patent/KR102000381B1/ko active Active
- 2016-01-26 JP JP2016572068A patent/JP6292323B2/ja active Active
- 2016-01-26 CA CA2974276A patent/CA2974276C/en active Active
- 2016-01-26 CN CN202110140236.5A patent/CN113142187B/zh active Active
- 2016-01-26 EP EP16743370.5A patent/EP3252145A4/en active Pending
- 2016-01-26 SG SG11201706009PA patent/SG11201706009PA/en unknown
-
2018
- 2018-01-25 US US15/880,457 patent/US10443037B2/en active Active
Patent Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS63196286A (ja) * | 1987-02-12 | 1988-08-15 | Sumitomo Electric Ind Ltd | 細胞培養用基材 |
| JPS63198975A (ja) * | 1987-02-13 | 1988-08-17 | Sumitomo Electric Ind Ltd | 細胞培養用基材 |
| JPS63198978A (ja) * | 1987-02-13 | 1988-08-17 | Sumitomo Electric Ind Ltd | 細胞培養用基材 |
| JP2003524404A (ja) * | 1999-05-19 | 2003-08-19 | ソニー インターナショナル (ヨーロッパ) ゲゼルシャフト ミット ベシュレンクテル ハフツング | 配向した軸索突起外殖のための基質構造の構成方法、基質構造、及び神経単位の監視装置 |
| JP2005536233A (ja) * | 2002-08-19 | 2005-12-02 | バイオプロセッサーズ コーポレイション | リアクター環境条件の決定および/または制御 |
| JP2004173563A (ja) * | 2002-11-26 | 2004-06-24 | Applied Cell Biotechnologies Inc | ヒト細胞培養に関する方法 |
| WO2010038873A1 (ja) * | 2008-10-02 | 2010-04-08 | 宇部興産株式会社 | 多孔質ポリイミド膜及びその製造方法 |
| JP2011172533A (ja) * | 2010-02-25 | 2011-09-08 | Fusao Komada | マイクロ空間構造体を用いた高密度三次元細胞培養法 |
| JP2011219585A (ja) * | 2010-04-07 | 2011-11-04 | Ube Industries Ltd | 多孔質ポリイミド膜及びその製造方法 |
| WO2015012415A1 (ja) * | 2013-07-26 | 2015-01-29 | 宇部興産株式会社 | 細胞の培養方法、細胞培養装置及びキット |
Non-Patent Citations (5)
| Title |
|---|
| HIROYOSHI KAWAKAMI: "Cell Culture on Nano- or Micro-relief pattern Surface", MEMBRANE, vol. 32, no. 5, 2007, pages 266 - 270, XP009503429, ISSN: 0385-1036 * |
| JULIEN, S. ET AL.: "Implantation of ultrathin, biofunctionalized polyimide membranes into the subretinal space of rats", BIOMATERIALS, vol. 32, 2011, pages 3890 - 3898, XP028370852, ISSN: 0142-9612 * |
| See also references of EP3252145A4 * |
| SEIFERT, B. ET AL.: "Polyetherimide: a new membrane-forming polymer for biomedical applications", ARTIFICIAL ORGANS, vol. 26, no. 2, 2002, pages 189 - 199, XP055131039, ISSN: 0160-564X * |
| TAO, C-T ET AL.: "Polyetherimide membrane formation by the cononsolvent system and its biocompatibility of MG 63 cell line", JOURNAL OF MEMBRANE SCIENCE, vol. 269, 2006, pages 66 - 74, XP024931301, ISSN: 0376-7388 * |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2020067434A1 (ja) * | 2018-09-27 | 2020-04-02 | テルモ株式会社 | 細胞の凍結保存方法 |
| US12284955B2 (en) | 2021-08-05 | 2025-04-29 | W. L. Gore & Associates, Inc. | Composite materials promoting the catchment and attachment of seaweed holdfasts |
| WO2024257852A1 (ja) | 2023-06-15 | 2024-12-19 | Ube株式会社 | 細胞の凍結保存方法、細胞培養方法および凍結保存基材 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN107208046A (zh) | 2017-09-26 |
| EP3406707A1 (en) | 2018-11-28 |
| US10443037B2 (en) | 2019-10-15 |
| BR112017015130A2 (ja) | 2018-01-23 |
| SG11201706009PA (en) | 2017-08-30 |
| CA2974276C (en) | 2019-07-30 |
| US10626365B2 (en) | 2020-04-21 |
| JPWO2016121767A1 (ja) | 2017-11-24 |
| CA2974276A1 (en) | 2016-08-04 |
| CN113142187B (zh) | 2022-07-01 |
| EP3252145A1 (en) | 2017-12-06 |
| KR20170093250A (ko) | 2017-08-14 |
| EP3252145A4 (en) | 2018-10-03 |
| CN107208046B (zh) | 2021-03-05 |
| KR102000381B1 (ko) | 2019-07-15 |
| CN113142187A (zh) | 2021-07-23 |
| US20180208887A1 (en) | 2018-07-26 |
| US20170369838A1 (en) | 2017-12-28 |
| JP6292323B2 (ja) | 2018-03-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6292323B2 (ja) | ポリイミド多孔質膜を用いる細胞の長期培養、及びポリイミド多孔質膜を用いる細胞の凍結保存方法 | |
| JP6288312B2 (ja) | ポリイミド多孔質膜を用いる細胞の大量培養方法、装置及びキット | |
| JP6225993B2 (ja) | 細胞の培養方法、細胞培養装置及びキット | |
| JP6502392B2 (ja) | 細胞の培養方法及びキット | |
| JP6288311B2 (ja) | 物質の産生方法 | |
| WO2018021362A1 (ja) | 脱分化しやすい細胞の脱分化を抑制する方法、当該細胞の調製方法、及び物質の産生方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 16743370 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2016572068 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2974276 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15545252 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11201706009P Country of ref document: SG |
|
| ENP | Entry into the national phase |
Ref document number: 20177020771 Country of ref document: KR Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2016743370 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112017015130 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 112017015130 Country of ref document: BR Kind code of ref document: A2 Effective date: 20170714 |